Research Papers-Philosophy/Download/6558
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Glossary Physics (I-Introduction)
1 Glossary Physics (I-introduction) - Efficiency: The percent of the work put into a machine that is converted into useful work output; = work done / energy used [-]. = eta In machines: The work output of any machine cannot exceed the work input (<=100%); in an ideal machine, where no energy is transformed into heat: work(input) = work(output), =100%. Energy: The property of a system that enables it to do work. Conservation o. E.: Energy cannot be created or destroyed; it may be transformed from one form into another, but the total amount of energy never changes. Equilibrium: The state of an object when not acted upon by a net force or net torque; an object in equilibrium may be at rest or moving at uniform velocity - not accelerating. Mechanical E.: The state of an object or system of objects for which any impressed forces cancels to zero and no acceleration occurs. Dynamic E.: Object is moving without experiencing acceleration. Static E.: Object is at rest.F Force: The influence that can cause an object to be accelerated or retarded; is always in the direction of the net force, hence a vector quantity; the four elementary forces are: Electromagnetic F.: Is an attraction or repulsion G, gravit. const.6.672E-11[Nm2/kg2] between electric charges: d, distance [m] 2 2 2 2 F = 1/(40) (q1q2/d ) [(CC/m )(Nm /C )] = [N] m,M, mass [kg] Gravitational F.: Is a mutual attraction between all masses: q, charge [As] [C] 2 2 2 2 F = GmM/d [Nm /kg kg 1/m ] = [N] 0, dielectric constant Strong F.: (nuclear force) Acts within the nuclei of atoms: 8.854E-12 [C2/Nm2] [F/m] 2 2 2 2 2 F = 1/(40) (e /d ) [(CC/m )(Nm /C )] = [N] , 3.14 [-] Weak F.: Manifests itself in special reactions among elementary e, 1.60210 E-19 [As] [C] particles, such as the reaction that occur in radioactive decay. -

An Atomic Physics Perspective on the New Kilogram Defined by Planck's Constant
An atomic physics perspective on the new kilogram defined by Planck’s constant (Wolfgang Ketterle and Alan O. Jamison, MIT) (Manuscript submitted to Physics Today) On May 20, the kilogram will no longer be defined by the artefact in Paris, but through the definition1 of Planck’s constant h=6.626 070 15*10-34 kg m2/s. This is the result of advances in metrology: The best two measurements of h, the Watt balance and the silicon spheres, have now reached an accuracy similar to the mass drift of the ur-kilogram in Paris over 130 years. At this point, the General Conference on Weights and Measures decided to use the precisely measured numerical value of h as the definition of h, which then defines the unit of the kilogram. But how can we now explain in simple terms what exactly one kilogram is? How do fixed numerical values of h, the speed of light c and the Cs hyperfine frequency νCs define the kilogram? In this article we give a simple conceptual picture of the new kilogram and relate it to the practical realizations of the kilogram. A similar change occurred in 1983 for the definition of the meter when the speed of light was defined to be 299 792 458 m/s. Since the second was the time required for 9 192 631 770 oscillations of hyperfine radiation from a cesium atom, defining the speed of light defined the meter as the distance travelled by light in 1/9192631770 of a second, or equivalently, as 9192631770/299792458 times the wavelength of the cesium hyperfine radiation. -

