Executive Summary
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Role of Nanobiosensors in Therapeutic Drug Monitoring
Journal of Personalized Medicine Review Personalized Medicine for Antibiotics: The Role of Nanobiosensors in Therapeutic Drug Monitoring Vivian Garzón 1, Rosa-Helena Bustos 2 and Daniel G. Pinacho 2,* 1 PhD Biosciences Program, Universidad de La Sabana, Chía 140013, Colombia; [email protected] 2 Therapeutical Evidence Group, Clinical Pharmacology, Universidad de La Sabana, Chía 140013, Colombia; [email protected] * Correspondence: [email protected]; Tel.: +57-1-8615555 (ext. 23309) Received: 21 August 2020; Accepted: 7 September 2020; Published: 25 September 2020 Abstract: Due to the high bacterial resistance to antibiotics (AB), it has become necessary to adjust the dose aimed at personalized medicine by means of therapeutic drug monitoring (TDM). TDM is a fundamental tool for measuring the concentration of drugs that have a limited or highly toxic dose in different body fluids, such as blood, plasma, serum, and urine, among others. Using different techniques that allow for the pharmacokinetic (PK) and pharmacodynamic (PD) analysis of the drug, TDM can reduce the risks inherent in treatment. Among these techniques, nanotechnology focused on biosensors, which are relevant due to their versatility, sensitivity, specificity, and low cost. They provide results in real time, using an element for biological recognition coupled to a signal transducer. This review describes recent advances in the quantification of AB using biosensors with a focus on TDM as a fundamental aspect of personalized medicine. Keywords: biosensors; therapeutic drug monitoring (TDM), antibiotic; personalized medicine 1. Introduction The discovery of antibiotics (AB) ushered in a new era of progress in controlling bacterial infections in human health, agriculture, and livestock [1] However, the use of AB has been challenged due to the appearance of multi-resistant bacteria (MDR), which have increased significantly in recent years due to AB mismanagement and have become a global public health problem [2]. -
2002 FSIS National Residue Program, Section 4
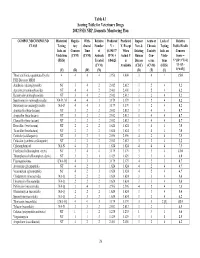
Table 4.1 Scoring Table for Veterinary Drugs 2002 FSIS NRP, Domestic Monitoring Plan COMPOUND/COMPOUND Historical Regula- With- Relative Predicted Predicted Impact Acute or Lack of Relative CLASS Testing tory drawal Number V = V, Except New & Chronic Testing Public Health Info. on Concern Time of (0.19437* When Existing Toxicity Info. on Concern Violations (CVM) (CVM) Animals R*N) + Actual V Human Con- Viola- Score = (FSIS) Treated 0.84625 is Disease cerns tions V*[(D+3*T)/4] (CVM) Available (CDC) (CVM) (FSIS) *{1+[(L- (V) (R) (W) (N) (D) (T) (L) 1)*0.05]} Those antibiotics quantitated by the 4 4 4 4 3.956 4.000 3 4 1 15.0 FSIS Bioassay MRM Amikacin (aminoglycoside) NT 3 4 2 2.012 2.012 3 2 4 5.2 Apramycin (aminoglycoside) NT 4 4 2 2.401 2.401 3 2 4 6.2 Kanamycin (aminoglycoside) NT 3 4 2 2.012 2.012 3 2 4 5.2 Spectinomycin (aminoglycoside) NA-D, M 4 4 3 3.179 3.179 3 2 4 8.2 Streptomycin (aminoglycoside) NA-D 4 4 3 3.179 3.179 3 2 4 8.2 Amoxicillin (beta-lactam) NT 3 2 2 2.012 2.012 3 4 4 8.7 Ampicillin (beta-lactam) NT 3 2 2 2.012 2.012 3 4 4 8.7 Cloxacillin (beta-lactam) NT 3 2 2 2.012 2.012 3 4 4 8.7 Hetacillin (beta-lactam) NT 2 2 2 1.624 1.624 3 4 4 7.0 Ticarcillin (beta-lactam) NT 2 2 2 1.624 1.624 3 4 4 7.0 Ceftiofur (cefalosporin) NT 3 2 3 2.596 2.596 4 2 4 7.5 Cefazolin (synthetic cefalosporin) NT 3 2 2 2.012 2.012 3 2 4 5.2 Chloramphenicol NA-N 4 2 1 1.624 1.624 4 4 4 7.5 Florfenicol (chloramphen. -

