Neuromodulation Today Vagus Nerve Stimulation Neural Network
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Regional Differences in Serotonin Content in the Nucleus of the Solitary Tract of Male Rats After Hypovolemia Produced by Polyethylene Glycol
J Physiol Sci (2013) 63:39–46 DOI 10.1007/s12576-012-0229-4 ORIGINAL PAPER Regional differences in serotonin content in the nucleus of the solitary tract of male rats after hypovolemia produced by polyethylene glycol J. Thomas Curtis • Michael B. Anderson • Kathleen S. Curtis Received: 30 June 2012 / Accepted: 6 August 2012 / Published online: 4 September 2012 Ó The Physiological Society of Japan and Springer 2012 Abstract Serotonin (5-HT) has been implicated in cen- and hormonal responses, occur over a comparatively long trally mediated compensatory responses to volume loss in period of time, whereas changes in autonomic nervous rats. Accordingly, we hypothesized that slowly developing, system activity occur in a much shorter time frame. Short- non-hypotensive hypovolemia increases serotonin in the term responses, in particular, involve low pressure baro- hindbrain nucleus of the solitary tract (NTS). We produced receptors in the heart and great veins which signal volume loss in adult male rats by administering hyperon- increased or decreased volume, and subsequently activate cotic polyethylene glycol (PEG) and then assessed 5-HT hindbrain baroreflex pathways, the connectivity and neu- levels in the NTS using measurements of tissue 5-HT rochemistry of which are well defined (for review, see [1]). content or 5-HT immunohistochemistry. The results show Activation in these pathways has been reported in response selective increases of 5-HT in the caudal NTS after PEG to increased volume [2–4] and, perhaps more familiarly, in treatment, but no change in the primary 5-HT metabolite, response to decreased volume [5–8], such as that produced 5-HIAA. -

The Responsiveness of Neurons in the Insular Gustatory Cortex of the Macaque Monkey Is Independent of Hunger
Physiology & Behavior, Vol. 42, pp. 223--229. Copyright ~ Pergamon Press plc, 1988. Printed in the U.S.A. 0031-9384/88 $3.00 + .00 The Responsiveness of Neurons in the Insular Gustatory Cortex of the Macaque Monkey is Independent of Hunger SIMON YAXLEY, EDMUND T. ROLLS 1 AND ZENON J. SIENKIEW1CZ University of Oxfi)rd, Department of Experimental Psychology South Parks Road, Oxford OXI 3UD, England Received 19 August 1986 YAXLEY, S., E. T. ROLLS AND Z. J. S1ENKIEWICZ. The responsiveness of neurons in the insular gustatory cortex of the macaque monkey is independent of hunger. PHYSIOL BEHAV 42(3) 223-229. 1988.--(1) In order to determine whether the responsiveness of neurons in the insular gustatory cortex is influenced by hunger, neuronal activity was analysed in it while macaque monkeys (Maeaca jhscicularis) were fed to satiety. The responses of single neurons in the insular gustatory cortex to the protypical taste stimuli glucose, NaCI, HCI and quinine HCI, and to fruit juice, were measured before, while, and after the monkey was fed to satiety with glucose or fruit juice. (2) While behavior turned from avid acceptance to active rejection upon repletion, the responsiveness of the neurons to the stimulus array, including the satiating solution, was unmodified. (3) It is concluded that in the insular gustatory cortex, neuronal responses to gustatory stimuli are not influenced by the normal transition from hunger to satiety. This is in contrast to the responses of a population of neurons recorded in the hypothalamus, which only respond to the taste of food when the monkey is hungry. -

