Comparison of St John's Wort And
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Effect of Selected Herbal Extracts on Lactic Acid Bacteria Activity
applied sciences Article The Effect of Selected Herbal Extracts on Lactic Acid Bacteria Activity Małgorzata Ziarno 1,* , Mariola Kozłowska 2 , Iwona Scibisz´ 3 , Mariusz Kowalczyk 4 , Sylwia Pawelec 4 , Anna Stochmal 4 and Bartłomiej Szleszy ´nski 5 1 Division of Milk Technology, Department of Food Technology and Assessment, Institute of Food Science, Warsaw University of Life Sciences–SGGW (WULS–SGGW), 02-787 Warsaw, Poland 2 Department of Chemistry, Institute of Food Science, Warsaw University of Life Sciences–SGGW (WULS–SGGW), 02-787 Warsaw, Poland; [email protected] 3 Division of Fruit, Vegetable and Cereal Technology, Department of Food Technology and Assessment, Institute of Food Science, Warsaw University of Life Sciences–SGGW (WULS–SGGW), 02-787 Warsaw, Poland; [email protected] 4 Department of Biochemistry and Crop Quality, Institute of Soil Science and Plant Cultivation, State Research Institute, 24-100 Puławy, Poland; [email protected] (M.K.); [email protected] (S.P.); [email protected] (A.S.) 5 Institute of Horticultural Sciences, Warsaw University of Life Sciences–SGGW (WULS–SGGW), 02-787 Warsaw, Poland; [email protected] * Correspondence: [email protected]; Tel.: +48-225-937-666 Abstract: This study aimed to investigate the effect of plant extracts (valerian Valeriana officinalis L., sage Salvia officinalis L., chamomile Matricaria chamomilla L., cistus Cistus L., linden blossom Tilia L., ribwort plantain Plantago lanceolata L., marshmallow Althaea L.) on the activity and growth of lactic acid bacteria (LAB) during the fermentation and passage of milk through a digestive system model. Citation: Ziarno, M.; Kozłowska, M.; The tested extracts were also characterized in terms of their content of polyphenolic compounds and Scibisz,´ I.; Kowalczyk, M.; Pawelec, S.; antioxidant activity. -

ADVISORY BOARDS Each Issue of Herbaigram Is Peer Reviewed by Various Members of Our Advisory Boards Prior to Publication
ADVISORY BOARDS Each issue of HerbaiGram is peer reviewed by various members of our Advisory Boards prior to publication . American Botanical Council Herb Research Dennis V. C. Awang, Ph.D., F.C.I.C., MediPiont Natural Gail B. Mahadr, Ph.D., Research Assistant Professor, Products Consul~ng Services, Ottowa, Ontario, Conodo Deportment o Medical Chemistry &Pharmacognosy, College of Foundation Pharmacy, University of Illinois, Chicago, Illinois Manuel F. Balandrin, R.Ph., Ph.D., Research Scien~st, NPS Rob McCaleb, President Pharmaceuticals, Salt LakeCity , Utah Robin J. Maries, Ph.D., Associate Professor of Botany, Brandon University, Brandon, Manitoba, Conodo Mi(hael J. Balidt, Ph.D., Director of the lns~tute of Econom ic Glenn Appelt, Ph.D., R.Ph., Author and Profess or Botany, the New York Botanical Gorden, Bronx, New York Dennis J. M(Kenna, Ph.D., Consulting Ethnophormocologist, Emeritus, University of Colorado, and with Boulder Beach Joseph M. Betz, Ph.D., Research Chemist, Center for Food Minneapolis, Minnesota Consulting Group Safety and Applied Nutri~on, Division of Natural Products, Food Daniel E. Moerman, Ph.D., William E. Stirton Professor of John A. Beutler, Ph.D., Natural Products Chemist, and Drug Administro~on , Washington, D.C. Anthropology, University of Michigon/ Deorbom, Dearborn, Michigan Notional Cancer Institute Donald J. Brown, N.D., Director, Natural Products Research Consultants; Faculty, Bastyr University, Seattle, Washington Samuel W. Page, Ph.D., Director, Division of Natural Products, Robert A. Bye, Jr., Ph.D., Professor of Ethnobotony, Notional University of Mexico Thomas J. Carlson, M.S., M.D., Senior Director, Center for Food Safety and Applied Nutri~on , Food and Drug Administro~on , Washington, D.C. -
