DOCCM-255606 Invertebrates: Search and Extraction Methods V1.1 2
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Heavy Duty Truck Technician
Heavy Duty Truck Technician BOOK AND TOOL LIST 2019-2020 Books* ISBN: 9781284110586 TRUCK TECHNICIAN BUNDLE: $550.00 Fundamentals of Medium/Heavy Duty Commercial Vehicle Systems Textbook Fundamentals of Medium/Heavy Duty Diesel Engines Textbook Two Year Online Access Bundle must be purchased prior to the first day of class SUPPLIES: (app. $385.00) Drug Test - $30.00 Leather safety toe work boots, ANSI- Z87 approved safety glasses, clear, no tint - $60.00 to $200.00 Handheld electronic calculator Miscellaneous supplies (3 ring binder, notebook paper, pens, pencils, highlighters) 3 Uniform Shirts with CWI logo, 3 uniform pants - $225.00 (Long Sleeve $5.00 add per shirt) FIRST YEAR TOOL LIST: Course Fee $5600.00* Required* (see footnote) Toolbox: KRA4107FPBO - Rollaway tool chest. Minimum capacity 15,000 cubic inches. Maximum width 41 inches. Must have matching top box optional. Pliers: PL400B 7" diagonal cutting pliers 6-1/2" long nose needle nose pliers 8" combinations slip joint pliers Rib lock pliers (channel lock) 9” long BLP10 - 10" vice grips (locking pliers) SRPC105 - External/Internal snap ring pliers fixed tip .038 & .070 tip - 0 & 90 degree Hammers: BPN16B - 16 oz. Ball peen hammer HD4SG - 4 lb. Short handled hammer HBFE32 - 32 oz Dead blow soft face hammer Electrical: FLU177 - Fluke 177 Digital Volt Ohm Meter with temperature probe PWC9 - Wire stripper/terminal crimpers pliers Punches and chisels: PPC710BK 3-piece Cold chisel set 3-piece Pin punch set 3-piece Taper punch set Center punch (1/2" x 6") PPB826A- Brass drift (13/16th x 8 inch) Pry bars: SPBS704AO - Pry bar set (4-pcs striking) PBS704 - 4 piece lady foot / line up bar set Screwdrivers: SGDX80BR 1 ¼ " stubby screwdriver-flat blade 3½" screwdriver – flat blade 4" screwdriver- flat blade 6" screwdriver- flat blade 8" screwdriver- flat blade #1 Phillips screwdriver #2 Phillips screwdriver #2 Stubby Phillips screwdriver Measuring tools: TPMA12 - 12 ft. -

Builders Tools & Accessories
BUILDERS TOOLS & ACCESSORIES CONSTRUCTEURS OUTILS ET ACCESSOIRES Great Deals On Building supplies SHOP NOW & SAVE CONTENT Pages Tile Cutter & Accessories 286 Laminate Cutter & Accessories 290 Floor Roller Tools 294 Floor Scraper & Accessories 296 Glass & Tile Nipper 298 Glass Cutter Tool & Accessories 300 Tile Spacers & Tile Installation Tools 301 Tile Grout Removal Tools & Accessories 306 Grout Bag & Clean Up Accessories 307 Tile Grouting Sponge 308 Sawhorse & Brackets 310 Drywall Lifting Tools & Accessories 312 Drywall Sanding & Abrasive Accessories 315 Drywall Taping & Installation Accessories 318 Magnesium Bull Float & Accessories 320 Caulking Guns & Accessories 322 Pointed & Brick Trowels 326 Margin Trowels 328 Tuck Pointer Trowels 330 Concrete Groover 331 Drywall Trowel 332 Pool Trowels 333 Notched Trowels 336 Masonry Tools & Accessories 342 Grout & Cement Floats 344 Plastering Hawks & Taping Knives 348 Putty Knives - Flexible Blades 350 Mixing Paddles & Mud Pans 352 Tool Boxes and Storage 355 Bags, Pouches & Belts 358 Tarpaulins 365 Fencing & Barriers 370 Glass & Tile Nippers 372 Drywall Tools & Accessories 373 Plaster & Finishing Trowels 374 Tuck Pointer & Concrete Edgers 375 Floats & Plastering Hawk Accessories 380 Drywall Tools & Accessories 384 Grouting Tools & Mixing Paddles 385 Tile Cutter & Accessories Professional Tile Cutter 4-Ball Bearing Revolutionary Sliding High Mechanism Leverage Handle Tungsten-Alloy Japanese Cutting Wheel Over 10,000 linear feet of cut with a single wheel! Rubber Padded Bed Reinforced Heavy Duty -

