Comparison of Efficacy and Safety of Benidipine With
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
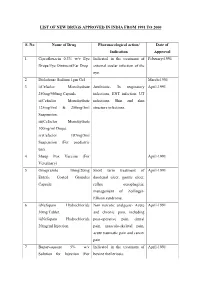
List of New Drugs Approved in India from 1991 to 2000
LIST OF NEW DRUGS APPROVED IN INDIA FROM 1991 TO 2000 S. No Name of Drug Pharmacological action/ Date of Indication Approval 1 Ciprofloxacin 0.3% w/v Eye Indicated in the treatment of February-1991 Drops/Eye Ointment/Ear Drop external ocular infection of the eye. 2 Diclofenac Sodium 1gm Gel March-1991 3 i)Cefaclor Monohydrate Antibiotic- In respiratory April-1991 250mg/500mg Capsule. infections, ENT infection, UT ii)Cefaclor Monohydrate infections, Skin and skin 125mg/5ml & 250mg/5ml structure infections. Suspension. iii)Cefaclor Monohydrate 100mg/ml Drops. iv)Cefaclor 187mg/5ml Suspension (For paediatric use). 4 Sheep Pox Vaccine (For April-1991 Veterinary) 5 Omeprazole 10mg/20mg Short term treatment of April-1991 Enteric Coated Granules duodenal ulcer, gastric ulcer, Capsule reflux oesophagitis, management of Zollinger- Ellison syndrome. 6 i)Nefopam Hydrochloride Non narcotic analgesic- Acute April-1991 30mg Tablet. and chronic pain, including ii)Nefopam Hydrochloride post-operative pain, dental 20mg/ml Injection. pain, musculo-skeletal pain, acute traumatic pain and cancer pain. 7 Buparvaquone 5% w/v Indicated in the treatment of April-1991 Solution for Injection (For bovine theileriosis. Veterinary) 8 i)Kitotifen Fumerate 1mg Anti asthmatic drug- Indicated May-1991 Tablet in prophylactic treatment of ii)Kitotifen Fumerate Syrup bronchial asthma, symptomatic iii)Ketotifen Fumerate Nasal improvement of allergic Drops conditions including rhinitis and conjunctivitis. 9 i)Pefloxacin Mesylate Antibacterial- In the treatment May-1991 Dihydrate 400mg Film Coated of severe infection in adults Tablet caused by sensitive ii)Pefloxacin Mesylate microorganism (gram -ve Dihydrate 400mg/5ml Injection pathogens and staphylococci). iii)Pefloxacin Mesylate Dihydrate 400mg I.V Bottles of 100ml/200ml 10 Ofloxacin 100mg/50ml & Indicated in RTI, UTI, May-1991 200mg/100ml vial Infusion gynaecological infection, skin/soft lesion infection. -

Migraine Prophylaxis: Which Drugs Work and Which Ones Don't
Migraine Prophylaxis: Which Drugs Work and Which Ones Don’t Hans-Christoph Diener, MD Department of Neurology, University Hospital Essen, Essen, Germany. J Gen Intern Med 28(9):1125–6 investigated in two properly powered and conducted studies DOI: 10.1007/s11606-013-2469-2 and found not to be effective compared to placebo.3, 4 Adding © Society of General Internal Medicine 2013 data from poorly controlled and underpowered studies in a meta-analysis, as in this paper, gives the wrong impression that he paper by Shamliyan et al.1 in this issue of JGIM is a very nimodipine is as effective in migraine prevention as propran- T important contribution to headache research. The authors olol. Another example illustrated in this meta-analysis involves conducted a systematic literature review of drug treatment for the gabapentin. Most of the published trials are poorly conducted, underpowered, or have manipulated statistical analyses, such as prevention of episodic migraine. They analysed randomised 5 controlled trials (RCTs) and performed meta-analyses where modified intention-to-treat analyses. The meta-analysis in this appropriate. The two important outcomes examined in the paper indicated possible efficacy. Yet, in the time between the analysis are a ≥ 50 % reduction in migraine frequency and submission and publication of this paper, a recent well-powered adverse events leading to treatment discontinuation. dose finding trial was published investigating four doses of gabapentin as compared with placebo for migraine prevention. 6 What are the strengths of this paper? This trial showed no benefit for gabapentin. Another problem inherent in meta-analyses that this paper The authors are experts in this kind of analysis. -