Central Neurocircuits Regulating Food Intake in Response to Gut Inputs—Preclinical Evidence
nutrients Review Central Neurocircuits Regulating Food Intake in Response to Gut Inputs—Preclinical Evidence Kirsteen N. Browning * and Kaitlin E. Carson Department of Neural and Behavioral Sciences, Penn State College of Medicine, Hershey, PA 17033, USA; [email protected] * Correspondence: [email protected]; Tel.: +1-717-531-8267 Abstract: The regulation of energy balance requires the complex integration of homeostatic and hedonic pathways, but sensory inputs from the gastrointestinal (GI) tract are increasingly recognized as playing critical roles. The stomach and small intestine relay sensory information to the central nervous system (CNS) via the sensory afferent vagus nerve. This vast volume of complex sensory information is received by neurons of the nucleus of the tractus solitarius (NTS) and is integrated with responses to circulating factors as well as descending inputs from the brainstem, midbrain, and forebrain nuclei involved in autonomic regulation. The integrated signal is relayed to the adjacent dorsal motor nucleus of the vagus (DMV), which supplies the motor output response via the efferent vagus nerve to regulate and modulate gastric motility, tone, secretion, and emptying, as well as intestinal motility and transit; the precise coordination of these responses is essential for the control of meal size, meal termination, and nutrient absorption. The interconnectivity of the NTS implies that many other CNS areas are capable of modulating vagal efferent output, emphasized by the many CNS disorders associated with dysregulated GI functions including feeding. This review will summarize the role of major CNS centers to gut-related inputs in the regulation of gastric function Citation: Browning, K.N.; Carson, with specific reference to the regulation of food intake. -

Optical Vagus Nerve Modulation of Heart and Respiration Via Heart‑Injected Retrograde AAV Arjun K
www.nature.com/scientificreports OPEN Optical vagus nerve modulation of heart and respiration via heart‑injected retrograde AAV Arjun K. Fontaine1,3,8*, Gregory L. Futia1,8, Pradeep S. Rajendran4,5, Samuel F. Littich1,3, Naoko Mizoguchi2,7, Kalyanam Shivkumar4,5, Jefrey L. Ardell4,5, Diego Restrepo2,9, John H. Caldwell2,9, Emily A. Gibson1,9 & Richard F. f Weir1,3,6,9 Vagus nerve stimulation has shown many benefts for disease therapies but current approaches involve imprecise electrical stimulation that gives rise to of‑target efects, while the functionally relevant pathways remain poorly understood. One method to overcome these limitations is the use of optogenetic techniques, which facilitate targeted neural communication with light‑sensitive actuators (opsins) and can be targeted to organs of interest based on the location of viral delivery. Here, we tested whether retrograde adeno‑associated virus (rAAV2‑retro) injected in the heart can be used to selectively express opsins in vagus nerve fbers controlling cardiac function. Furthermore, we investigated whether perturbations in cardiac function could be achieved with photostimulation at the cervical vagus nerve. Viral injection in the heart resulted in robust, primarily aferent, opsin reporter expression in the vagus nerve, nodose ganglion, and brainstem. Photostimulation using both one‑ photon stimulation and two‑photon holography with a GRIN‑lens incorporated nerve cuf, was tested on the pilot‑cohort of injected mice. Changes in heart rate, surface electrocardiogram, and respiratory responses were observed in response to both one‑ and two‑photon photostimulation. The results demonstrate feasibility of retrograde labeling for organ targeted optical neuromodulation. Improved techniques are necessary for selective modulation of peripheral nerve fbers, particularly within the autonomic nervous system. -
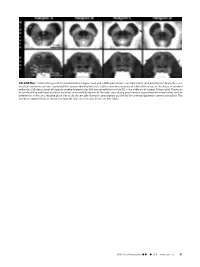
ON-LINE FIG 1. Selected Images of the Caudal Midbrain (Upper Row
ON-LINE FIG 1. Selected images of the caudal midbrain (upper row) and middle pons (lower row) from 4 of 13 total postmortem brains illustrate excellent anatomic contrast reproducibility across individual datasets. Subtle variations are present. Note differences in the shape of cerebral peduncles (24), decussation of superior cerebellar peduncles (25), and spinothalamic tract (12) in the midbrain of subject D (top right). These can be attributed to individual anatomic variation, some mild distortion of the brain stem during procurement at postmortem examination, and/or differences in the axial imaging plane not easily discernable during its prescription parallel to the anterior/posterior commissure plane. The numbers in parentheses in the on-line legends refer to structures in the On-line Table. AJNR Am J Neuroradiol ●:●●2019 www.ajnr.org E1 ON-LINE FIG 3. Demonstration of the dentatorubrothalamic tract within the superior cerebellar peduncle (asterisk) and rostral brain stem. A, Axial caudal midbrain image angled 10° anterosuperior to posteroinferior relative to the ACPC plane demonstrates the tract traveling the midbrain to reach the decussation (25). B, Coronal oblique image that is perpendicular to the long axis of the hippocam- pus (structure not shown) at the level of the ventral superior cerebel- lar decussation shows a component of the dentatorubrothalamic tract arising from the cerebellar dentate nucleus (63), ascending via the superior cerebellar peduncle to the decussation (25), and then enveloping the contralateral red nucleus (3). C, Parasagittal image shows the relatively long anteroposterior dimension of this tract, which becomes less compact and distinct as it ascends toward the thalamus. ON-LINE FIG 2. -