(12) United States Patent (10) Patent N0.: US 8,343,962 B2 Kisak Et Al
US008343962B2 (12) United States Patent (10) Patent N0.: US 8,343,962 B2 Kisak et al. (45) Date of Patent: *Jan. 1, 2013 (54) TOPICAL FORMULATION (58) Field of Classi?cation Search ............. .. 514/226.5, 514/334, 420, 557, 567 (75) Inventors: Edward T. Kisak, San Diego, CA (US); See application ?le fOr Complete Search history. John M. NeWsam, La Jolla, CA (US); _ Dominic King-Smith, San Diego, CA (56) References C‘ted (US); Pankaj Karande, Troy, NY (US); Samir Mitragotri, Goleta, CA (US) US' PATENT DOCUMENTS 5,602,183 A 2/1997 Martin et al. (73) Assignee: NuvoResearchOntano (CA) Inc., Mississagua, 6,328,979 2B1 12/2001 Yamashita et a1. 7,001,592 B1 2/2006 Traynor et a1. ( * ) Notice: Subject to any disclaimer, the term of this 7,795,309 B2 9/2010 Kisak eta1~ patent is extended or adjusted under 35 2002/0064524 A1 5/2002 Cevc U.S.C. 154(b) by 212 days. FOREIGN PATENT DOCUMENTS This patent is subject to a terminal dis- W0 WO 2005/009510 2/2005 claimer- OTHER PUBLICATIONS (21) APPI' NO‘, 12/848,792 International Search Report issued on Aug. 8, 2008 in application No. PCT/lB2007/0l983 (corresponding to US 7,795,309). _ Notice ofAlloWance issued on Apr. 29, 2010 by the Examiner in US. (22) Med Aug- 2’ 2010 Appl. No. 12/281,561 (US 7,795,309). _ _ _ Of?ce Action issued on Dec. 30, 2009 by the Examiner in US. Appl. (65) Prior Publication Data No, 12/281,561 (Us 7,795,309), Us 2011/0028460 A1 Feb‘ 3’ 2011 Primary Examiner * Raymond Henley, 111 Related U 5 Application Data (74) Attorney, Agent, or Firm * Foley & Lardner LLP (63) Continuation-in-part of application No. -

Allosteric Drug Transport Mechanism of Multidrug Transporter Acrb
ARTICLE https://doi.org/10.1038/s41467-021-24151-3 OPEN Allosteric drug transport mechanism of multidrug transporter AcrB ✉ Heng-Keat Tam 1,3,4 , Wuen Ee Foong 1,4, Christine Oswald1,2, Andrea Herrmann1, Hui Zeng1 & ✉ Klaas M. Pos 1 Gram-negative bacteria maintain an intrinsic resistance mechanism against entry of noxious compounds by utilizing highly efficient efflux pumps. The E. coli AcrAB-TolC drug efflux pump + 1234567890():,; contains the inner membrane H /drug antiporter AcrB comprising three functionally inter- dependent protomers, cycling consecutively through the loose (L), tight (T) and open (O) state during cooperative catalysis. Here, we present 13 X-ray structures of AcrB in inter- mediate states of the transport cycle. Structure-based mutational analysis combined with drug susceptibility assays indicate that drugs are guided through dedicated transport chan- nels toward the drug binding pockets. A co-structure obtained in the combined presence of erythromycin, linezolid, oxacillin and fusidic acid shows binding of fusidic acid deeply inside the T protomer transmembrane domain. Thiol cross-link substrate protection assays indicate that this transmembrane domain-binding site can also accommodate oxacillin or novobiocin but not erythromycin or linezolid. AcrB-mediated drug transport is suggested to be allos- terically modulated in presence of multiple drugs. 1 Institute of Biochemistry, Goethe-University Frankfurt, Frankfurt am Main, Germany. 2 Sosei Heptares, Steinmetz Building, Granta Park, Great Abington, Cambridge, UK. 3Present -

Coccidiosis in Large and Small Ruminants
