Homeoprotein Phox2b Commands a Somatic-To-Visceral Switch in Cranial Sensory Pathways
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Regional Differences in Serotonin Content in the Nucleus of the Solitary Tract of Male Rats After Hypovolemia Produced by Polyethylene Glycol
J Physiol Sci (2013) 63:39–46 DOI 10.1007/s12576-012-0229-4 ORIGINAL PAPER Regional differences in serotonin content in the nucleus of the solitary tract of male rats after hypovolemia produced by polyethylene glycol J. Thomas Curtis • Michael B. Anderson • Kathleen S. Curtis Received: 30 June 2012 / Accepted: 6 August 2012 / Published online: 4 September 2012 Ó The Physiological Society of Japan and Springer 2012 Abstract Serotonin (5-HT) has been implicated in cen- and hormonal responses, occur over a comparatively long trally mediated compensatory responses to volume loss in period of time, whereas changes in autonomic nervous rats. Accordingly, we hypothesized that slowly developing, system activity occur in a much shorter time frame. Short- non-hypotensive hypovolemia increases serotonin in the term responses, in particular, involve low pressure baro- hindbrain nucleus of the solitary tract (NTS). We produced receptors in the heart and great veins which signal volume loss in adult male rats by administering hyperon- increased or decreased volume, and subsequently activate cotic polyethylene glycol (PEG) and then assessed 5-HT hindbrain baroreflex pathways, the connectivity and neu- levels in the NTS using measurements of tissue 5-HT rochemistry of which are well defined (for review, see [1]). content or 5-HT immunohistochemistry. The results show Activation in these pathways has been reported in response selective increases of 5-HT in the caudal NTS after PEG to increased volume [2–4] and, perhaps more familiarly, in treatment, but no change in the primary 5-HT metabolite, response to decreased volume [5–8], such as that produced 5-HIAA. -

The Responsiveness of Neurons in the Insular Gustatory Cortex of the Macaque Monkey Is Independent of Hunger
Physiology & Behavior, Vol. 42, pp. 223--229. Copyright ~ Pergamon Press plc, 1988. Printed in the U.S.A. 0031-9384/88 $3.00 + .00 The Responsiveness of Neurons in the Insular Gustatory Cortex of the Macaque Monkey is Independent of Hunger SIMON YAXLEY, EDMUND T. ROLLS 1 AND ZENON J. SIENKIEW1CZ University of Oxfi)rd, Department of Experimental Psychology South Parks Road, Oxford OXI 3UD, England Received 19 August 1986 YAXLEY, S., E. T. ROLLS AND Z. J. S1ENKIEWICZ. The responsiveness of neurons in the insular gustatory cortex of the macaque monkey is independent of hunger. PHYSIOL BEHAV 42(3) 223-229. 1988.--(1) In order to determine whether the responsiveness of neurons in the insular gustatory cortex is influenced by hunger, neuronal activity was analysed in it while macaque monkeys (Maeaca jhscicularis) were fed to satiety. The responses of single neurons in the insular gustatory cortex to the protypical taste stimuli glucose, NaCI, HCI and quinine HCI, and to fruit juice, were measured before, while, and after the monkey was fed to satiety with glucose or fruit juice. (2) While behavior turned from avid acceptance to active rejection upon repletion, the responsiveness of the neurons to the stimulus array, including the satiating solution, was unmodified. (3) It is concluded that in the insular gustatory cortex, neuronal responses to gustatory stimuli are not influenced by the normal transition from hunger to satiety. This is in contrast to the responses of a population of neurons recorded in the hypothalamus, which only respond to the taste of food when the monkey is hungry. -

