Development and Application of a Green-Chemistry
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

High Purity Inorganics
High Purity Inorganics www.alfa.com INCLUDING: • Puratronic® High Purity Inorganics • Ultra Dry Anhydrous Materials • REacton® Rare Earth Products www.alfa.com Where Science Meets Service High Purity Inorganics from Alfa Aesar Known worldwide as a leading manufacturer of high purity inorganic compounds, Alfa Aesar produces thousands of distinct materials to exacting standards for research, development and production applications. Custom production and packaging services are part of our regular offering. Our brands are recognized for purity and quality and are backed up by technical and sales teams dedicated to providing the best service. This catalog contains only a selection of our wide range of high purity inorganic materials. Many more products from our full range of over 46,000 items are available in our main catalog or online at www.alfa.com. APPLICATION FOR INORGANICS High Purity Products for Crystal Growth Typically, materials are manufactured to 99.995+% purity levels (metals basis). All materials are manufactured to have suitably low chloride, nitrate, sulfate and water content. Products include: • Lutetium(III) oxide • Niobium(V) oxide • Potassium carbonate • Sodium fluoride • Thulium(III) oxide • Tungsten(VI) oxide About Us GLOBAL INVENTORY The majority of our high purity inorganic compounds and related products are available in research and development quantities from stock. We also supply most products from stock in semi-bulk or bulk quantities. Many are in regular production and are available in bulk for next day shipment. Our experience in manufacturing, sourcing and handling a wide range of products enables us to respond quickly and efficiently to your needs. CUSTOM SYNTHESIS We offer flexible custom manufacturing services with the assurance of quality and confidentiality. -

Chemical Safety and Waste Management Manual
Chemical Safety and Waste Management Manual University of Alabama at Birmingham Department of Occupational Health & Safety Chemical Safety Division 2002 EDITION 1. INTRODUCTION In a comparatively short time, the University of Alabama at Birmingham has gained significant recognition as a center of excellence for teaching, medical services and research programs. This is a highly commendable achievement and one that could not have been realized without the continued support and dedication of faculty, staff members, and employees. Similar unfailing cooperation and support are necessary for the institution to be equally successful in its development of a comprehensive occupational health and safety program for the protection of University personnel, students, and the surrounding community. An important part of this program is concerned with the safe and prudent handling of chemicals and their proper legal disposal as regulated by the Environmental Protection Agency (EPA) and the Alabama Department of Environmental Management (ADEM). Almost every laboratory and many allied and support personnel at UAB use chemicals in their daily activities. It is the purpose of this manual to describe the operation of the Chemical Safety Program and to provide guidance in establishing safe work practices for the use of chemicals. This program applies to all work operations at this University where employees may be exposed to hazardous substances under normal working conditions or during an emergency. The Chemical Safety and Waste Management Manual combines both the Chemical Hygiene Plan for laboratories and the Hazard Communication Program for maintenance, environmental services, and other support personnel. The Occupational Safety and Health Administration (OSHA) Hazard Communication Standard may be found at : http://www.osha- slc.gov/OshStd_data/1910_1200.html. -
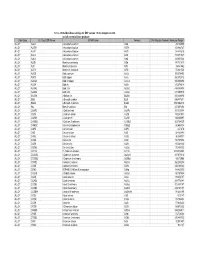
Database Full Listing
16-Nov-06 OLI Data Base Listings for ESP version 7.0.46, Analyzers 2.0.46 and all current alliance products Data Base OLI Tag (ESP) Name IUPAC Name Formula CAS Registry Number Molecular Weight ALLOY AL2U 2-Aluminum uranium Al2U 291.98999 ALLOY AL3TH 3-Aluminum thorium Al3Th 312.982727 ALLOY AL3TI 3-Aluminum titanium Al3Ti 128.824615 ALLOY AL3U 3-Aluminum uranium Al3U 318.971527 ALLOY AL4U 4-Aluminum uranium Al4U 345.953064 ALLOY ALSB Aluminum antimony AlSb 148.731537 ALLOY ALTI Aluminum titanium AlTi 74.861542 ALLOY ALTI3 Aluminum 3-titanium AlTi3 170.621536 ALLOY AUCD Gold cadmium AuCd 309.376495 ALLOY AUCU Gold copper AuCu 260.512512 ALLOY AUCU3 Gold 3-copper AuCu3 387.604492 ALLOY AUSN Gold tin AuSn 315.676514 ALLOY AUSN2 Gold 2-tin AuSn2 434.386505 ALLOY AUSN4 Gold 4-tin AuSn4 671.806519 ALLOY BA2SN 2-Barium tin Ba2Sn 393.369995 ALLOY BI2U 2-Bismuth uranium Bi2U 655.987671 ALLOY BI4U3 4-Bismuth 3-uranium Bi4U3 1550.002319 ALLOY BIU Bismuth uranium BiU 447.007294 ALLOY CA2PB 2-Calcium lead Ca2Pb 287.355988 ALLOY CA2SI 2-Calcium silicon Ca2Si 108.241501 ALLOY CA2SN 2-Calcium tin Ca2Sn 198.865997 ALLOY CA3SB2 3-Calcium 2-antimony Ca3Sb2 363.734009 ALLOY CAMG2 Calcium 2-magnesium CaMg2 88.688004 ALLOY CAPB Calcium lead CaPb 247.278 ALLOY CASI Calcium silicon CaSi 68.163498 ALLOY CASI2 Calcium 2-silicon CaSi2 96.249001 ALLOY CASN Calcium tin CaSn 158.787994 ALLOY CAZN Calcium zinc CaZn 105.468002 ALLOY CAZN2 Calcium 2-zinc CaZn2 170.858002 ALLOY CD11U 11-Cadmium uranium Cd11U 1474.536865 ALLOY CD3AS2 3-Cadmium 2-arsenic As2Cd3 487.073212 -