Optum Essential Health Benefits Enhanced Formulary PDL January
PENICILLINS ketorolac tromethamineQL GENERIC mefenamic acid amoxicillin/clavulanate potassium nabumetone amoxicillin/clavulanate potassium ER naproxen January 2016 ampicillin naproxen sodium ampicillin sodium naproxen sodium CR ESSENTIAL HEALTH BENEFITS ampicillin-sulbactam naproxen sodium ER ENHANCED PREFERRED DRUG LIST nafcillin sodium naproxen DR The Optum Preferred Drug List is a guide identifying oxacillin sodium oxaprozin preferred brand-name medicines within select penicillin G potassium piroxicam therapeutic categories. The Preferred Drug List may piperacillin sodium/ tazobactam sulindac not include all drugs covered by your prescription sodium tolmetin sodium drug benefit. Generic medicines are available within many of the therapeutic categories listed, in addition piperacillin sodium/tazobactam Fenoprofen Calcium sodium to categories not listed, and should be considered Meclofenamate Sodium piperacillin/tazobactam as the first line of prescribing. Tolmetin Sodium Amoxicillin/Clavulanate Potassium LOW COST GENERIC PREFERRED For benefit coverage or restrictions please check indomethacin your benefit plan document(s). This listing is revised Augmentin meloxicam periodically as new drugs and new prescribing LOW COST GENERIC naproxen kit information becomes available. It is recommended amoxicillin that you bring this list of medications when you or a dicloxacillin sodium CARDIOVASCULAR covered family member sees a physician or other penicillin v potassium ACE-INHIBITORS healthcare provider. GENERIC QUINOLONES captopril ANTI-INFECTIVES -

Neurontin (Gabapentin)
Texas Prior Authorization Program Clinical Criteria Drug/Drug Class Gabapentin Clinical Criteria Information Included in this Document Neurontin (gabapentin) • Drugs requiring prior authorization: the list of drugs requiring prior authorization for this clinical criteria • Prior authorization criteria logic: a description of how the prior authorization request will be evaluated against the clinical criteria rules • Logic diagram: a visual depiction of the clinical criteria logic • Supporting tables: a collection of information associated with the steps within the criteria (diagnosis codes, procedure codes, and therapy codes); provided when applicable • References: clinical publications and sources relevant to this clinical criteria Note: Click the hyperlink to navigate directly to that section. Gralise (gabapentin Extended Release) • Drugs requiring prior authorization: the list of drugs requiring prior authorization for this clinical criteria • Prior authorization criteria logic: a description of how the prior authorization request will be evaluated against the clinical criteria rules • Logic diagram: a visual depiction of the clinical criteria logic • Supporting tables: a collection of information associated with the steps within the criteria (diagnosis codes, procedure codes, and therapy codes); provided when applicable • References: clinical publications and sources relevant to this clinical criteria Note: Click the hyperlink to navigate directly to that section. March 29, 2019 Copyright © 2019 Health Information Designs, LLC 1 Horizant -