Central Neurocircuits Regulating Food Intake in Response to Gut Inputs—Preclinical Evidence
nutrients Review Central Neurocircuits Regulating Food Intake in Response to Gut Inputs—Preclinical Evidence Kirsteen N. Browning * and Kaitlin E. Carson Department of Neural and Behavioral Sciences, Penn State College of Medicine, Hershey, PA 17033, USA; [email protected] * Correspondence: [email protected]; Tel.: +1-717-531-8267 Abstract: The regulation of energy balance requires the complex integration of homeostatic and hedonic pathways, but sensory inputs from the gastrointestinal (GI) tract are increasingly recognized as playing critical roles. The stomach and small intestine relay sensory information to the central nervous system (CNS) via the sensory afferent vagus nerve. This vast volume of complex sensory information is received by neurons of the nucleus of the tractus solitarius (NTS) and is integrated with responses to circulating factors as well as descending inputs from the brainstem, midbrain, and forebrain nuclei involved in autonomic regulation. The integrated signal is relayed to the adjacent dorsal motor nucleus of the vagus (DMV), which supplies the motor output response via the efferent vagus nerve to regulate and modulate gastric motility, tone, secretion, and emptying, as well as intestinal motility and transit; the precise coordination of these responses is essential for the control of meal size, meal termination, and nutrient absorption. The interconnectivity of the NTS implies that many other CNS areas are capable of modulating vagal efferent output, emphasized by the many CNS disorders associated with dysregulated GI functions including feeding. This review will summarize the role of major CNS centers to gut-related inputs in the regulation of gastric function Citation: Browning, K.N.; Carson, with specific reference to the regulation of food intake. -

Optical Vagus Nerve Modulation of Heart and Respiration Via Heart‑Injected Retrograde AAV Arjun K
www.nature.com/scientificreports OPEN Optical vagus nerve modulation of heart and respiration via heart‑injected retrograde AAV Arjun K. Fontaine1,3,8*, Gregory L. Futia1,8, Pradeep S. Rajendran4,5, Samuel F. Littich1,3, Naoko Mizoguchi2,7, Kalyanam Shivkumar4,5, Jefrey L. Ardell4,5, Diego Restrepo2,9, John H. Caldwell2,9, Emily A. Gibson1,9 & Richard F. f Weir1,3,6,9 Vagus nerve stimulation has shown many benefts for disease therapies but current approaches involve imprecise electrical stimulation that gives rise to of‑target efects, while the functionally relevant pathways remain poorly understood. One method to overcome these limitations is the use of optogenetic techniques, which facilitate targeted neural communication with light‑sensitive actuators (opsins) and can be targeted to organs of interest based on the location of viral delivery. Here, we tested whether retrograde adeno‑associated virus (rAAV2‑retro) injected in the heart can be used to selectively express opsins in vagus nerve fbers controlling cardiac function. Furthermore, we investigated whether perturbations in cardiac function could be achieved with photostimulation at the cervical vagus nerve. Viral injection in the heart resulted in robust, primarily aferent, opsin reporter expression in the vagus nerve, nodose ganglion, and brainstem. Photostimulation using both one‑ photon stimulation and two‑photon holography with a GRIN‑lens incorporated nerve cuf, was tested on the pilot‑cohort of injected mice. Changes in heart rate, surface electrocardiogram, and respiratory responses were observed in response to both one‑ and two‑photon photostimulation. The results demonstrate feasibility of retrograde labeling for organ targeted optical neuromodulation. Improved techniques are necessary for selective modulation of peripheral nerve fbers, particularly within the autonomic nervous system. -
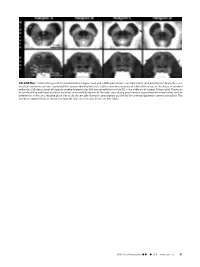
ON-LINE FIG 1. Selected Images of the Caudal Midbrain (Upper Row
ON-LINE FIG 1. Selected images of the caudal midbrain (upper row) and middle pons (lower row) from 4 of 13 total postmortem brains illustrate excellent anatomic contrast reproducibility across individual datasets. Subtle variations are present. Note differences in the shape of cerebral peduncles (24), decussation of superior cerebellar peduncles (25), and spinothalamic tract (12) in the midbrain of subject D (top right). These can be attributed to individual anatomic variation, some mild distortion of the brain stem during procurement at postmortem examination, and/or differences in the axial imaging plane not easily discernable during its prescription parallel to the anterior/posterior commissure plane. The numbers in parentheses in the on-line legends refer to structures in the On-line Table. AJNR Am J Neuroradiol ●:●●2019 www.ajnr.org E1 ON-LINE FIG 3. Demonstration of the dentatorubrothalamic tract within the superior cerebellar peduncle (asterisk) and rostral brain stem. A, Axial caudal midbrain image angled 10° anterosuperior to posteroinferior relative to the ACPC plane demonstrates the tract traveling the midbrain to reach the decussation (25). B, Coronal oblique image that is perpendicular to the long axis of the hippocam- pus (structure not shown) at the level of the ventral superior cerebel- lar decussation shows a component of the dentatorubrothalamic tract arising from the cerebellar dentate nucleus (63), ascending via the superior cerebellar peduncle to the decussation (25), and then enveloping the contralateral red nucleus (3). C, Parasagittal image shows the relatively long anteroposterior dimension of this tract, which becomes less compact and distinct as it ascends toward the thalamus. ON-LINE FIG 2. -