Strontium and Strontium Compounds
This report contains the collective views of an international group of experts and does not necessarily represent the decisions or the stated policy of the United Nations Environment Programme, the International Labour Organization or the World Health Organization. Concise International Chemical Assessment Document 77 STRONTIUM AND STRONTIUM COMPOUNDS First draft prepared by Mr Peter Watts, Toxicology Advice & Consulting Ltd, Sutton, England; and Mr Paul Howe, Centre for Ecology and Hydrology, Monks Wood, England Published under the joint sponsorship of the United Nations Environment Programme, the International Labour Organization and the World Health Organization, and produced within the framework of the Inter-Organization Programme for the Sound Management of Chemicals. The International Programme on Chemical Safety (IPCS), established in 1980, is a joint venture of the United Nations Environment Programme (UNEP), the International Labour Organization (ILO) and the World Health Organization (WHO). The overall objectives of the IPCS are to establish the scientific basis for assessment of the risk to human health and the environment from exposure to chemicals, through international peer review processes, as a prerequisite for the promotion of chemical safety, and to provide technical assistance in strengthening national capacities for the sound management of chemicals. The Inter-Organization Programme for the Sound Management of Chemicals (IOMC) was established in 1995 by UNEP, ILO, the Food and Agriculture Organization of the United Nations, WHO, the United Nations Industrial Development Organization, the United Nations Institute for Training and Research and the Organisation for Economic Co-operation and Development (Participating Organizations), following recommendations made by the 1992 UN Conference on Environment and Development to strengthen cooperation and increase coordination in the field of chemical safety. -

Danh Mục Số Cas A
CÔNG TY CỔ PHẦN CÔNG NGHIỆP VIỆT XUÂN NHÀ CUNG CẤP KHÍ HELI, KHÍ METAN, KHÍ SF6, KHÍ PROPAN Số 80B Nguyễn Văn Cừ - Long Biên - Hà Nội - Việt Nam www.vietxuangas.com. -

Development and Application of a Green-Chemistry Solution
Downloaded from orbit.dtu.dk on: Dec 20, 2017 Development and application of a green-chemistry solution deposition technique for buffer layer coating on cube-textured metal substrates in view of further deposition of rare-earth based superconductors Pallewatta, Pallewatta G A P; Grivel, Jean-Claude Publication date: 2014 Document Version Peer reviewed version Link back to DTU Orbit Citation (APA): Pallewatta, P. G. A. P., & Grivel, J-C. (2014). Development and application of a green-chemistry solution deposition technique for buffer layer coating on cube-textured metal substrates in view of further deposition of rare-earth based superconductors. Department of Energy Conversion and Storage, Technical University of Denmark. General rights Copyright and moral rights for the publications made accessible in the public portal are retained by the authors and/or other copyright owners and it is a condition of accessing publications that users recognise and abide by the legal requirements associated with these rights. • Users may download and print one copy of any publication from the public portal for the purpose of private study or research. • You may not further distribute the material or use it for any profit-making activity or commercial gain • You may freely distribute the URL identifying the publication in the public portal If you believe that this document breaches copyright please contact us providing details, and we will remove access to the work immediately and investigate your claim. Ph.D. Thesis Development and application of a green-chemistry solution deposition technique for buffer layer coating on cube-textured metal substrates in view of further deposition of rare-earth based superconductors P.