Hand Tool Safety
Hand Tool Safety Slide Show Notes • Welcome to the training session on hand tools, which has been designed for anyone that uses hand tools in the course of their work. 1 Learning Objectives You will be able to: • Identify the hazards of hand tools • Take precautions against injury • Understand common safety practices for specific types of tools © Business & Legal Reports, Inc. 0512 Slide Show Notes By the end of the training session, you will be able to: • Identify the hazards of hand tools • Take precautions against injury • Understand common safety practices for specific types of tools 2 Types of Hand Tools • Hand tools include anything from axes to wrenches • Other common examples include screwdrivers, hammers, punches, handsaws, chisels, files, snips, axes, knives, box cutters, wrenches, pry bars, hooks, and pliers © Business & Legal Reports, Inc. 0512 Slide Show Notes • Hand tools are nonpowered tools that require human force in order to function and include anything from axes to wrenches. • Other common examples of hand tools include screwdrivers, hammers, punches, handsaws, chisels, files, snips, axes, knives, box cutters, wrenches, pry bars, hooks, and pliers Modify this slide to list the specific types of hand tools used at your facility. Bring examples of the hand tools used at your workplace. 3 Hand Tool Hazards Greatest hazards of hand tools result from misuse and improper maintenance • Using a screwdriver as a chisel and the tip breaks • Wooden handle is damaged and the head of the hammer flies off • Wrench, with jaws sprung, slips and your hand strikes another object • Impact tool, such as a chisel, with a mushroomed head shatters into fragments © Business & Legal Reports, Inc. -

Big Help Out!
FLOOR COVERING TOOLS BIG HELP OUT! NO. 280 AIR LIFTER NEW LONGER AIR PLATES HELP KEEP LOADS LEVEL! 2018 CATALOG Corners No. 575 Multi-Undercut Saw This versatile saw undercuts along walls, door jambs, under toe spaces, and will also undercut inside corners. The height of cut is easily adjustable from flush to one inch above the floor. The large, flat base prevents tipping and assures accurate, level cutting. The dual-angle depth gauge allows undercutting at a “straight-on” or a 45° an- gle. The visual scale provides precision depth control at 1 No. 575 Toe-Spaces /8" increments. Each saw comes with a 36-tooth No. 556 Carbide Blade. The blade is bell-shaped for added rigidity and should not be used on any other saw. Also comes with custom molded carrying case, a hex key for blade removal, and detailed instructions. Order No. Description Volts Amps. Weight 575 Multi-Undercut Saw 120 6.2 11 lbs. 556 Carbide Blade (36 tooth) --- --- 6 oz. No. 812 Super Saw FULLY UNDERCUTS INSIDE The Super Saw provides the most powerful motor of any 1 CORNER AREAS undercut saw available. The 13 AMP motor and 6 /2" di- HEIGHT COMES WITH CASE, CARBIDE BLADE, AND ameter flush-cutting blade can fully undercut the inside ADJUSTMENT UNDERCUTS DOORS AS MASONRY BLADE corner even in extremely tight areas. Doors as thick as 3 THICK AS 1¾" WITHOUT 1 /4" can be undercut without removal. Blade height is 1 REMOVAL adjustable from a flush cut to a maximum of 1 /4" off the floor. -