Non-Einsteinian Interpretations of the Photoelectric Effect
NON-EINSTEINIAN INTERPRETATIONS ------ROGER H. STUEWER------ serveq spectral distribution for black-body radiation, and it led inexorably ~o _the ~latantl~ f::se conclusion that the total radiant energy in a cavity is mfimte. (This ultra-violet catastrophe," as Ehrenfest later termed it, was point~d out independently and virtually simultaneously, though not Non-Einsteinian Interpretations of the as emphatically, by Lord Rayleigh.2) Photoelectric Effect Einstein developed his positive argument in two stages. First, he derived an expression for the entropy of black-body radiation in the Wien's law spectral region (the high-frequency, low-temperature region) and used this expression to evaluate the difference in entropy associated with a chan.ge in volume v - Vo of the radiation, at constant energy. Secondly, he Few, if any, ideas in the history of recent physics were greeted with ~o~s1dere? the case of n particles freely and independently moving around skepticism as universal and prolonged as was Einstein's light quantum ms1d: a given v~lume Vo _and determined the change in entropy of the sys hypothesis.1 The period of transition between rejection and acceptance tem if all n particles accidentally found themselves in a given subvolume spanned almost two decades and would undoubtedly have been longer if v ~ta r~ndomly chosen instant of time. To evaluate this change in entropy, Arthur Compton's experiments had been less striking. Why was Einstein's Emstem used the statistical version of the second law of thermodynamics hypothesis not accepted much earlier? Was there no experimental evi in _c?njunction with his own strongly physical interpretation of the prob dence that suggested, even demanded, Einstein's hypothesis? If so, why ~b1ht~. -

Physics Nobel Prize 1975
Physics Nobel Prize 1975 Nobel prize winners, left to right, A. Bohr, B. Mottelson and J. Rainwater. 0. Kofoed-Hansen (Photos Keystone Press, Photopress) Before 1949 every physicist knew that mooted, it was a major breakthrough from 1943-45. From December 1943 the atomic nucleus does not rotate. in thinking about the nucleus and he was actually in the USA. Back in The reasoning behind this erroneous opened a whole new field of research Denmark after the war, he obtained belief came from the following argu• in nuclear physics. This work has for his Ph. D. in 1954 for work on rota• ments. A quantum-mechanical rotator years been guided by the inspiration tional states in atomic nuclei. His with the moment of inertia J can take of Bohr and Mottelson. thesis was thus based on the work for up various energy levels with rota• An entire industry of nuclear which he has now been recognized at tional energies equal to h2(l + 1)| research thus began with Rainwater's the very highest level. He is Professor /8TC2J, where h is Planck's constant brief contribution to Physical Review at the Niels Bohr Institute of the ^nd I is the spin. As an example, I may in 1950 entitled 'Nuclear energy level University of Copenhagen and a nave values 0, 2, 4, ... etc. If the argument for a spheroidal nuclear member of the CERN Scientific Policy nucleus is considered as a rigid body, model'. Today a fair-sized library is Committee. then the moment of inertia J is very needed in order to contain all the Ben Mottelson was born in the USA large and the rotational energies papers written on deformed nuclei and in 1926 and has, for many years, become correspondingly very small. -
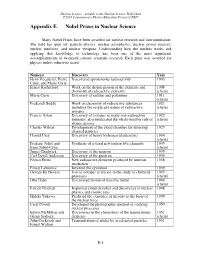
Appendix E Nobel Prizes in Nuclear Science
Nuclear Science—A Guide to the Nuclear Science Wall Chart ©2018 Contemporary Physics Education Project (CPEP) Appendix E Nobel Prizes in Nuclear Science Many Nobel Prizes have been awarded for nuclear research and instrumentation. The field has spun off: particle physics, nuclear astrophysics, nuclear power reactors, nuclear medicine, and nuclear weapons. Understanding how the nucleus works and applying that knowledge to technology has been one of the most significant accomplishments of twentieth century scientific research. Each prize was awarded for physics unless otherwise noted. Name(s) Discovery Year Henri Becquerel, Pierre Discovered spontaneous radioactivity 1903 Curie, and Marie Curie Ernest Rutherford Work on the disintegration of the elements and 1908 chemistry of radioactive elements (chem) Marie Curie Discovery of radium and polonium 1911 (chem) Frederick Soddy Work on chemistry of radioactive substances 1921 including the origin and nature of radioactive (chem) isotopes Francis Aston Discovery of isotopes in many non-radioactive 1922 elements, also enunciated the whole-number rule of (chem) atomic masses Charles Wilson Development of the cloud chamber for detecting 1927 charged particles Harold Urey Discovery of heavy hydrogen (deuterium) 1934 (chem) Frederic Joliot and Synthesis of several new radioactive elements 1935 Irene Joliot-Curie (chem) James Chadwick Discovery of the neutron 1935 Carl David Anderson Discovery of the positron 1936 Enrico Fermi New radioactive elements produced by neutron 1938 irradiation Ernest Lawrence -