Tonsillar Branch of the Glossopharyngeal Nerve and the Superior Laryngeal Nerve in Lamb
THE JOURNAL OF COMPARATIVE NEUROLOGY 245:471-482 (1986) Central Connections of the Lingual- Tonsillar Branch of the Glossopharyngeal Nerve and the Superior Laryngeal Nerve in Lamb ROBERT D. SWEAZEY AND ROBERT M. BRADLEY Department of Oral Biology, School of Dentistry (R.D.S., R.M.B.), The Department of Physiology, School of Medicine (R.M.B.), University of Michigan, Ann Arbor, Michigan 48109 ABSTRACT Afferent and efferent central connections of the lingual-tonsillar branch of the glossopharyngeal nerve (LT-IX) and the superior laryngeal nerve (SLN) in the lamb were traced with horseradish peroxidase (HRP) histochem- istry. After entering the brainstem, most LT-IX and SLN afferent fibers turned caudally in the solitary tract (ST). Some afferent fibers of LT-IX terminated in the medial nucleus of the solitary tract slightly caudal to their level of entry. The remaining fibers projected to the dorsolateral, ventrolat- eral, and interstitial areas of the nucleus of the solitary tract (NST) at the level of the area postrema. Superior laryngeal nerve afferent fibers termi- nated extensively in the medial and ventral NST at levels near the rostral pole of the area postrema. Further caudal, near the level of obex, SLN afferent terminations were concentrated in the region ventrolateral to the ST and in the interstitial NST. The caudal extent of LT-IX and the rostral extent of SLN terminals projected to similar levels of the NST, but only a relatively small proportion of the total projections overlapped. Lingual- tonsillar and SLN fibers also coursed rostrally to terminate in the caudal pons within and medial to the dorsomedial principal sensory trigeminal nucleus. -

The Heart Side of Brain Neuromodulation
The heart side of brain neuromodulation rsta.royalsocietypublishing.org Simone Rossi1, Emiliano Santarnecchi1,2, Gaetano Valenza3,4,5 and Monica Ulivelli1 Review 1Department of Medicine, Surgery and Neuroscience, Unit of Neurology and Clinical Neurophysiology, Brain Investigation and Cite this article: Rossi S, Santarnecchi E, Neuromodulation Lab. (Si-BIN Lab.), Azienda Ospedaliera Valenza G, Ulivelli M. 2016 The heart side of Universitaria Senese, University of Siena, 53100 Siena, Italy Phil.Trans.R.Soc.A brain neuromodulation. 2Berenson–Allen Center for Noninvasive Brain Stimulation, 374:20150187. Department of Neurology, Beth Israel Deaconess Medical Center, http://dx.doi.org/10.1098/rsta.2015.0187 Boston, MA 02215, USA 3Department of Information Engineering, and Research Center Accepted: 11 January 2016 E. Piaggio, University of Pisa, 56122 Pisa, Italy 4Neuroscience Statistics Research Lab, Harvard Medical School, One contribution of 16 to a theme issue Massachusetts General Hospital, Boston, MA 02115, USA ‘Uncovering brain–heart information through 5Massachusetts Institute of Technology, Cambridge, MA 02139, USA advanced signal and image processing’. SR, 0000-0001-6697-9459 Subject Areas: biomedical engineering Neuromodulation refers to invasive, minimally invasive or non-invasive techniques to stimulate Keywords: discrete cortical or subcortical brain regions with deep brain stimulation, vagal nerve therapeutic purposes in otherwise intractable patients: for example, thousands of advanced Parkinsonian stimulation, repetitive transcranial -