Coccidiosis in Large and Small Ruminants a, b Sarah Tammy Nicole Keeton, PhD, MS *, Christine B. Navarre, DVM, MS KEYWORDS Coccidia Coccidiosis Diarrhea Ruminants Cattle Sheep Goats Ionophores KEY POINTS Coccidiosis is an important parasitic disease of ruminant livestock caused by the proto- zoan parasite of the genus Eimeria. Calves between 6 and 12 months of age and lambs and kids between 1 and 6 months of age are most susceptible. Subclinical disease is characterized by poor growth. Clinical disease is most commonly characterized by diarrhea. Control of coccidiosis is based on sound management, the use of preventive medications, and treatment of clinical cases as necessary. INTRODUCTION: NATURE OF THE PROBLEM Coccidiosis is a parasitic disease of vertebrate animals, including domestic ruminants.1 It is economically significant, with losses from both clinical and subclinical disease. Coccidiosis is caused by the protozoan parasite of the genus Eimeria. Eimeria are host specific, meaning that an Eimeria species that infect goats does not infect sheep or cattle and vice versa. Certain species of Eimeria are nonpathogenic and do not cause disease. The pathogenic species and sites of infection are listed in Table 1. Mixed infections with multiple pathogenic and nonpathogenic species is common. LIFE CYCLE Proper treatment and control of coccidiosis requires an understanding of the complex life cycle and transmission of Eimeria spp (Fig. 1). The life cycle can be divided into Disclosure: The authors have nothing to disclose. a Department of Veterinary Clinical Sciences, School of Veterinary Medicine, Louisiana State University, Skip Bertman Drive, Baton Rouge, LA 70803, USA; b LSU AgCenter, School of Animal Sciences, Louisiana State University, 111 Dalrymple Bldg, 110 LSU Union Square, Baton Rouge, LA 70803-0106, USA * Corresponding author. -

Microbiological Profile of Telithromycin, the First Ketolide Antimicrobial
View metadata, citation and similar papers at core.ac.uk brought to you by CORE Paperprovided by 48Elsevier Disc - Publisher Connector Microbiological profile of telithromycin, the first ketolide antimicrobial D. Felmingham GR Micro Ltd, London, UK ABSTRACT Telithromycin, the first of the ketolide antimicrobials, has been specifically designed to provide potent activity against common and atypical/intracellular or cell-associated respiratory pathogens, including those that are resistant to b-lactams and/or macrolide±lincosamide±streptograminB (MLSB) antimicrobials. Against Gram- positive cocci, telithromycin possesses more potent activity in vitro and in vivo than the macrolides clarithromycin and azithromycin. It retains its activity against erm-(MLSB)ormef-mediated macrolide-resistant Streptococcus pneumoniae and Streptococcus pyogenes and against Staphylococcus aureus resistant to macrolides through inducible MLSB mechanisms. Telithromycin also possesses high activity against the Gram-negative pathogens Haemophilus influenzae and Moraxella catarrhalis, regardless of b-lactamase production. In vitro, it shows similar activity to azithromycin against H. influenzae, while in vivo its activity against H. influenzae is higher than that of azithromycin. Telithromycin's spectrum of activity also extends to the atypical, intracellular and cell-associated pathogens Legionella pneumophila, Mycoplasma pneumoniae and Chlamydia pneumoniae. In vitro, telithromycin does not induce MLSB resistance and it shows low potential to select for resistance or cross-resistance to other antimicrobials. These characteristics indicate that telithromycin will have an important clinical role in the Ahed empirical treatment of community-acquired respiratory tract infections. Bhed Clin Microbiol Infect 2001: 7 (Supplement 3): 2±10 Ched Dhed Ref marker Fig marker these agents. Resistance to penicillin, particularly among S. -