List of Common Covered Over-The-Counter Medications*
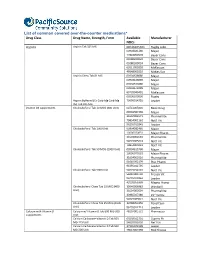
List of common covered over-the-counter medications* Drug Class Drug Name, Strength, Form Available Manufacturer NDCs Aspirin Aspirin Tab 325 MG 00536105301 Rugby Labo 00904681180 Major 12843054578 Bayer Cons 00280200020 Bayer Cons 00280200024 Bayer Cons 62011002003 McKesson 49348000110 McKes Sun Aspirin Chew Tab 81 MG 00904628880 Major 00904628889 Major 00904679480 Major 00904679489 Major 63739043401 McKesson 00536100836 Rugby Aspirin Buffered (Ca Carb-Mg Carb-Mg 70000014701 Leader Ox) Tab 325 MG Vitamin D3 supplements Cholecalciferol Tab 10 MCG (400 Unit) 00761005820 Basic Drug 00904582360 Major 31604002671 PharmaVite 79854001162 Nat’l Vit 96295012845 Leader Cholecalciferol Tab 1000 Unit 00904582460 Major 10006070033 Major Pharm 31604002683 PharmaVite 54629005024 Nat’l Vit 79854005023 Nat’l Vit Cholecalciferol Tab 50 MCG (2000 Unit) 00904615760 Major 10006070162 Major Pharm 31604002516 PharmaVite 51645092199 Plus Pharm 96295012795 Leader Cholecalciferol Tab 5000 Unit 54629794101 Nat’l Vit 58487003702 Freeda Vit 96295012846 Leader 43292056338 Magno-Hump Cholecalciferol Chew Tab 10 MCG (400 35046006982 Windwill Unit) 31604002604 PharmaVite 40985027380 21st Centu 54629089821 Nat’l Vit Cholecalciferol Chew Tab 25 MCG (1000 30768050356 Rexall Sun Unit) 96295012713 Leader Calcium with Vitamin D Calcium w/ Vitamin D Tab 600 MG-200 48107005122 Pharmassur supplements Unit Calcium Carbonate-Vitamin D Tab 500 60258012701 Cypress Ph MG-125 Unit 54629031630 Nat’l Vit Calcium Carbonate-Vitamin D Tab 500 37205043198 Leader MG-200 Unit 49614067298 -

Petition for a Writ of Mandamus
Case: 18-114 Document: 2-1 Page: 1 Filed: 12/01/2017 (1 of 257) No. 17-______ IN THE UNITED STATES COURT OF APPEALS FOR THE FEDERAL CIRCUIT ________________ IN RE AMARIN PHARMA, INC. AND AMARIN PHARMACEUTICALS IRELAND LTD., Petitioners. ________________ On Petition for a Writ of Mandamus to the United States International Trade Commission, In the Matter of Certain Synthetically Produced, Predominantly EPA Omega-3 Products in Ethyl Ester or Re-esterified Triglyceride Form, ITC Docket No. 3247. ________________ PETITION FOR A WRIT OF MANDAMUS ________________ Of Counsel: Ashley C. Parrish Joseph T. Kennedy Principal Counsel Barbara Kurys Jeffrey M. Telep AMARIN PHARMA, INC. Lisa M. Dwyer* 1430 Route 206 Jesse Snyder Bedminster, NJ 07921 KING & SPALDING LLP Telephone: (908) 719-1315 1700 Pennsylvania Ave. N.W. Facsimile: (908) 719-3012 Washington, DC 20006-4706 Telephone: (202) 737-0500 Facsimile: (202) 626-3737 Email: [email protected] Counsel for Petitioners Amarin Pharma, Inc. and Amarin Pharmaceuticals Ireland Ltd. December 1, 2017 *Admission Pending Case: 18-114 Document: 2-1 Page: 2 Filed: 12/01/2017 (2 of 257) CERTIFICATE OF INTEREST Counsel for Amarin Pharma, Inc. and Amarin Pharmaceuticals Ireland Ltd. certify the following: 1. The full name of every party or amicus represented herein: Amarin Pharma, Inc. and Amarin Pharmaceuticals Ireland Ltd. 2. The name of the real party in interest (if the party named in the caption is not the real party in interest) represented us: Not applicable. 3. All parent corporations and any publicly held companies that own 10 percent or more of the stock of the party or amicus curiae represented herein: Amarin Pharma, Inc. -

Differential Effects of Tranylcypromine and Imidazole on Mammary Carcinogenesis in Rats Fed Low and High Fat Diets1