Dorset Medicines Advisory Group
DORSET CARDIOLOGY WORKING GROUP GUIDELINE FOR CALCIUM CHANNEL BLOCKERS IN HYPERTENSION SUMMARY The pan-Dorset cardiology working group continues to recommend the use of amlodipine (a third generation dihydropyridine calcium-channel blocker) as first choice calcium channel blocker on the pan-Dorset formulary for hypertension. Lercanidipine is second choice, lacidipine third choice and felodipine is fourth choice. This is due to preferable side effect profiles in terms of ankle oedema and relative costs of the preparations. Note: where angina is the primary indication or is a co-morbidity prescribers must check against the specific product characteristics (SPC) for an individual drug to confirm this is a licensed indication. N.B. Lacidipine and lercandipine are only licensed for use in hypertension. Chapter 02.06.02 CCBs section of the Formulary has undergone an evidence-based review. A comprehensive literature search was carried out on NHS Evidence, Medline, EMBASE, Cochrane Database, and UK Duets. This was for recent reviews or meta-analyses on calcium channel blockers from 2009 onwards (comparative efficacy and side effects) and randomised controlled trials (RCTs). REVIEW BACKGROUND Very little good quality evidence exists. No reviews, meta-analyses or RCTs were found covering all calcium channel blockers currently on the formulary. Another limitation was difficulty obtaining full text original papers for some of the references therefore having to use those from more obscure journals instead. Some discrepancies exist between classification of generations of dihydropyridine CCBs, depending upon the year of publication of the reference/authors’ interpretation. Dihydropyridine (DHP) CCBs tend to be more potent vasodilators than non-dihydropyridine (non-DHP) CCBs (diltiazem, verapamil), but the latter have greater inotropic effects. -

Drug Name Plate Number Well Location % Inhibition, Screen Axitinib 1 1 20 Gefitinib (ZD1839) 1 2 70 Sorafenib Tosylate 1 3 21 Cr
Drug Name Plate Number Well Location % Inhibition, Screen Axitinib 1 1 20 Gefitinib (ZD1839) 1 2 70 Sorafenib Tosylate 1 3 21 Crizotinib (PF-02341066) 1 4 55 Docetaxel 1 5 98 Anastrozole 1 6 25 Cladribine 1 7 23 Methotrexate 1 8 -187 Letrozole 1 9 65 Entecavir Hydrate 1 10 48 Roxadustat (FG-4592) 1 11 19 Imatinib Mesylate (STI571) 1 12 0 Sunitinib Malate 1 13 34 Vismodegib (GDC-0449) 1 14 64 Paclitaxel 1 15 89 Aprepitant 1 16 94 Decitabine 1 17 -79 Bendamustine HCl 1 18 19 Temozolomide 1 19 -111 Nepafenac 1 20 24 Nintedanib (BIBF 1120) 1 21 -43 Lapatinib (GW-572016) Ditosylate 1 22 88 Temsirolimus (CCI-779, NSC 683864) 1 23 96 Belinostat (PXD101) 1 24 46 Capecitabine 1 25 19 Bicalutamide 1 26 83 Dutasteride 1 27 68 Epirubicin HCl 1 28 -59 Tamoxifen 1 29 30 Rufinamide 1 30 96 Afatinib (BIBW2992) 1 31 -54 Lenalidomide (CC-5013) 1 32 19 Vorinostat (SAHA, MK0683) 1 33 38 Rucaparib (AG-014699,PF-01367338) phosphate1 34 14 Lenvatinib (E7080) 1 35 80 Fulvestrant 1 36 76 Melatonin 1 37 15 Etoposide 1 38 -69 Vincristine sulfate 1 39 61 Posaconazole 1 40 97 Bortezomib (PS-341) 1 41 71 Panobinostat (LBH589) 1 42 41 Entinostat (MS-275) 1 43 26 Cabozantinib (XL184, BMS-907351) 1 44 79 Valproic acid sodium salt (Sodium valproate) 1 45 7 Raltitrexed 1 46 39 Bisoprolol fumarate 1 47 -23 Raloxifene HCl 1 48 97 Agomelatine 1 49 35 Prasugrel 1 50 -24 Bosutinib (SKI-606) 1 51 85 Nilotinib (AMN-107) 1 52 99 Enzastaurin (LY317615) 1 53 -12 Everolimus (RAD001) 1 54 94 Regorafenib (BAY 73-4506) 1 55 24 Thalidomide 1 56 40 Tivozanib (AV-951) 1 57 86 Fludarabine -