Nitrofurazone
PATIENT INFORMATION SHEET Nitrofurazone (N-005) Your patch testing results indicate that you have a contact allergy to Nitrofurazone . It is important that you familiarize yourself with this chemical and take steps to avoid coming in contact with it. i What is Nitrofurazone and where is it found? This chemical is used as a topical bactericide for surface infections. If is also used in anti ‐infective therapy for second and third degree burns and in skin grafting. It is also an antibacterial agent for the treatment or prevention of infections in a variety of conditions involving skin, eyes, ears, nose and genitourinary tract. It is used on pyodermas, ulcers and wounds. Further research may identify additional product or industrial usages of this chemical. i What else is Nitrofurazone called? This chemical can be identified by different names, including: 2‐[(5 ‐Nitro ‐2‐furanyl)methylene]‐hydrazinecarboxamide 5‐nitro ‐2‐furfuraldehyde semicarbazone (5 ‐nitro ‐2‐furfurylideneamino)urea 5‐nitrofurfural semicarbazone 5‐Nitro ‐2‐furaldehyde Semicarbazone 6‐nitrofuraldehyde semicarbazide 5‐nitrofuraldehyde semicarbazide NF 5‐nitrofuran ‐2‐aldehyde semicarbazone NF ‐7 5‐nitro ‐2‐furancarboxaldehyde semicarbazone NFS This may not be a complete list as manufacturers introduce and delete chemicals from their product lines. THINGS YOU CAN DO TO HELP MANAGE YOUR CONTACT ALLERGY Be vigilant read the product label. Always take the time to read the ingredient listing on product packages. This should be your first step each time you purchase a product as manufacturers sometimes change product ingredients. If you have any concerns ask your pharmacist or your doctor. Test the product first. -

Combination of Minocycline and Rifampicin Against Methicillin- and Gentamicin-Resistant Staphylococcus Aureus
J Clin Pathol: first published as 10.1136/jcp.34.5.559 on 1 May 1981. Downloaded from J Clin Pathol 1981 ;34:559-563 Combination of minocycline and rifampicin against methicillin- and gentamicin-resistant Staphylococcus aureus E YOURASSOWSKY, MP VAN DER LINDEN, MJ LISMONT, F CROKAERT From the H6pital Universitaire Brugmann, Service de Biologie Clinique, 1020 Bruxelles, Belgique SUMMARY Methicillin- and gentamicin-resistant Staphylococcus aureus may remain sensitive to minocycline and to rifampicin. A study of growth curves has shown that at inhibitory concentrations (0-4 ,ug/ml), minocycline prevents the development of mutants resistant to rifampicin. Staphylococcus aureus resistant to methicillin and strains of different phage type were selected for this gentamicin is responsible for an increasing number of investigation. hospital infections, some of which are severe.1-7 A number of treatments have been suggested although Microbial strains vancomycin is often the only major antibiotic which Minocycline HCL (Cyanamid Benelux, batch no is active against these strains. However, it is necessary 7116B-172). Rifampicin (Lepetit, batch no P/4) copyright. to assess the effect of the "second choice" antibiotics. (solution in dimethyl formamide). The MICs of The risk of rapid development of resistance to minocycline (tube dilution method in Mueller Hinton rifampicin is well known,8 9 and in spite of the medium, inoculum 106 micro-organisms/ml) were excellent penetration particularly in polymor- 0-2 ,ug/ml for all the strains. Rifampicin showed phonuclear cells of this antibiotic,10 11 its use alone is minimal inhibitory activity in liquid medium up to contraindicated. Minocycline is active against Staph a concentration of 0-01 ,ug/ml. -