[CANCER RESEARCH 49, 3168-3172, June 15, 1989] Differential Effects of Tranylcypromine and Imidazole on Mammary Carcinogenesis in Rats Fed Low and High Fat Diets1 David L. McCormick,2 Ann M. Spicer, and Jacqueline L. Hollister Life Sciences Department, IIT Research Institute, Chicago, Illinois 60616 ABSTRACT studies with this class of compounds used inhibitors of the cyclooxygenase pathway of arachidonic acid catabolism; exper Neoplastic development in the rat mammary gland can be suppressed iments performed in our laboratory and by Ip and coworkers by inhibition of the activity of several enzymes involved in eicosanoid biosynthesis. In order to investigate the potential utility of prostacyclin demonstrated that the postcarcinogen phase of rat mammary and thromboxane synthetases as targets for mammary cancer chemopre- carcinogenesis can be suppressed by dietary administration of vention, experiments were conducted to determine the influence of tran- indomethacin (7, 8) or flurbiprofen (9). However, although the ylcypromine (TCP), an inhibitor of prostacyclin synthetase, and ¡mida/ole anticarcinogenic activity of indomethacin is similar to that of (IMI), an inhibitor of thromboxane synthetase, on mammary carcinogen- more widely studied inhibitors of mammary carcinogenesis such esis induced in rats by 7V-methyl-/V-nitrosourea. Fifty-day-old female as retinyl acetate (10) and selenium (11), the dose levels of Sprague-Dawley |Hsd:SD(BR)l rats received a single s.c. dose of 0 or 40 indomethacin required for chemopreventive efficacy in rats are mg of .V-mcth>l-.Y-nitrosoiirea per kg of body weight. Beginning 7 days close to the threshold of lethal toxicity (12). For this reason, after carcinogen administration, groups of rats were fed isoenergetic, studies are ongoing to identify additional modifiers of arachi casein-based diets containing 3 or 20% corn oil (w/w), supplemented with donic acid metabolism whose administration provides an effec (per kg of diet) 10 mg of TCP, 1000 mg of IMI, or sucrose carrier only. -

Curative Effect of Selenium Against Indomethacin-Induced Gastric Ulcers in Rats
J. Microbiol. Biotechnol. (2011), 21(4), 400–404 doi: 10.4014/jmb.1012.12019 First published online 19 January 2011 Curative Effect of Selenium Against Indomethacin-Induced Gastric Ulcers in Rats Kim, Jeong-Hwan1, Byung-Woo Kim1,3,4, Hyun-Ju Kwon1,3,4, and Soo-Wan Nam1,2,4* 1Department of Biomaterial Control, 2Department of Biotechnology and Bioengineering, 3Department of Life Science and Biotechnology, and 4Blue-Bio Industry RIC, Dong-Eui University, Busan 614-714, Korea Received: December 16, 2010 / Revised: December 30, 2010 / Accepted: December 31, 2010 Indomethacin is a nonsteroid anti-inflammatory agent erosions, ulcerative lesions, and petechial bleeding in the that is known to induce severe gastric mucosal lesions. In mucosa of stomach as serious side effects [10, 18]. According this study, we investigated the effect of selenium on gastric to previous reports, the oral administration of indomethacin in mucosal lesions in rats. To confirm the curative effect of rats causes ulcerative lesions in the gastric mucosa [7, 13]. selenium against indomethacin-induced gastric ulcers, Furthermore, the development of the gastric mucosal lesions gastric ulcers were induced by oral administration of induced by indomethacin is mainly mediated through 25 mg/kg indomethacin, and then different doses (10, 50, generation of oxygen free radicals and lipid peroxidation and 100 µg/kg of body weight) of selenium or vehicle were [6, 22, 25, 26, 28, 29]. treated by oral gavage for 3 days. Oral administration of Selenium is an essential nutrient of fundamental importance indomethacin clearly increased the gastric ulcer area in to human biology. It has important metabolic functions in the stomach, whereas selenium applied for 3 days significantly animals, including protection of membrane lipids and decreased the gastric ulcer area in a dose-dependent macromolecules from oxidative damage produced by peroxides manner. -