Medicine for Prevention of and Treatment for Arteriosclerosis and Hypertension
Europäisches Patentamt *EP001604664A1* (19) European Patent Office Office européen des brevets (11) EP 1 604 664 A1 (12) EUROPEAN PATENT APPLICATION published in accordance with Art. 158(3) EPC (43) Date of publication: (51) Int Cl.7: A61K 31/4422, A61K 45/06, 14.12.2005 Bulletin 2005/50 A61P 9/00, A61P 9/10, A61P 43/00, A61P 9/12, (21) Application number: 04706359.9 A61P 13/12 (22) Date of filing: 29.01.2004 (86) International application number: PCT/JP2004/000861 (87) International publication number: WO 2004/067003 (12.08.2004 Gazette 2004/33) (84) Designated Contracting States: • IWAI, Masaru AT BE BG CH CY CZ DE DK EE ES FI FR GB GR Shigenobu-cho, Onsen-gun, HU IE IT LI LU MC NL PT RO SE SI SK TR Ehime 791-0204 (JP) Designated Extension States: • SADA, Toshio, Sankyo Company, Limited AL LT LV MK Tokyo 140-8710 (JP) • MIZUNO, Makoto, Sankyo Company, Limited (30) Priority: 31.01.2003 JP 2003022990 Tokyo 140-8710 (JP) 07.02.2003 JP 2003030830 (74) Representative: Gibson, Christian John Robert (71) Applicant: Sankyo Company, Limited Marks & Clerk Tokyo 103-8426 (JP) 90 Long Acre London WC2E 9RA (GB) (72) Inventors: • HORIUCHI, Masatsugu Onsen-gun, Ehime 791-0204 (JP) (54) MEDICINE FOR PREVENTION OF AND TREATMENT FOR ARTERIOSCLEROSIS AND HYPERTENSION (57) A medicament comprising the following composition: (A) an angiotensin II receptor antagonist selected from the group of a compound having a general formula (I), pharmacologically acceptable esters thereof and pharmacolog- ically acceptable salts thereof (for example, olmesartan medoxomil and the like); EP 1 604 664 A1 Printed by Jouve, 75001 PARIS (FR) (Cont. -

Antioxidant Effects of Calcium Antagonists Rat Myocardial
223 Antioxidant Effects of Calcium Antagonists ['Ii] Rat Myocardial Membrane Lipid Peroxidation Hitoshi Sugawara, Katsuyuki Tobise, and Kenjiro Kikuchi We studied the antioxidant effects of nine calcium antagonists (nisoldipine, benidipine, nilvadipine, felo- dipine, nicardipine; nitrendipine, nifedipine, verapamil, and diltiazem) by means of rat myocardial membrane lipid peroxidation with a nonenzymatic active oxygen-generating system (DHF/FeC13-ADP). The order of antioxidant potency of these agents was nilvadipine > nisoldipine > felodipine > nicardipine > verapamil > benidipine. Their IC50 values (,uM) were 25.1, 28.2, 42.0, 150.0, 266.1, and 420.0, re- spectively. In contrast, nitrendipine, nifedipine, and diltiazem had little inhibitory effect on lipid peroxi- dation.These six calcium antagonists could be divided into four types on the basis of their antioxidant mechanisms. Nilvadipine, nisoldipine, and verapamil, which showed antioxidant effects both before and after the addition of active oxygen, and reduced the dihydroxyfumarate (DHF) auto-oxidation rate, were chain-breaking and preventive antioxidants. Felodipine, which showed antioxidant effects both before and after exposure to active oxygen and increased the DHF auto-oxidation rate, was only a chain-break- ing antioxidant. Nicardipine, which showed an antioxidant effect only before exposure to active oxygen and reduced the DHF auto-oxidation rate, was mainly a preventive antioxidant. Benidipine, which showed an antioxidant effect only before exposure to active oxygen and had no appreciable effect on the DHF auto-oxidation rate, could interrupt the chain reaction of lipid peroxidation at the initial step alone. Although these results suggest that the antioxidant properties of some calcium antagonists may be beneficial clinically in protecting against cellular damage caused by lipid peroxidation, further studies are required to establish the antioxidant effects of these agents in vivo. -