High-Yield Neuroanatomy, FOURTH EDITION
LWBK110-3895G-FM[i-xviii].qxd 8/14/08 5:57 AM Page i Aptara Inc. High-Yield TM Neuroanatomy FOURTH EDITION LWBK110-3895G-FM[i-xviii].qxd 8/14/08 5:57 AM Page ii Aptara Inc. LWBK110-3895G-FM[i-xviii].qxd 8/14/08 5:57 AM Page iii Aptara Inc. High-Yield TM Neuroanatomy FOURTH EDITION James D. Fix, PhD Professor Emeritus of Anatomy Marshall University School of Medicine Huntington, West Virginia With Contributions by Jennifer K. Brueckner, PhD Associate Professor Assistant Dean for Student Affairs Department of Anatomy and Neurobiology University of Kentucky College of Medicine Lexington, Kentucky LWBK110-3895G-FM[i-xviii].qxd 8/14/08 5:57 AM Page iv Aptara Inc. Acquisitions Editor: Crystal Taylor Managing Editor: Kelley Squazzo Marketing Manager: Emilie Moyer Designer: Terry Mallon Compositor: Aptara Fourth Edition Copyright © 2009, 2005, 2000, 1995 Lippincott Williams & Wilkins, a Wolters Kluwer business. 351 West Camden Street 530 Walnut Street Baltimore, MD 21201 Philadelphia, PA 19106 Printed in the United States of America. All rights reserved. This book is protected by copyright. No part of this book may be reproduced or transmitted in any form or by any means, including as photocopies or scanned-in or other electronic copies, or utilized by any information storage and retrieval system without written permission from the copyright owner, except for brief quotations embodied in critical articles and reviews. Materials appearing in this book prepared by individuals as part of their official duties as U.S. government employees are not covered by the above-mentioned copyright. To request permission, please contact Lippincott Williams & Wilkins at 530 Walnut Street, Philadelphia, PA 19106, via email at [email protected], or via website at http://www.lww.com (products and services). -

Tonsillar Branch of the Glossopharyngeal Nerve and the Superior Laryngeal Nerve in Lamb
THE JOURNAL OF COMPARATIVE NEUROLOGY 245:471-482 (1986) Central Connections of the Lingual- Tonsillar Branch of the Glossopharyngeal Nerve and the Superior Laryngeal Nerve in Lamb ROBERT D. SWEAZEY AND ROBERT M. BRADLEY Department of Oral Biology, School of Dentistry (R.D.S., R.M.B.), The Department of Physiology, School of Medicine (R.M.B.), University of Michigan, Ann Arbor, Michigan 48109 ABSTRACT Afferent and efferent central connections of the lingual-tonsillar branch of the glossopharyngeal nerve (LT-IX) and the superior laryngeal nerve (SLN) in the lamb were traced with horseradish peroxidase (HRP) histochem- istry. After entering the brainstem, most LT-IX and SLN afferent fibers turned caudally in the solitary tract (ST). Some afferent fibers of LT-IX terminated in the medial nucleus of the solitary tract slightly caudal to their level of entry. The remaining fibers projected to the dorsolateral, ventrolat- eral, and interstitial areas of the nucleus of the solitary tract (NST) at the level of the area postrema. Superior laryngeal nerve afferent fibers termi- nated extensively in the medial and ventral NST at levels near the rostral pole of the area postrema. Further caudal, near the level of obex, SLN afferent terminations were concentrated in the region ventrolateral to the ST and in the interstitial NST. The caudal extent of LT-IX and the rostral extent of SLN terminals projected to similar levels of the NST, but only a relatively small proportion of the total projections overlapped. Lingual- tonsillar and SLN fibers also coursed rostrally to terminate in the caudal pons within and medial to the dorsomedial principal sensory trigeminal nucleus. -