Chronic Administration of Probiotic L. Rhamnosus Increases Anxiety-Like Behavior in Group-Housed Male Long Evans Rats
University of Dayton eCommons Honors Theses University Honors Program 4-2018 Chronic Administration of Probiotic L. rhamnosus Increases Anxiety-like Behavior in Group-housed Male Long Evans Rats Parker Maddison Griff University of Dayton Follow this and additional works at: https://ecommons.udayton.edu/uhp_theses Part of the Biology Commons, and the Psychology Commons eCommons Citation Griff, Parker Maddison, "Chronic Administration of Probiotic L. rhamnosus Increases Anxiety-like Behavior in Group-housed Male Long Evans Rats" (2018). Honors Theses. 157. https://ecommons.udayton.edu/uhp_theses/157 This Honors Thesis is brought to you for free and open access by the University Honors Program at eCommons. It has been accepted for inclusion in Honors Theses by an authorized administrator of eCommons. For more information, please contact [email protected], [email protected]. Chronic Administration of Probiotic L. rhamnosus Increases Anxiety-like Behavior in Group-housed Male Long Evans Rats Honors Thesis Parker Maddison Griff Department: Psychology and Biology Advisor: Tracy Butler, Ph.D. and Yvonne Sun, Ph.D. April 2018 Chronic Administration of Probiotic L. rhamnosus Increases Anxiety-like Behavior in Group-housed Male Long Evans Rats Honors Thesis Parker Maddison Griff Department: Psychology and Biology Advisor: Tracy Butler, Ph.D. and Yvonne Sun, Ph.D. April 2018 Abstract Early life stress is a risk factor for later development of alcohol use disorders and anxiety disorders in humans. Using rodent experimental models, we know that rats experiencing social isolation as early-life stress exhibit greater anxiety-like behavior and alcohol consumption than rats housed in groups. Examining potential preventive strategies, we investigated the effects of probiotics, which have previously been shown to decrease rodent anxiety-like behavior, on the relationship between early-life stress and anxiety-like behavior in rats. -

Antifungal Activity of Lactobacillus Pentosus ŁOCK 0979 in the Presence of Polyols and Galactosyl-Polyols
Probiotics & Antimicro. Prot. https://doi.org/10.1007/s12602-017-9344-0 Antifungal Activity of Lactobacillus pentosus ŁOCK 0979 in the Presence of Polyols and Galactosyl-Polyols Lidia Lipińska1 & Robert Klewicki2 & Michał Sójka2 & Radosław Bonikowski3 & Dorota Żyżelewicz2 & Krzysztof Kołodziejczyk2 & Elżbieta Klewicka 1 # The Author(s) 2017. This article is an open access publication Abstract The antifungal activity of Lactobacillus pentosus Keywords Antifungal activity . Galactosyl-polyols . ŁOCK 0979 depends both on the culture medium and on the Lactobacillus . Metabolites . Polyols . SEM fungal species. In the control medium, the strain exhibited limited antagonistic activity against indicator food-borne molds and yeasts. However, the supplementation of the bac- Introduction terial culture medium with polyols (erythritol, lactitol, maltitol, mannitol, sorbitol, xylitol) or their galactosyl deriva- Filamentous fungi and yeasts are present in almost all types of tives (gal-erythritol, gal-sorbitol, gal-xylitol) enhanced the an- ecosystems due to their high adaptation ability and low nutri- tifungal properties of Lactobacillus pentosus ŁOCK 0979. Its tional requirements. Filamentous fungi are widespread food metabolites were identified and quantified by enzymatic spoilage microorganisms responsible