Migraine Headache Prophylaxis Hien Ha, Pharmd, and Annika Gonzalez, MD, Christus Santa Rosa Family Medicine Residency Program, San Antonio, Texas
Migraine Headache Prophylaxis Hien Ha, PharmD, and Annika Gonzalez, MD, Christus Santa Rosa Family Medicine Residency Program, San Antonio, Texas Migraines impose significant health and financial burdens. Approximately 38% of patients with episodic migraines would benefit from preventive therapy, but less than 13% take prophylactic medications. Preventive medication therapy reduces migraine frequency, severity, and headache-related distress. Preventive therapy may also improve quality of life and prevent the progression to chronic migraines. Some indications for preventive therapy include four or more headaches a month, eight or more headache days a month, debilitating headaches, and medication- overuse headaches. Identifying and managing environmental, dietary, and behavioral triggers are useful strategies for preventing migraines. First-line med- ications established as effective based on clinical evidence include divalproex, topiramate, metoprolol, propranolol, and timolol. Medications such as ami- triptyline, venlafaxine, atenolol, and nadolol are probably effective but should be second-line therapy. There is limited evidence for nebivolol, bisoprolol, pindolol, carbamazepine, gabapentin, fluoxetine, nicardipine, verapamil, nimodipine, nifedipine, lisinopril, and candesartan. Acebutolol, oxcarbazepine, lamotrigine, and telmisartan are ineffective. Newer agents target calcitonin gene-related peptide pain transmission in the migraine pain pathway and have recently received approval from the U.S. Food and Drug Administration; how- ever, more studies of long-term effectiveness and adverse effects are needed. The complementary treatments petasites, feverfew, magnesium, and riboflavin are probably effective. Nonpharmacologic therapies such as relaxation training, thermal biofeedback combined with relaxation training, electromyographic feedback, and cognitive behavior therapy also have good evidence to support their use in migraine prevention. (Am Fam Physician. 2019; 99(1):17-24. -

Original Article Comparison of Therapeutic Effects of Two Ccbs on Glaucoma and Analysis of Their Possible Mechanisms
Int J Clin Exp Med 2017;10(7):10560-10564 www.ijcem.com /ISSN:1940-5901/IJCEM0056272 Original Article Comparison of therapeutic effects of two CCBs on glaucoma and analysis of their possible mechanisms Tao Liang, Lingyun Zhang, Yanhua Gao, Yanru Xiang, Yan Gao Department of Ophthalmology, The Affiliated Hospital of Qingdao University, Qingdao, Shandong, China Received April 26, 2017; Accepted May 26, 2017; Epub July 15, 2017; Published July 30, 2017 Abstract: Objective: To respectively compare the therapeutic effects of nimodipine and nifedipine on glaucoma, and then analyze the possible protective effects of these two calcium channel blockers (CCBs) on glaucomatous retinal ganglion cells (RGCs). Methods: Fifty-four patients with glaucoma were divided into control group (n=15), treat- ment group 1 (n=20) and treatment group 2 (n=19) in accordance with a random number table. General clinical treatment of glaucoma was performed in all three groups, while nimodipine was applied in treatment group 1 and nifedipine was applied in treatment group 2. The therapeutic effects and incidence of adverse reactions (intraocular pressure (IOP), eyesight, retinal light sensitivity, progressive visual field damage and adverse drug reaction) were compared among the three groups. Results: There were no significant differences in IOP and eyesight before and after treatment among the three groups (P>0.05). The retinal light sensitivity in control group began to decline from the sixth month after treatment, which was significantly different from treatment group 1 and treatment group 2 (P=0.03; P=0.04). The survival curve of visual field damage indicated that the visual field damage in control group was obviously more serious than that in the two treatment groups with the increase of sick time (P=0.03). -