Cochleovestibular Nerve Development Is Integrated with Migratory Neural Crest Cells
Developmental Biology 385 (2014) 200–210 Contents lists available at ScienceDirect Developmental Biology journal homepage: www.elsevier.com/locate/developmentalbiology Cochleovestibular nerve development is integrated with migratory neural crest cells Lisa L. Sandell a,n, Naomi E. Butler Tjaden b,c, Amanda J. Barlow d, Paul A. Trainor b,c a University of Louisville, Department of Molecular, Cellular and Craniofacial Biology, Louisville, KY 40201, USA b Stowers Institute for Medical Research, Kansas City, MO 64110, USA c Department of Anatomy and Cell Biology, University of Kansas Medical Center, Kansas City, KS 66160, USA d Department of Surgery, University of Wisconsin, Madison, WI 53792, USA article info abstract Article history: The cochleovestibular (CV) nerve, which connects the inner ear to the brain, is the nerve that enables the Received 28 August 2013 senses of hearing and balance. The aim of this study was to document the morphological development of Received in revised form the mouse CV nerve with respect to the two embryonic cells types that produce it, specifically, the otic 1 November 2013 vesicle-derived progenitors that give rise to neurons, and the neural crest cell (NCC) progenitors that give Accepted 8 November 2013 rise to glia. Otic tissues of mouse embryos carrying NCC lineage reporter transgenes were whole mount Available online 16 November 2013 immunostained to identify neurons and NCC. Serial optical sections were collected by confocal Keywords: microscopy and were compiled to render the three dimensional (3D) structure of the developing CV Ear nerve. Spatial organization of the NCC and developing neurons suggest that neuronal and glial Otic populations of the CV nerve develop in tandem from early stages of nerve formation. -

Functional Anatomy of the Facial Nerve Revealed by Ramsay Hunt Syndrome
EDITORIAL DON GILDEN, MD Louise Baum Endowed Chair and Professor, Department of Neurology and Microbiology, University of Colorado School of Medicine, Aurora, CO Functional anatomy of the facial nerve revealed by Ramsay Hunt syndrome aricella-zoster virus (VZV) is a highly neuro- facial paralysis (geniculate zoster) not only around the V tropic and ubiquitous alpha-herpesvirus. Primary ear, but also on the hard palate or on the anterior two- infection causes varicella (chickenpox), after which thirds of the tongue.2 the virus becomes latent in ganglionic neurons along In geniculate ganglionitis, a rash is usually seen in the entire neuraxis. Reactivation decades later usually one but not all three of these skin and mucosal sites. results in zoster (shingles), pain with a dermatomal Yet in this issue of the Cleveland Clinic Journal of Medi- distribution, and rash. Unlike herpes simplex virus cine, Grillo et al3 describe a patient with facial palsy and type 1 (HSV-1), which becomes latent exclusively in rash in all three sites. This remarkable finding under- cranial nerve ganglia and reactivates to produce re- scores the importance of distinguishing Ramsay Hunt current vesicular lesions around the mouth, and un- syndrome from Bell palsy by checking for rash on the like HSV type 2, which becomes latent exclusively ear, tongue, and hard palate in any patient with acute in sacral ganglia and reactivates to produce genital unilateral peripheral facial weakness. Ramsay Hunt herpes, VZV may reactivate from any ganglia to cause syndrome results from active VZV replication in the zoster anywhere on the body. geniculate ganglion and requires treatment with an- tiviral drugs, whereas Bell palsy is usually treated with See related article, page 76 steroids. -

The Heart Side of Brain Neuromodulation
The heart side of brain neuromodulation rsta.royalsocietypublishing.org Simone Rossi1, Emiliano Santarnecchi1,2, Gaetano Valenza3,4,5 and Monica Ulivelli1 Review 1Department of Medicine, Surgery and Neuroscience, Unit of Neurology and Clinical Neurophysiology, Brain Investigation and Cite this article: Rossi S, Santarnecchi E, Neuromodulation Lab. (Si-BIN Lab.), Azienda Ospedaliera Valenza G, Ulivelli M. 2016 The heart side of Universitaria Senese, University of Siena, 53100 Siena, Italy Phil.Trans.R.Soc.A brain neuromodulation. 2Berenson–Allen Center for Noninvasive Brain Stimulation, 374:20150187. Department of Neurology, Beth Israel Deaconess Medical Center, http://dx.doi.org/10.1098/rsta.2015.0187 Boston, MA 02215, USA 3Department of Information Engineering, and Research Center Accepted: 11 January 2016 E. Piaggio, University of Pisa, 56122 Pisa, Italy 4Neuroscience Statistics Research Lab, Harvard Medical School, One contribution of 16 to a theme issue Massachusetts General Hospital, Boston, MA 02115, USA ‘Uncovering brain–heart information through 5Massachusetts Institute of Technology, Cambridge, MA 02139, USA advanced signal and image processing’. SR, 0000-0001-6697-9459 Subject Areas: biomedical engineering Neuromodulation refers to invasive, minimally invasive or non-invasive techniques to stimulate Keywords: discrete cortical or subcortical brain regions with deep brain stimulation, vagal nerve therapeutic purposes in otherwise intractable patients: for example, thousands of advanced Parkinsonian stimulation, repetitive transcranial -