for significant economic methods, HPLC, UHPLC-MS coupled with QuEChERS, losses in the agri-food industry [6]; they are also a major and GC-MS. The presence of polyols and gal-polyols signif- health concern due to mycotoxin production. The most com- icantly affected the acid metabolite profile of the bacterial mon genera of spoilage fungi include Penicillium, Fusarium, culture supernatant. In addition, lactitol and mannitol were Aspergillus, Cladosporium,andRhizopus [21]. Commercial used by bacteria as alternative carbon sources. A number of foodstuffs are usually protected from such microorganisms by compounds with potential antifungal properties were identi- physical and chemical techniques. -

Potential Adverse Effects of Resveratrol: a Literature Review
International Journal of Molecular Sciences Review Potential Adverse Effects of Resveratrol: A Literature Review Abdullah Shaito 1 , Anna Maria Posadino 2, Nadin Younes 3, Hiba Hasan 4 , Sarah Halabi 5, Dalal Alhababi 3, Anjud Al-Mohannadi 3, Wael M Abdel-Rahman 6 , Ali H. Eid 7,*, Gheyath K. Nasrallah 3,* and Gianfranco Pintus 6,2,* 1 Department of Biological and Chemical Sciences, Lebanese International University, 1105 Beirut, Lebanon; [email protected] 2 Department of Biomedical Sciences, University of Sassari, 07100 Sassari, Italy; [email protected] 3 Department of Biomedical Science, College of Health Sciences, and Biomedical Research Center Qatar University, P.O Box 2713 Doha, Qatar; [email protected] (N.Y.); [email protected] (D.A.); [email protected] (A.A.-M.) 4 Institute of Anatomy and Cell Biology, Justus-Liebig-University Giessen, 35392 Giessen, Germany; [email protected] 5 Biology Department, Faculty of Arts and Sciences, American University of Beirut, 1105 Beirut, Lebanon; [email protected] 6 Department of Medical Laboratory Sciences, College of Health Sciences and Sharjah Institute for Medical Research, University of Sharjah, Sharjah P.O Box: 27272, United Arab Emirates; [email protected] 7 Department of Pharmacology and Toxicology, Faculty of Medicine, American University of Beirut, P.O. Box 11-0236 Beirut, Lebanon * Correspondence: [email protected] (A.H.E.); [email protected] (G.K.N.); [email protected] (G.P.) Received: 13 December 2019; Accepted: 15 March 2020; Published: 18 March 2020 Abstract: Due to its health benefits, resveratrol (RE) is one of the most researched natural polyphenols. -

Common Name: SELENIUM SULFIDE HAZARD SUMMARY
Common Name: SELENIUM SULFIDE CAS Number: 7446-34-6 RTK Substance number: 1653 DOT Number: UN 2657 Date: October 1995 Revision: October 2001 ------------------------------------------------------------------------- ------------------------------------------------------------------------- HAZARD SUMMARY * Selenium Sulfide can affect you when breathed in and by * If you think you are experiencing any work-related health passing through your skin. problems, see a doctor trained to recognize occupational * Selenium Sulfide should be handled as a CARCINOGEN- diseases. Take this Fact Sheet with you. -WITH EXTREME CAUTION. * Contact can irritate the eyes with possible eye damage. WORKPLACE EXPOSURE LIMITS * Breathing Selenium Sulfide can irritate the nose and The following exposure limits are for Selenium compounds throat. (measured as Selenium): * High exposure may cause headache, nausea, vomiting, garlic odor of the breath, metallic taste and coated tongue. OSHA: The legal airborne permissible exposure limit * Repeated exposure can cause pallor, nervousness and (PEL) is 0.2 mg/m3 averaged over an 8-hour mood changes. workshift. * Selenium Sulfide may damage the liver and kidneys. NIOSH: The recommended airborne exposure limit is IDENTIFICATION 0.2 mg/m3 averaged over a 10-hour workshift. Selenium Sulfide is a bright orange powder. It is used in medicated shampoos. ACGIH: The recommended airborne exposure limit is 3 0.2 mg/m averaged over an 8-hour workshift. REASON FOR CITATION * Selenium Sulfide is on the Hazardous Substance List * Selenium Sulfide may be a CARCINOGEN in humans. because it is regulated by OSHA and cited by ACGIH, There may be no safe level of exposure to a carcinogen, so DOT, NIOSH, NTP, DEP, HHAG and EPA. all contact should be reduced to the lowest possible level. -