Benidipine, a Dihydropyridine-Ca2+ Channel Blocker, Increases the Endothelial Differentiation of Endothelial Progenitor Cells in Vitro
1047 Hypertens Res Vol.29 (2006) No.12 p.1047-1054 Original Article Benidipine, a Dihydropyridine-Ca2+ Channel Blocker, Increases the Endothelial Differentiation of Endothelial Progenitor Cells In Vitro Hiroshi ANDO1), Kosuke NAKANISHI2), Mami SHIBATA1), Kazuhide HASEGAWA2), Kozo YAO2), and Hiromasa MIYAJI1) Benidipine is a dihydropyridine-Ca2+ channel blocker used in the treatment of hypertension and angina pec- toris. In the present study, we examined the effects of benidipine on the endothelial differentiation of circu- lating endothelial progenitor cells (EPCs) using an in vitro culture method. Peripheral blood derived mononuclear cells (PBMCs) containing EPCs were isolated from C57BL/6 mice, and then the cells were cul- tured on vitronectin/gelatin-coated slide glasses. After 7 days of culture, endothelial cells differentiated from EPCs were identified as adherent cells with 1,1′-dioctadecyl-3,3,3′,3′-tetramethyl-indocarbocyanine–labeled acetylated low density lipoprotein (DiI-Ac-LDL) uptake and lectin binding under a fluorescent microscope. Incubation of PBMCs for 7 days with benidipine (0.01–1 µmol/l) significantly increased the number of DiI- Ac-LDL+/fluorescein isothiocyanate-lectin (FITC-Lectin)+ cells. Wortmannin, a phosphoinositide-3 kinase (PI3K) inhibitor, selectively attenuated the effect of benidipine on the endothelial differentiation. In addition, benidipine treatment augmented the phosphorylation of Akt, indicating that the PI3K/Akt pathway contrib- uted, at least in part, to the endothelial differentiation induced by benidipine. These results suggest that the treatment with benidipine may increase the endothelial differentiation of circulating EPCs and contribute to endothelial protection, prevention of cardiovascular disease, and/or an improvement of the prognosis after ischemic damage. -

Benidipine Reduces Albuminuria and Plasma Aldosterone in Mild-To-Moderate Stage Chronic Kidney Disease with Albuminuria
Hypertension Research (2011) 34, 268–273 & 2011 The Japanese Society of Hypertension All rights reserved 0916-9636/11 $32.00 www.nature.com/hr ORIGINAL ARTICLE Benidipine reduces albuminuria and plasma aldosterone in mild-to-moderate stage chronic kidney disease with albuminuria Masanori Abe1, Kazuyoshi Okada1, Noriaki Maruyama1, Shiro Matsumoto1, Takashi Maruyama1, Takayuki Fujita1, Koichi Matsumoto1 and Masayoshi Soma1,2 Benidipine inhibits both L- and T-type Ca channels, and has been shown to dilate the efferent arterioles as effectively as the afferent arterioles. In this study, we conducted an open-label and randomized trial to compare the effects of benidipine with those of amlodipine on blood pressure (BP), albuminuria and aldosterone concentration in hypertensive patients with mild-to- moderate stage chronic kidney disease (CKD). Patients with BPX130/80 mm Hg, with estimated glomerular filtration rate (eGFR) of 30–90 ml minÀ1 per 1.73 m2, and with albuminuria430 mg per g creatinine (Cr), despite treatment with the maximum recommended dose of angiotensin II receptor blockers (ARBs) were randomly assigned to two groups. Patients received either of the following two treatment regimens: 2 mg per day benidipine, which was increased up to a dose of 8 mg per day (n¼52), or 2.5 mg per day amlodipine, which was increased up to a dose of 10 mg per day (n¼52). After 6 months of treatment, a significant and comparable reduction in the systolic and diastolic BP was observed in both groups. The decrease in the urinary albumin to Cr ratio in the benidipine group was significantly lower than that in the amlodipine group.