1. Physical Constants 1 1
1. Physical constants 1 1. PHYSICAL CONSTANTS Table 1.1. Reviewed 1998 by B.N. Taylor (NIST). Based mainly on the “1986 Adjustment of the Fundamental Physical Constants” by E.R. Cohen and B.N. Taylor, Rev. Mod. Phys. 59, 1121 (1987). The last group of constants (beginning with the Fermi coupling constant) comes from the Particle Data Group. The figures in parentheses after the values give the 1-standard- deviation uncertainties in the last digits; the corresponding uncertainties in parts per million (ppm) are given in the last column. This set of constants (aside from the last group) is recommended for international use by CODATA (the Committee on Data for Science and Technology). Since the 1986 adjustment, new experiments have yielded improved values for a number of constants, including the Rydberg constant R∞, the Planck constant h, the fine- structure constant α, and the molar gas constant R,and hence also for constants directly derived from these, such as the Boltzmann constant k and Stefan-Boltzmann constant σ. The new results and their impact on the 1986 recommended values are discussed extensively in “Recommended Values of the Fundamental Physical Constants: A Status Report,” B.N. Taylor and E.R. Cohen, J. Res. Natl. Inst. Stand. Technol. 95, 497 (1990); see also E.R. Cohen and B.N. Taylor, “The Fundamental Physical Constants,” Phys. Today, August 1997 Part 2, BG7. In general, the new results give uncertainties for the affected constants that are 5 to 7 times smaller than the 1986 uncertainties, but the changes in the values themselves are smaller than twice the 1986 uncertainties. -

Determination of Planck's Constant Using the Photoelectric Effect
Determination of Planck’s Constant Using the Photoelectric Effect Lulu Liu (Partner: Pablo Solis)∗ MIT Undergraduate (Dated: September 25, 2007) Together with my partner, Pablo Solis, we demonstrate the particle-like nature of light as charac- terized by photons with quantized and distinct energies each dependent only on its frequency (ν) and scaled by Planck’s constant (h). Using data obtained through the bombardment of an alkali metal surface (in our case, potassium) by light of varying frequencies, we calculate Planck’s constant, h to be 1.92 × 10−15 ± 1.08 × 10−15eV · s. 1. THEORY AND MOTIVATION This maximum kinetic energy can be determined by applying a retarding potential (Vr) across a vacuum gap It was discovered by Heinrich Hertz that light incident in a circuit with an amp meter. On one side of this gap upon a matter target caused the emission of electrons is the photoelectron emitter, a metal with work function from the target. The effect was termed the Hertz Effect W0. We let light of different frequencies strike this emit- (and later the Photoelectric Effect) and the electrons re- ter. When eVr = Kmax we will cease to see any current ferred to as photoelectrons. It was understood that the through the circuit. By finding this voltage we calculate electrons were able to absorb the energy of the incident the maximum kinetic energy of the electrons emitted as a light and escape from the coulomb potential that bound function of radiation frequency. This relationship (with it to the nucleus. some variation due to error) should model Equation 2 According to classical wave theory, the energy of a above. -