(V,) Type in the Nucleus of the Solitary Tract of the Rat Mediate Direct Neuronal Excitation
The Journal of Neuroscience, November 1999, 9(11): 39293936 Vasopressin Receptors of the Vasopressor (V,) Type in the Nucleus of the Solitary Tract of the Rat Mediate Direct Neuronal Excitation Mario Raggenbass, Eliane Tribollet, Michel Dubois-Dauphin, and Jean Jacques Dreifuss Department of Physiology, University Medical Center, Geneva, Switzerland The existence of vasopressin-sensitive neurons in the nu- have been detected in the nucleus of the solitary tract and in cleus of the solitary tract of the rat and the presence in this the dorsal motor nucleus of the vagus nerve, respectively (Dorsa brain area of vasopressin binding sites were investigated et al., 1983; Dreifuss et al., 1988; Tribollet et al., 1988). Using using extracellular single-unit recordings from brain-stem brain-stem slices, we have shown that vagal motoneurons are slices and light microscopic autoradiography. About 45% of directly depolarized by oxytocin and that this effect is mediated the recorded neurons responded to vasopressin at 5-2000 by oxytocin receptors (Charpak et al., 1984; Raggenbass et al., nM by a reversible, concentration-dependent increase in fir- 1987a). ing rate. The action of vasopressin was direct, was sup- The nucleus of the solitary tract is involved in the regulation pressed by a vasopressor antagonist, and was mimicked by of cardiovascular functions. It contains second-order neurons a vasopressor agonist. Oxytocin was 1 O-l 00 times less ef- of the baroreceptor reflex arc, and bilateral lesions of this nucleus ficient than vasopressin and a specific antidiuretic agonist cause hypertension in rats (for reviews, see Bystrzycka and Nail, was without effect. -

Homeoprotein Phox2b Commands a Somatic-To-Visceral Switch in Cranial Sensory Pathways
Homeoprotein Phox2b commands a somatic-to-visceral switch in cranial sensory pathways Fabien D’Autréauxa,b,c, Eva Coppolaa,b,c, Marie-Rose Hirscha,b,c, Carmen Birchmeierd, and Jean-François Bruneta,b,c,1 aInstitut de Biologie de l’École Normale Supérieure 75005 Paris, France; bCentre National de la Recherche Scientifique, Unité Mixte de Recherche 8197, 75005 Paris, France; cInstitut National de la Santé et de la Recherche Médicale U1024, 75005 Paris, France; and dDepartment of Neuroscience, Max-Delbrück-Centrum for Molecular Medicine, D-13125 Berlin-Buch, Germany Edited by Yuh-Nung Jan, Howard Hughes Medical Institute, San Francisco, CA, and approved November 8, 2011 (received for review June 28, 2011) Taste and most sensory inputs required for the feedback regula- Dopamine-β-hydroxylase expression at embryonic day (E)9.5 and tion of digestive, respiratory, and cardiovascular organs are show attenuated expression of the tyrosine kinase receptor Ret at conveyed to the central nervous system by so-called “visceral” E10.5 (2). At E11.5, they are capable of projecting fibers to the sensory neurons located in three cranial ganglia (geniculate, pe- periphery (8). At E13.5, the ganglion cells are fewer than in the trosal, and nodose) and integrated in the hindbrain by relay sen- wild type (9), they have turned off the Phox2b locus as assessed sory neurons located in the nucleus of the solitary tract. Visceral by lacZ expression from the Phox2b locus in Phox2bLacZ/LacZ sensory ganglia and the nucleus of the solitary tract all depend for embryos, but still express peripherin (9). Therefore, a contingent their formation on the pan-visceral homeodomain transcription of epibranchial ganglion cells acquire a neuronal identity in the factor Phox2b, also required in efferent neurons to the viscera. -

A Circumscribed Projection from the Nucleus of the Solitary Tract to The
The Journal of Neuroscience, May 1989, g(5): 1668-l 682 A Circumscribed Projection from the Nucleus of the Solitary Tract to the Nucleus Ambiguus in the Rat: Anatomical Evidence for Somatostatin-284mmunoreactive Interneurons Subserving Reflex Control of Esophageal Motility E. T. Cunningham, Jr.‘s21a and P. E. Sawchenko2 ‘Department of Neurosciences, University of California, San Diego, La Jolla, California 92093, and 2The Salk Institute for Biological Studies and The Clayton Foundation for Research-California Division, La Jolla, California 92037 Axonal transport and immunohistochemical methods were The nucleus of the solitary tract (NTS) occupies a pivotal po- used to investigate the anatomical and biochemical orga- sition in the central visceromotor system. Projections of the nization of projections from the nucleus of the solitary tract Vth, VIIth, IXth, and Xth cranial nerves terminate centrally (NTS) to the rostral, esophageal, part of the nucleus ambig- within the NTS, conveying sensoryinformation from the oral, uus (NA) in the rat. Discrete iontophoretic deposits of a ret- thoracic, and abdominal cavities. The NTS, in turn, projects to rogradely transported tracer, fluorogold, placed in the rostra1 virtually all levels of the neuraxis, so as to coordinate appro- NA labeled a column of cells within the NTS, termed the priate behavioral, neuroendocrine, and autonomic responses central part of the NTS (after Ross et al., 1985) situated just (seeSawchenko, 1983; Norgren, 1984, for reviews). medial to the solitary tract and extending -