Development of a Canine Coccidiosis Model and the Anticoccidial Effects of a New Chemotherapeutic Agent
DEVELOPMENT OF A CANINE COCCIDIOSIS MODEL AND THE ANTICOCCIDIAL EFFECTS OF A NEW CHEMOTHERAPEUTIC AGENT Sheila Mitchell Dissertation submitted to the faculty of the Virginia Polytechnic Institute and State University in partial fulfillment of the requirements for the degree of Doctor of Philosophy In Biomedical and Veterinary Sciences David S. Lindsay Anne M. Zajac Nammalwar Sriranganathan Sharon G. Witonsky Byron L. Blagburn April 11, 2008 Blacksburg, Virginia Keywords: Apicomplexa, coccidia, canine, animal model, monozoic cyst, cell culture, Toxoplasma gondii, treatment DEVELOPMENT OF A CANINE COCCIDIOSIS MODEL AND THE ANTICOCCIDIAL EFFECTS OF A NEW CHEMOTHERAPEUTIC AGENT Sheila Mitchell ABSTRACT Coccidia are obligate intracellular parasites belonging to the phylum Apicomplexa. Many coccidia are of medical and veterinary importance such as Cystoisospora species and Toxoplasma gondii. The need to discover new anticoccidial therapies has increased due to development of resistance by the parasite or toxicity issues in the patient. The goals of this work were to develop a model for canine coccidiosis while proving that Cystoisospora canis is a true primary pathogen in dogs and to determine the efficacy of a new anticoccidial agent. A canine coccidiosis model would be useful in evaluating new anticoccidial treatments. Oral infections with 5 X 104 (n=2) and 1 X 105 (n=20) sporulated C. canis oocysts were attempted in 22 purpose bred beagle puppies. Clinical signs associated with disease were observed in all dogs. Bacterial and viral pathogens were ruled out by transmission electron microscopy (TEM) and bacterial growth assays. Development of C. canis in cell culture was also evaluated. The efficacy of ponazuril, a new anticoccidial drug, was examined in T. -

Infant Antibiotic Exposure Search EMBASE 1. Exp Antibiotic Agent/ 2
Infant Antibiotic Exposure Search EMBASE 1. exp antibiotic agent/ 2. (Acedapsone or Alamethicin or Amdinocillin or Amdinocillin Pivoxil or Amikacin or Aminosalicylic Acid or Amoxicillin or Amoxicillin-Potassium Clavulanate Combination or Amphotericin B or Ampicillin or Anisomycin or Antimycin A or Arsphenamine or Aurodox or Azithromycin or Azlocillin or Aztreonam or Bacitracin or Bacteriocins or Bambermycins or beta-Lactams or Bongkrekic Acid or Brefeldin A or Butirosin Sulfate or Calcimycin or Candicidin or Capreomycin or Carbenicillin or Carfecillin or Cefaclor or Cefadroxil or Cefamandole or Cefatrizine or Cefazolin or Cefixime or Cefmenoxime or Cefmetazole or Cefonicid or Cefoperazone or Cefotaxime or Cefotetan or Cefotiam or Cefoxitin or Cefsulodin or Ceftazidime or Ceftizoxime or Ceftriaxone or Cefuroxime or Cephacetrile or Cephalexin or Cephaloglycin or Cephaloridine or Cephalosporins or Cephalothin or Cephamycins or Cephapirin or Cephradine or Chloramphenicol or Chlortetracycline or Ciprofloxacin or Citrinin or Clarithromycin or Clavulanic Acid or Clavulanic Acids or clindamycin or Clofazimine or Cloxacillin or Colistin or Cyclacillin or Cycloserine or Dactinomycin or Dapsone or Daptomycin or Demeclocycline or Diarylquinolines or Dibekacin or Dicloxacillin or Dihydrostreptomycin Sulfate or Diketopiperazines or Distamycins or Doxycycline or Echinomycin or Edeine or Enoxacin or Enviomycin or Erythromycin or Erythromycin Estolate or Erythromycin Ethylsuccinate or Ethambutol or Ethionamide or Filipin or Floxacillin or Fluoroquinolones -