Heisenberg's Visit to Niels Bohr in 1941 and the Bohr Letters
Klaus Gottstein Max-Planck-Institut für Physik (Werner-Heisenberg-Institut) Föhringer Ring 6 D-80805 Munich, Germany 26 February, 2002 New insights? Heisenberg’s visit to Niels Bohr in 1941 and the Bohr letters1 The documents recently released by the Niels Bohr Archive do not, in an unambiguous way, solve the enigma of what happened during the critical brief discussion between Bohr and Heisenberg in 1941 which so upset Bohr and made Heisenberg so desperate. But they are interesting, they show what Bohr remembered 15 years later. What Heisenberg remembered was already described by him in his memoirs “Der Teil und das Ganze”. The two descriptions are complementary, they are not incompatible. The two famous physicists, as Hans Bethe called it recently, just talked past each other, starting from different assumptions. They did not finish their conversation. Bohr broke it off before Heisenberg had a chance to complete his intended mission. Heisenberg and Bohr had not seen each other since the beginning of the war in 1939. In the meantime, Heisenberg and some other German physicists had been drafted by Army Ordnance to explore the feasibility of a nuclear bomb which, after the discovery of fission and of the chain reaction, could not be ruled out. How real was this theoretical possibility? By 1941 Heisenberg, after two years of intense theoretical and experimental investigations by the drafted group known as the “Uranium Club”, had reached the conclusion that the construction of a nuclear bomb would be feasible in principle, but technically and economically very difficult. He knew in principle how it could be done, by Uranium isotope separation or by Plutonium production in reactors, but both ways would take many years and would be beyond the means of Germany in time of war, and probably also beyond the means of Germany’s adversaries. -

Quantifying the Quantum Stephan Schlamminger Looks at the Origins of the Planck Constant and Its Current Role in Redefining the Kilogram
measure for measure Quantifying the quantum Stephan Schlamminger looks at the origins of the Planck constant and its current role in redefining the kilogram. child on a swing is an everyday mechanical constant (h) with high precision, measure a approximation of a macroscopic a macroscopic experiment is necessary, force — in A harmonic oscillator. The total energy because the unit of mass, the kilogram, can this case of such a system depends on its amplitude, only be accessed with high precision at the the weight while the frequency of oscillation is, at one-kilogram value through its definition of a mass least for small amplitudes, independent as the mass of the International Prototype m — as of the amplitude. So, it seems that the of the Kilogram (IPK). The IPK is the only the product energy of the system can be adjusted object on Earth whose mass we know for of a current continuously from zero upwards — that is, certain2 — all relative uncertainties increase and a voltage the child can swing at any amplitude. This from thereon. divided by a velocity, is not the case, however, for a microscopic The revised SI, likely to come into mg = UI/v (with g the oscillator, the physics of which is described effect in 2019, is based on fixed numerical Earth’s gravitational by the laws of quantum mechanics. A values, without uncertainties, of seven acceleration). quantum-mechanical swing can change defining constants, three of which are Meanwhile, the its energy only in steps of hν, the product already fixed in the present SI; the four discoveries of the Josephson of the Planck constant and the oscillation additional ones are the elementary and quantum Hall effects frequency — the (energy) quantum of charge and the Planck, led to precise electrical the oscillator. -

