Helium Adsorption on Lithium Substrates
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

An Alternate Graphical Representation of Periodic Table of Chemical Elements Mohd Abubakr1, Microsoft India (R&D) Pvt
An Alternate Graphical Representation of Periodic table of Chemical Elements Mohd Abubakr1, Microsoft India (R&D) Pvt. Ltd, Hyderabad, India. [email protected] Abstract Periodic table of chemical elements symbolizes an elegant graphical representation of symmetry at atomic level and provides an overview on arrangement of electrons. It started merely as tabular representation of chemical elements, later got strengthened with quantum mechanical description of atomic structure and recent studies have revealed that periodic table can be formulated using SO(4,2) SU(2) group. IUPAC, the governing body in Chemistry, doesn‟t approve any periodic table as a standard periodic table. The only specific recommendation provided by IUPAC is that the periodic table should follow the 1 to 18 group numbering. In this technical paper, we describe a new graphical representation of periodic table, referred as „Circular form of Periodic table‟. The advantages of circular form of periodic table over other representations are discussed along with a brief discussion on history of periodic tables. 1. Introduction The profoundness of inherent symmetry in nature can be seen at different depths of atomic scales. Periodic table symbolizes one such elegant symmetry existing within the atomic structure of chemical elements. This so called „symmetry‟ within the atomic structures has been widely studied from different prospects and over the last hundreds years more than 700 different graphical representations of Periodic tables have emerged [1]. Each graphical representation of chemical elements attempted to portray certain symmetries in form of columns, rows, spirals, dimensions etc. Out of all the graphical representations, the rectangular form of periodic table (also referred as Long form of periodic table or Modern periodic table) has gained wide acceptance. -

Atomic Spectra of Alkali Elements
Atomic Spectra of Alkali Elements S. R. Kulkarni April 10, 2020 Rydberg primarily focused on studying the lines of alkali metals (Lithium, Potassium and Sodium).1 Rydberg organized the various features by their appearance on the pho- tographs:The alkali spectra were more complicated than that of hydrogen. Rydberg recog- nized that were three different types of lines: lines which looked \sharp" (on photographic plates), \principal" (strong lines that showed up in emission and absorption) and those which appeared ‘diffuse”. These series were abbreviated to S, P, D. Later \Fundamental" (F) was added. As noted earlier, Rydberg preferred to work with wavenumbers. Using data from Liveing and Deware he recast Angstrom's formula as follows: N k = k − (1) n 1 (n + µ)2 where kn is the wavenumber of the nth line in a given series. Rydberg kept N fixed to the value measured by Balmer with k1 µ being free parameters. Rydberg found the following formulae for Lithium R ks = ks − ; 2; 3; 4; ::: (2) n 1 (n + S)2 R kp = kp − 1; 2; 3; ::: (3) n 1 (n + P )2 R kd = kd − 2; 3; 4; ::: (4) n 1 (n + D)2 s −1 p −1 where S = 0:5951, P = 0:9596, D = 0:9974, k1 = 28601:6 cm k1 = 43487:7 cm , d −1 k1 = 28598:5 cm and we have switched to the modern notation in which N is replaced by R (the value he found was R = 109721:6 cm−1). Rydberg had confidence in the data that he was able to find a deeper connection between the constants between the series. -

The Development of the Periodic Table and Its Consequences Citation: J
Firenze University Press www.fupress.com/substantia The Development of the Periodic Table and its Consequences Citation: J. Emsley (2019) The Devel- opment of the Periodic Table and its Consequences. Substantia 3(2) Suppl. 5: 15-27. doi: 10.13128/Substantia-297 John Emsley Copyright: © 2019 J. Emsley. This is Alameda Lodge, 23a Alameda Road, Ampthill, MK45 2LA, UK an open access, peer-reviewed article E-mail: [email protected] published by Firenze University Press (http://www.fupress.com/substantia) and distributed under the terms of the Abstract. Chemistry is fortunate among the sciences in having an icon that is instant- Creative Commons Attribution License, ly recognisable around the world: the periodic table. The United Nations has deemed which permits unrestricted use, distri- 2019 to be the International Year of the Periodic Table, in commemoration of the 150th bution, and reproduction in any medi- anniversary of the first paper in which it appeared. That had been written by a Russian um, provided the original author and chemist, Dmitri Mendeleev, and was published in May 1869. Since then, there have source are credited. been many versions of the table, but one format has come to be the most widely used Data Availability Statement: All rel- and is to be seen everywhere. The route to this preferred form of the table makes an evant data are within the paper and its interesting story. Supporting Information files. Keywords. Periodic table, Mendeleev, Newlands, Deming, Seaborg. Competing Interests: The Author(s) declare(s) no conflict of interest. INTRODUCTION There are hundreds of periodic tables but the one that is widely repro- duced has the approval of the International Union of Pure and Applied Chemistry (IUPAC) and is shown in Fig.1. -