Cardiovascular Function Is Altered by Picomole Injections of Glutamate Into Rat Medulla
The Journal of Neuroscience, May 1988, E(5): 1884-1693 Cardiovascular Function Is Altered by Picomole Injections of Glutamate into Rat Medulla D. 0. Nelson, Howard L. Cohen, Jack L. Feldman,” and Donald Ft. McCrimmon Departments of Physiology, Anesthesia, and Surgery, Northwestern University Medical School, Chicago, Illinois 60611 Local neural circuitry in the nucleus tractus solitarius (NTS) ferent paths provides the neuroanatomical substrate for inte- involved in cardiovascular control was studied by injecting gration and modulation of cardiovascular responses to mecha- nanoliter volumes of excitatory amino acids into the struc- noreceptor afferent signals. Thus, the NTS must function as ture. Experiments were performed on urethane-anesthe- more than a simple relay nucleus. tized, artificially ventilated rats. Multibarrel micropipettes The neuronal integration within the NTS requires informa- were used for pressure ejection of drugs or a dye for marking tion transfer between neurons in a complex network. The de- ejection sites. Ejected volumes, ranging from 200 pl to 25 tailed nuclear substructure and microanatomy of the NTS sug- nl, were directly monitored for every injection. Injections of gests that small aggregates of neurons may act as functional as little as 200 fmol of L-glutamate in 200 pl into the medial units. Yet, despite a growing body of neuroanatomical literature and lateral NTS region rostra1 to the obex elicited marked, indicating a complex substructure within the NTS (Kalia and site-specific .decreases in arterial pressure and heart rate. Mesulum, 1980; Paxinos and Watson, 1986; Kalia et al., 1984; The majority of these responses were eliminated by block- Kalia and Fuxe, 1985) this structure is often considered to ade of parasympathetic and sympathetic neural outflow. -

Neuropeptides in the Human Brainstem
Investigación Clínica ISSN: 0535-5133 Universidad del Zulia Duque-Díaz, Ewing; Martínez-Rangel, Diana; Ruiz-Roa, Silvia Neuropeptides in the human brainstem Investigación Clínica, vol. 59, no. 2, 2018, April-June, pp. 161-178 Universidad del Zulia DOI: https://doi.org/10.22209/IC.v59n2a06 Available in: https://www.redalyc.org/articulo.oa?id=372960175007 How to cite Complete issue Scientific Information System Redalyc More information about this article Network of Scientific Journals from Latin America and the Caribbean, Spain and Journal's webpage in redalyc.org Portugal Project academic non-profit, developed under the open access initiative Invest Clin 59(2): 161 - 178, 2018 https//doi.org/10.22209/IC.v59n2a06 Neuropeptides in the human brainstem. Ewing Duque-Díaz1, Diana Martínez-Rangel1 and Silvia Ruiz-Roa1,2. 1Universidad de Santander UDES. Laboratory of Neuroscience, School of Medicine, Bucaramanga, Colombia. 2Universidad de Santander UDES. Laboratory of Neuroscience, Nursing Program, Bucaramanga, Colombia. Key words: neuropeptides; mesencephalon; pons; medulla oblongata; human. Abstract. The physiological importance of the brainstem has made it one of the most studied structures of the central nervous system of mammals (in- cluding human). This structure receives somatic and visceral inputs and its neurons send motor efferences by means of the cranial nerves, which innervate the head, neck and sensory organs, and it mediates in several actions such as movement, pain, cardiovascular, respiratory, salivary, sleep, vigil and sexual mechanisms. Most of these actions are mediated by neuroactive substances denominated neuropeptides, which are short amino acid chains widespread dis- tributed in the nervous system, that play a role in neurotransmission, neuro- modulation (paracrine and autocrine actions), and act as neurohormones.