1. Physical Constants 101
1. Physical constants 101 1. PHYSICAL CONSTANTS Table 1.1. Reviewed 2010 by P.J. Mohr (NIST). Mainly from the “CODATA Recommended Values of the Fundamental Physical Constants: 2006” by P.J. Mohr, B.N. Taylor, and D.B. Newell in Rev. Mod. Phys. 80 (2008) 633. The last group of constants (beginning with the Fermi coupling constant) comes from the Particle Data Group. The figures in parentheses after the values give the 1-standard-deviation uncertainties in the last digits; the corresponding fractional uncertainties in parts per 109 (ppb) are given in the last column. This set of constants (aside from the last group) is recommended for international use by CODATA (the Committee on Data for Science and Technology). The full 2006 CODATA set of constants may be found at http://physics.nist.gov/constants. See also P.J. Mohr and D.B. Newell, “Resource Letter FC-1: The Physics of Fundamental Constants,” Am. J. Phys, 78 (2010) 338. Quantity Symbol, equation Value Uncertainty (ppb) speed of light in vacuum c 299 792 458 m s−1 exact∗ Planck constant h 6.626 068 96(33)×10−34 Js 50 Planck constant, reduced ≡ h/2π 1.054 571 628(53)×10−34 Js 50 = 6.582 118 99(16)×10−22 MeV s 25 electron charge magnitude e 1.602 176 487(40)×10−19 C = 4.803 204 27(12)×10−10 esu 25, 25 conversion constant c 197.326 9631(49) MeV fm 25 conversion constant (c)2 0.389 379 304(19) GeV2 mbarn 50 2 −31 electron mass me 0.510 998 910(13) MeV/c = 9.109 382 15(45)×10 kg 25, 50 2 −27 proton mass mp 938.272 013(23) MeV/c = 1.672 621 637(83)×10 kg 25, 50 = 1.007 276 466 77(10) u = 1836.152 672 47(80) me 0.10, 0.43 2 deuteron mass md 1875.612 793(47) MeV/c 25 12 2 −27 unified atomic mass unit (u) (mass C atom)/12 = (1 g)/(NA mol) 931.494 028(23) MeV/c = 1.660 538 782(83)×10 kg 25, 50 2 −12 −1 permittivity of free space 0 =1/μ0c 8.854 187 817 .. -

Release of Documents Relating to 1941 Bohr-Heisenberg Meeting
Release of documents relating to 1941 Bohr-Heisenberg meeting DOCUMENTS RELEASED 6 FEBRUARY 2002 The family of Niels Bohr has decided to release all documents deposited at the Niels Bohr Archive, either written or dictated by Niels Bohr, pertaining specifically to the meeting between Bohr and Heisenberg in September 1941. There are in all eleven documents. The decision has been made in order to avoid possible misunderstandings regarding the contents of the documents. The documents supplement and confirm previously published statements of Bohr's recollections of the meeting, especially those of his son, Aage Bohr. The documents have now been organised, transcribed and translated into English at the Niels Bohr Archive. Because of the overwhelming interest in the material, it has been decided that the material should be published in full instead of being made available to scholars upon individual application, as is normal practice at the Niels Bohr Archive. This has been done by placing facsimiles, transcriptions and translations on this website here. An illustrated edition of the transcriptions and translations is published in the journal Naturens Verden, Vol. 84, No. 8 - 9. Reprints can be obtained from the Niels Bohr Archive for USD8/EUR8/DKK50 (prepaid). Release of documents relating to 1941 Bohr-Heisenberg meeting. Document 1. Draft of letter from Bohr to Heisenberg, never sent. In the handwriting of Niels Bohr's assistant, Aage Petersen. Undated, but written after the first publication, in 1957, of the Danish translation of Robert Jungk, Heller als Tausend Sonnen, the first edition of Jungk's book to contain Heisenberg's letter. -

Absolute Zero, Absolute Temperature. Absolute Zero Is the Lowest
Contents Radioactivity: The First Puzzles................................................ 1 The “Uranic Rays” of Henri Becquerel .......................................... 1 The Discovery ............................................................... 2 Is It Really Phosphorescence? .............................................. 4 What Is the Nature of the Radiation?....................................... 5 A Limited Impact on Scientists and the Public ............................ 6 Why 1896? .................................................................. 7 Was Radioactivity Discovered by Chance? ................................ 7 Polonium and Radium............................................................. 9 Marya Skłodowska .......................................................... 9 Pierre Curie .................................................................. 10 Polonium and Radium: Pierre and Marie Curie Invent Radiochemistry.. 11 Enigmas...................................................................... 14 Emanation from Thorium ......................................................... 17 Ernest Rutherford ........................................................... 17 Rutherford Studies Radioactivity: ˛-and ˇ-Rays.......................... 18 ˇ-Rays Are Electrons ....................................................... 19 Rutherford in Montreal: The Radiation of Thorium, the Exponential Decrease........................................... 19 “Induced” and “Excited” Radioactivity .................................... 20 Elster