Lithium Data Sheet
98 LITHIUM (Data in metric tons of lithium content unless otherwise noted) Domestic Production and Use: The only lithium production in the United States was from a brine operation in Nevada. Two companies produced a wide range of downstream lithium compounds in the United States from domestic or imported lithium carbonate, lithium chloride, and lithium hydroxide. Domestic production data were withheld to avoid disclosing company proprietary data. Although lithium markets vary by location, global end-use markets are estimated as follows: batteries, 65%; ceramics and glass, 18%; lubricating greases, 5%; polymer production, 3%; continuous casting mold flux powders, 3%; air treatment, 1%; and other uses, 5%. Lithium consumption for batteries has increased significantly in recent years because rechargeable lithium batteries are used extensively in the growing market for portable electronic devices and increasingly are used in electric tools, electric vehicles, and grid storage applications. Lithium minerals were used directly as ore concentrates in ceramics and glass applications. Salient Statistics—United States: 2015 2016 2017 2018 2019e Production W W W W W Imports for consumption 2,750 3,140 3,330 3,420 2,500 Exports 1,790 1,520 1,960 1,660 1,700 Consumption, estimated1 2,000 3,000 3,000 3,000 2,000 Price, annual average, battery-grade lithium carbonate, dollars per metric ton2 6,500 8,650 15,000 17,000 13,000 Employment, mine and mill, number 70 70 70 70 70 Net import reliance3 as a percentage of estimated consumption >25 >50 >50 >50 >25 Recycling: One domestic company has recycled lithium metal and lithium-ion batteries since 1992 at its facility in British Columbia, Canada. -

Of the Periodic Table
of the Periodic Table teacher notes Give your students a visual introduction to the families of the periodic table! This product includes eight mini- posters, one for each of the element families on the main group of the periodic table: Alkali Metals, Alkaline Earth Metals, Boron/Aluminum Group (Icosagens), Carbon Group (Crystallogens), Nitrogen Group (Pnictogens), Oxygen Group (Chalcogens), Halogens, and Noble Gases. The mini-posters give overview information about the family as well as a visual of where on the periodic table the family is located and a diagram of an atom of that family highlighting the number of valence electrons. Also included is the student packet, which is broken into the eight families and asks for specific information that students will find on the mini-posters. The students are also directed to color each family with a specific color on the blank graphic organizer at the end of their packet and they go to the fantastic interactive table at www.periodictable.com to learn even more about the elements in each family. Furthermore, there is a section for students to conduct their own research on the element of hydrogen, which does not belong to a family. When I use this activity, I print two of each mini-poster in color (pages 8 through 15 of this file), laminate them, and lay them on a big table. I have students work in partners to read about each family, one at a time, and complete that section of the student packet (pages 16 through 21 of this file). When they finish, they bring the mini-poster back to the table for another group to use. -