(V,) Type in the Nucleus of the Solitary Tract of the Rat Mediate Direct Neuronal Excitation
The Journal of Neuroscience, November 1999, 9(11): 39293936 Vasopressin Receptors of the Vasopressor (V,) Type in the Nucleus of the Solitary Tract of the Rat Mediate Direct Neuronal Excitation Mario Raggenbass, Eliane Tribollet, Michel Dubois-Dauphin, and Jean Jacques Dreifuss Department of Physiology, University Medical Center, Geneva, Switzerland The existence of vasopressin-sensitive neurons in the nu- have been detected in the nucleus of the solitary tract and in cleus of the solitary tract of the rat and the presence in this the dorsal motor nucleus of the vagus nerve, respectively (Dorsa brain area of vasopressin binding sites were investigated et al., 1983; Dreifuss et al., 1988; Tribollet et al., 1988). Using using extracellular single-unit recordings from brain-stem brain-stem slices, we have shown that vagal motoneurons are slices and light microscopic autoradiography. About 45% of directly depolarized by oxytocin and that this effect is mediated the recorded neurons responded to vasopressin at 5-2000 by oxytocin receptors (Charpak et al., 1984; Raggenbass et al., nM by a reversible, concentration-dependent increase in fir- 1987a). ing rate. The action of vasopressin was direct, was sup- The nucleus of the solitary tract is involved in the regulation pressed by a vasopressor antagonist, and was mimicked by of cardiovascular functions. It contains second-order neurons a vasopressor agonist. Oxytocin was 1 O-l 00 times less ef- of the baroreceptor reflex arc, and bilateral lesions of this nucleus ficient than vasopressin and a specific antidiuretic agonist cause hypertension in rats (for reviews, see Bystrzycka and Nail, was without effect. -

Clinical Presentations and Outcome Studies of Cranial Nerve Involvement in Herpes Zoster Infection: a Retrospective Single-Center Analysis
Journal of Clinical Medicine Article Clinical Presentations and Outcome Studies of Cranial Nerve Involvement in Herpes Zoster Infection: A Retrospective Single-Center Analysis 1, 1, 1 2 3 Po-Wei Tsau y , Ming-Feng Liao y , Jung-Lung Hsu , Hui-Ching Hsu , Chi-Hao Peng , Yu-Ching Lin 4 , Hung-Chou Kuo 1 and Long-Sun Ro 1,* 1 Department of Neurology, Chang Gung Memorial Hospital, 199 Tung Hwa North Road, Taipei 105, Taiwan; [email protected] (P.-W.T.); [email protected] (M.-F.L.); [email protected] (J.-L.H.); [email protected] (H.-C.K.) 2 Department of Traditional Chinese Medicine, Division of Chinese Acupuncture and Traumatology, Chang Gung Memorial Hospital, Taipei 105, Taiwan; [email protected] 3 Division of Chinese Internal Medicine, Center for Traditional Chinese Medicine, Chang Gung Memorial Hospital, Taipei 105, Taiwan; [email protected] 4 Department of Medical Imaging and Intervention, Chang Gung Memorial Hospital, Taipei 105, Taiwan; [email protected] * Correspondence: [email protected]; Tel.: +886-3-3281200-8351 First authors: Both equally contributed to the concept and writing. y Received: 17 February 2020; Accepted: 24 March 2020; Published: 30 March 2020 Abstract: Varicella-zoster virus (VZV) infection can cause chickenpox and herpes zoster. It sometimes involves cranial nerves, and rarely, it can involve multiple cranial nerves. We aimed to study clinical presentations of cranial nerve involvement in herpes zoster infection. We included patients who had the diagnosis of herpes zoster infection and cranial nerve involvement. The diagnosis was confirmed by typical vesicles and a rash. We excluded patients who had cranial neuralgias or neuropathies but without typical skin lesions (zoster sine herpete or post-herpetic neuralgia).