Early-Life Antibiotic Use and Risk of Asthma and Eczema: Results of a Discordant Twin Study
Early View Original article Early-life antibiotic use and risk of asthma and eczema: results of a discordant twin study Elise M.A. Slob, Bronwyn K. Brew, Susanne J.H. Vijverberg, Chantal J.A.R. Kats, Cristina Longo, Mariëlle W. Pijnenburg, Toos C.E.M. van Beijsterveldt, Conor V. Dolan, Meike Bartels, Patrick Magnusson, Paul Lichtenstein, Tong Gong, Gerard H. Koppelman, Catarina Almqvist, Dorret I. Boomsma, Anke H. Maitland-van der Zee Please cite this article as: Slob EMA, Brew BK, Vijverberg SJH, et al. Early-life antibiotic use and risk of asthma and eczema: results of a discordant twin study. Eur Respir J 2020; in press (https://doi.org/10.1183/13993003.02021-2019). This manuscript has recently been accepted for publication in the European Respiratory Journal. It is published here in its accepted form prior to copyediting and typesetting by our production team. After these production processes are complete and the authors have approved the resulting proofs, the article will move to the latest issue of the ERJ online. Copyright ©ERS 2020 Early-life antibiotic use and risk of asthma and eczema: results of a discordant twin study Elise M.A. Slob1,2 Bronwyn K. Brew3,4 Susanne J.H. Vijverberg1,2 Chantal J.A.R. Kats1 Cristina Longo1 Mariëlle W. Pijnenburg5 Toos C.E.M. van Beijsterveldt6 Conor V. Dolan6 Meike Bartels6 Patrick Magnusson3 Paul Lichtenstein3 Tong Gong3 Gerard H. Koppelman7,8 Catarina Almqvist3,9 Dorret I. Boomsma6 Anke H. Maitland-van der Zee1,2* 1. Department of Respiratory Medicine, Amsterdam University Medical Centers, University of Amsterdam, P.O. -

Antimicrobial Resistance Benchmark 2020 Antimicrobial Resistance Benchmark 2020
First independent framework for assessing pharmaceutical company action Antimicrobial Resistance Benchmark 2020 Antimicrobial Resistance Benchmark 2020 ACKNOWLEDGEMENTS The Access to Medicine Foundation would like to thank the following people and organisations for their contributions to this report.1 FUNDERS The Antimicrobial Resistance Benchmark research programme is made possible with financial support from UK AID and the Dutch Ministry of Health, Welfare and Sport. Expert Review Committee Research Team Reviewers Hans Hogerzeil - Chair Gabrielle Breugelmans Christine Årdal Gregory Frank Fatema Rafiqi Karen Gallant Nina Grundmann Adrián Alonso Ruiz Hans Hogerzeil Magdalena Kettis Ruth Baron Hitesh Hurkchand Joakim Larsson Dulce Calçada Joakim Larsson Marc Mendelson Moska Hellamand Marc Mendelson Margareth Ndomondo-Sigonda Kevin Outterson Katarina Nedog Sarah Paulin (Observer) Editorial Team Andrew Singer Anna Massey Deirdre Cogan ACCESS TO MEDICINE FOUNDATION Rachel Jones The Access to Medicine Foundation is an independent Emma Ross non-profit organisation based in the Netherlands. It aims to advance access to medicine in low- and middle-income Additional contributors countries by stimulating and guiding the pharmaceutical Thomas Collin-Lefebvre industry to play a greater role in improving access to Alex Kong medicine. Nestor Papanikolaou Address Contact Naritaweg 227-A For more information about this publication, please contact 1043 CB, Amsterdam Jayasree K. Iyer, Executive Director The Netherlands [email protected] +31 (0) 20 215 35 35 www.amrbenchmark.org 1 This acknowledgement is not intended to imply that the individuals and institutions referred to above endorse About the cover: Young woman from the Antimicrobial Resistance Benchmark methodology, Brazil, where 40%-60% of infections are analyses or results.