Expansion of Domestic Production of Lithium Carbonate and Lithium Hydroxide to Supply US Battery Industry
Expansion of Domestic Production of Lithium Carbonate and Lithium Hydroxide to Supply US Battery Industry John Groves Jeff Davis Chemetall Foote Corp May 11, 2011 Project ID# ARRAVT010 This presentation does not contain any proprietary, confidential, or otherwise restricted information Overview Expand Lithium Raw Material Base in US Timeline Barriers Start Date: April 14, 2010 Geothermal Resource Strength and Viability of End Date: February, 2013 Geothermal resource Partners Budget Engineering: BE&K® (a KBR DOE Share - $28.4 million company) Rockwood Share - $39.5 Environmental Assessment: million Nevada Bureau of Land Mgmt Relevance: Domestic Source of Strategic Materials • Objectives – Expand domestic lithium carbonate and lithium hydroxide production to supply the US electric drive automotive market. – Deliver high quality lithium products to battery component manufacturers to produce high quality lithium ion batteries. – Create construction jobs over the first two years in the US and permanent jobs for production of lithium raw materials. – Stimulate the US economy with worthwhile long term benefits that will support the conversion to electric drive mobility. Relevance: Domestic Source of Strategic Materials • Milestones – Deliver battery grade lithium products to the DOE and component manufacturers in 2012 from this project. – Maintain the long term viability of domestic production of lithium raw materials by lowering operating cost and at the same time reducing fossil fuel based energy consumption. – Job Creation throughout 2010-2012 -
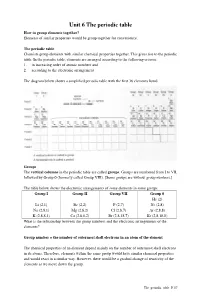
Unit 6 the Periodic Table How to Group Elements Together? Elements of Similar Properties Would Be Group Together for Convenience
Unit 6 The periodic table How to group elements together? Elements of similar properties would be group together for convenience. The periodic table Chemists group elements with similar chemical properties together. This gives rise to the periodic table. In the periodic table, elements are arranged according to the following criteria: 1. in increasing order of atomic numbers and 2. according to the electronic arrangement The diagram below shows a simplified periodic table with the first 36 elements listed. Groups The vertical columns in the periodic table are called groups . Groups are numbered from I to VII, followed by Group 0 (formerly called Group VIII). [Some groups are without group numbers.] The table below shows the electronic arrangements of some elements in some groups. Group I Group II Group VII Group 0 He (2) Li (2,1) Be (2,2) F (2,7) Ne (2,8) Na (2,8,1) Mg (2,8,2) Cl (2,8,7) Ar (2,8,8) K (2,8,8,1) Ca (2,8,8,2) Br (2,8,18,7) Kr (2,8,18,8) What is the relationship between the group numbers and the electronic arrangements of the elements? Group number = the number of outermost shell electrons in an atom of the element The chemical properties of an element depend mainly on the number of outermost shell electrons in its atoms. Therefore, elements within the same group would have similar chemical properties and would react in a similar way. However, there would be a gradual change of reactivity of the elements as we move down the group. -

Global Lithium Sources—Industrial Use and Future in the Electric Vehicle Industry: a Review
resources Review Global Lithium Sources—Industrial Use and Future in the Electric Vehicle Industry: A Review Laurence Kavanagh * , Jerome Keohane, Guiomar Garcia Cabellos, Andrew Lloyd and John Cleary EnviroCORE, Department of Science and Health, Institute of Technology Carlow, Kilkenny, Road, Co., R93-V960 Carlow, Ireland; [email protected] (J.K.); [email protected] (G.G.C.); [email protected] (A.L.); [email protected] (J.C.) * Correspondence: [email protected] Received: 28 July 2018; Accepted: 11 September 2018; Published: 17 September 2018 Abstract: Lithium is a key component in green energy storage technologies and is rapidly becoming a metal of crucial importance to the European Union. The different industrial uses of lithium are discussed in this review along with a compilation of the locations of the main geological sources of lithium. An emphasis is placed on lithium’s use in lithium ion batteries and their use in the electric vehicle industry. The electric vehicle market is driving new demand for lithium resources. The expected scale-up in this sector will put pressure on current lithium supplies. The European Union has a burgeoning demand for lithium and is the second largest consumer of lithium resources. Currently, only 1–2% of worldwide lithium is produced in the European Union (Portugal). There are several lithium mineralisations scattered across Europe, the majority of which are currently undergoing mining feasibility studies. The increasing cost of lithium is driving a new global mining boom and should see many of Europe’s mineralisation’s becoming economic. The information given in this paper is a source of contextual information that can be used to support the European Union’s drive towards a low carbon economy and to develop the field of research. -

Periodic Table 1 Periodic Table
Periodic table 1 Periodic table This article is about the table used in chemistry. For other uses, see Periodic table (disambiguation). The periodic table is a tabular arrangement of the chemical elements, organized on the basis of their atomic numbers (numbers of protons in the nucleus), electron configurations , and recurring chemical properties. Elements are presented in order of increasing atomic number, which is typically listed with the chemical symbol in each box. The standard form of the table consists of a grid of elements laid out in 18 columns and 7 Standard 18-column form of the periodic table. For the color legend, see section Layout, rows, with a double row of elements under the larger table. below that. The table can also be deconstructed into four rectangular blocks: the s-block to the left, the p-block to the right, the d-block in the middle, and the f-block below that. The rows of the table are called periods; the columns are called groups, with some of these having names such as halogens or noble gases. Since, by definition, a periodic table incorporates recurring trends, any such table can be used to derive relationships between the properties of the elements and predict the properties of new, yet to be discovered or synthesized, elements. As a result, a periodic table—whether in the standard form or some other variant—provides a useful framework for analyzing chemical behavior, and such tables are widely used in chemistry and other sciences. Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. -