Role of Selenium in HIV Infection
Role of selenium in HIV infection The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters Citation Stone, Cosby A, Kosuke Kawai, Roland Kupka, and Wafaie W Fawzi. 2010. “Role of Selenium in HIV Infection.” Nutrition Reviews 68 (11) (October 20): 671–681. doi:10.1111/j.1753-4887.2010.00337.x. Published Version doi:10.1111/j.1753-4887.2010.00337.x Citable link http://nrs.harvard.edu/urn-3:HUL.InstRepos:26951080 Terms of Use This article was downloaded from Harvard University’s DASH repository, and is made available under the terms and conditions applicable to Other Posted Material, as set forth at http:// nrs.harvard.edu/urn-3:HUL.InstRepos:dash.current.terms-of- use#LAA NIH Public Access Author Manuscript Nutr Rev. Author manuscript; available in PMC 2011 November 1. NIH-PA Author ManuscriptPublished NIH-PA Author Manuscript in final edited NIH-PA Author Manuscript form as: Nutr Rev. 2010 November ; 68(11): 671±681. doi:10.1111/j.1753-4887.2010.00337.x. The Role of Selenium in HIV Infection Cosby A Stone, Kosuke Kawai, Roland Kupka, Wafaie W Fawzi Harvard School of Public Health Cosby A Stone, School of Public Health and School of Medicine, University of Alabama at Birmingham, Birmingham, AL, USA Kosuke Kawai, Department of Epidemiology, Harvard School of Public Health, Boston, MA, USA Roland Kupka, and Department of Nutrition, Harvard School of Public Health, Boston, MA, USA and United Nations Children’s Fund, Regional Office for West and Central Africa, Dakar, Senegal Wafaie W. -

A Proteomic View of Probiotic Lactobacillus Rhamnosus GG
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Helsingin yliopiston digitaalinen arkisto Department of Veterinary Biosciences Faculty of Veterinary Medicine University of Helsinki Helsinki, Finland A proteomic view of probioƟ c Lactobacillus rhamnosus GG KerƩ u Koskenniemi ACADEMIC DISSERTATION To be presented, with the permission of the Faculty of Veterinary Medicine of the University of Helsinki, for public examinaƟ on in the Walter Auditorium, Agnes Sjöbergin katu 2, Helsinki, on January 13th 2012, at 12 o’clock noon. Helsinki 2012 Supervisor Docent Pekka Varmanen Division of Food Technology Department of Food and Environmental Sciences University of Helsinki Helsinki, Finland Reviewers Professor Sirpa Kärenlampi Department of Biosciences University of Eastern Finland Kuopio, Finland Docent Jaana Mättö Finnish Red Cross Blood Service Helsinki, Finland Opponent Professor Oscar Kuipers Department of Molecular Genetics Groningen Biomolecular Sciences and Biotechnology Institute University of Groningen Groningen, the Netherlands. Layout: Tinde Päivärinta Cover fi gure: A 2-D DIGE gel image (photo Kerttu Koskenniemi) ISBN 978-952-10-7403-5 (paperback) ISBN 978-952-10-7404-2 (PDF) ISSN 1799-7372 http://ethesis.helsinki.fi Unigrafi a Helsinki 2012 CONTENTS Abstract List of abbreviations List of original publications 1. Review of the literature ..........................................................................................1 1.1. Probiotic bacteria .....................................................................................................1