The Noble Gases
INTERCHAPTER K The Noble Gases When an electric discharge is passed through a noble gas, light is emitted as electronically excited noble-gas atoms decay to lower energy levels. The tubes contain helium, neon, argon, krypton, and xenon. University Science Books, ©2011. All rights reserved. www.uscibooks.com Title General Chemistry - 4th ed Author McQuarrie/Gallogy Artist George Kelvin Figure # fig. K2 (965) Date 09/02/09 Check if revision Approved K. THE NOBLE GASES K1 2 0 Nitrogen and He Air P Mg(ClO ) NaOH 4 4 2 noble gases 4.002602 1s2 O removal H O removal CO removal 10 0 2 2 2 Ne Figure K.1 A schematic illustration of the removal of O2(g), H2O(g), and CO2(g) from air. First the oxygen is removed by allowing the air to pass over phosphorus, P (s) + 5 O (g) → P O (s). 20.1797 4 2 4 10 2s22p6 The residual air is passed through anhydrous magnesium perchlorate to remove the water vapor, Mg(ClO ) (s) + 6 H O(g) → Mg(ClO ) ∙6 H O(s), and then through sodium hydroxide to remove 18 0 4 2 2 4 2 2 the carbon dioxide, NaOH(s) + CO2(g) → NaHCO3(s). The gas that remains is primarily nitrogen Ar with about 1% noble gases. 39.948 3s23p6 36 0 The Group 18 elements—helium, K-1. The Noble Gases Were Kr neon, argon, krypton, xenon, and Not Discovered until 1893 83.798 radon—are called the noble gases 2 6 4s 4p and are noteworthy for their rela- In 1893, the English physicist Lord Rayleigh noticed 54 0 tive lack of chemical reactivity. -

Periodic Table Key Concepts
Periodic Table Key Concepts Periodic Table Basics The periodic table is a table of all the elements which make up matter Elements initially grouped in a table by Dmitri Mendeleev Symbols – each element has a symbol which is either a Capital Letter or a Capital Letter followed by a lower case letter Atomic Number – the number above an element’s symbol which shows the number of protons Atomic Mass – the number found below an elements symbol which shows the mass of the element. Mass = the number of protons + the number of neutrons Metals – the elements which have the properties of malleability, luster, and conductivity o These elements are good conductors of electricity & heat. o Found to the left of the zig-zag line on the periodic table Nonmetals – do not have the properties of metals. Found to the right of the zig-zag line Metalloids – elements found along the zig-zag line of the periodic table and have some properties of metals and nonmetals (B, Si, Ge, As, Sb, Te, and Po) Groups The columns going up and down (There are 18 groups) Group 1: Hydrogen, Lithium, Sodium, Potassium, Rubidium, Cesium, and Francium Elements arranged so that elements with similar properties would be in the same group. o Group 1 Alkali Metals - highly reactive metals o Group 2 Alkali Earth Metals – reactive metals o Group 3-12 Transition Metals o Group 17 Halogens – highly reactive non-metals o Group 18 Noble Gases - do not react or combine with any other elements. Elements are grouped according to their properties or reactivity Reactivity is determined by the number of electrons in an element’s outer energy level These electrons are called valence electrons Periods The rows that run from left to right on the periodic table (There are 7 periods) Period 1 contains 2 elements, Hydrogen and Helium. -

Expansion of Domestic Production of Lithium Carbonate and Lithium Hydroxide to Supply US Battery Industry
Expansion of Domestic Production of Lithium Carbonate and Lithium Hydroxide to Supply US Battery Industry Austin Devaney Jeff Davis Chemetall Foote Corp May 16, 2012 Project ID# ARRAVT010 This presentation does not contain any proprietary, confidential, or otherwise restricted information Overview Expand Lithium Raw Material Base in US Timeline Barriers Start Date: April 14, 2010 Geothermal Resource Strength and Viability of End Date: December, 2013 Geothermal resource Partners Budget Engineering: BE&K (a KBR DOE Share - $28.4 million company) Rockwood Share - $46.0 Environmental Assessment: million Nevada Bureau Land Mgmt Relevance: Domestic Source of Strategic Materials • Objectives – Expand domestic lithium carbonate and lithium hydroxide production to supply the US electric drive automotive market. – Deliver high quality lithium products to battery component manufacturers to produce high quality lithium ion batteries. – Create construction jobs over three years in the US and permanent jobs for production of lithium raw materials. – Stimulate the US economy with worthwhile long term benefits that will support the conversion to electric drive mobility. Relevance: Domestic Source of Strategic Materials • Milestones – Deliver battery grade lithium products to the DOE and component manufacturers in 2012 from this project. – Maintain the long term viability of domestic production of lithium raw materials by lowering operating cost and at the same time reducing fossil fuel based energy consumption. – Job Creation throughout 2010-2013 for