Lineman's Tools
Lineman’s Tools Klein Tools has established a reputation for manufacturing high quality tools to help linemen do their jobs. The company remains dedicated to making the best hand tools in the industry, and supporting linemen around the world with quality and reliability they can count on every day. Lineman’s Tools W E A R E Y E P R OTECTI ON M 39 Cable Cutters Utility Cable Cutter • Forged steel with black-oxide finish for long life. • Shear-type hook jaws grab and hold cable while shear-cutting action makes clean cuts. • Beveled tips for positive mating. • Hinge bolt with pin-set nut for proper blade tension. Non-slip vinyl grips. • 63035 • Not designed for cutting steel or ACSR. Cat. No. Overall Length MCM Cable-Cutting Maximum Capacity Handle Color Weight (lbs.) 63035 16-3/4" (425 mm) 350 copper, 350 aluminum red 1.70 Standard Cable Cutters • Lightweight, yet efficient shear-type cable cutters. • Replaceable hook-jaw blades grab and hold cable while shear-cutting action makes clean cuts. • Exceptionally strong fiberglass handles with heavy vinyl grips for comfort and slip resistance. • Heavy-duty pins in head serve as blade-stops. • Jaws are forged tool steel with black-oxide finish for long life. • Makes a clean, even cut for ease in fitting lugs and terminals. • Beveled tips assure positive mating, and hinge on a rugged 63041 bolt with a pin-set nut to maintain proper blade tension. • Communications Cable Cutter (Cat. No, 63047) cuts lead or rubber-covered communications cable. Cable Cutters • Not designed for cutting steel or ACSR. -

HAND TOOLS Hablamos Español
1-877-AZTool1 (1-877-298-6651) HAND TOOLS Hablamos Español Wrench Sets Drive Bits Master Hex Bit Set ST EE L SKU 094663 25952 4999 1/4” Drive: ST EE L 1/8”, 5/32”, 3/16” and 7/32” 3/8” Drive: 1/4”, 9/32”, 5/16” and 11/32” 35-Piece Master Torx Bit Set 44-Piece Master Torx and Hex Bit Set 1/2” Drive: 3/8”, 1/2”, 9/16”, 3/4” and 5/8” SKU 466622 SKU 311698 99 99 1/4” Drive: 25951 49 25950 46 2.5, 3, 4 and 5 mm • Star: T8, T10, T15, T20, T25, T27, T30, T40, T45, Hex:, 2 2.5, 3, 4, 5, 6, 7, 8, 10; 1/8”, 5/32”, 3/16”, 7/32”, 1/4”, 5/16” and 3/8” 3/8” Drive: ST EE L T47, T50, T55 and T60 Star: T10, T15, T20, T25, T27, T30, T40, T45, T47, T50, T55 and T60 6, 7, 8 and 9 mm 16-Piece • Tamper Proof Star: T7, T8, T9, T10, T15, T20, T25, Phillips: #1, #2 and #3 1/2” Drive: 10, 12, 14, 17 and 19 mm Combo Wrench Set - SAE T30, T40, T45, T50 and T55 Slotted: 3/16”, 1/4” and 5/16” • Female Star: E4, E5, E6, E7, E8, E10, E12, E14, SKU 531642 99 E16 and E18 Bits: T7H, T8H, T9H, T10H, T15H, T20H, T25H, T27H and T30H 17329 49 11-Piece Tamper Proof Torx Plus Bit Socket Set • Sizes Include: 3/8, 7/16, 1/2, 9/16, 5/8, 11/16, 3/4, 13/16, 7/8, 15/16, 1, 1-1/16, 1-1/8 and 1-1/4" SKU 255725 • Durable Nylon Storage Pouch 13-Piece 1/4” Drive 22973 Tamper-Resistant Star Security Bits • Raised Panel Design SKU 447157 99 99 24293 29 59 • Sizes: T7-T50 • Set Includes: (1/4” Drive) 8-TPT, 10-TPT, 15-TPT, 20-TPT and 25-TPT, (3/8” Drive) 27-TPT, 30-TPT, 40-TPT, 45-TPT, 50-TPT and 55-TPT Stubby Star Drive Sets Tamper Proof 13-Piece 1/4” Drive • 3/4" Overall Length Designed -
HVACR Tool List.Xlsx
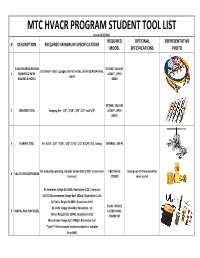
MTC HVACR PROGRAM STUDENT TOOL LIST revised 12/15/2017 REQUIRED OPTIONAL REPRESENTATIVE # DESCRIPTION REQUIRED MINIMUM SPECIFICATIONS MODEL SPECIFICATIONS PHOTO 3‐WAY REFRIGERATION RITCHIE: YELLOW 3‐1/8‐inch ‐ class 1 gauges, 60‐inch hoses, and R‐22/410A temp. 1 MANIFOLD WITH JACKET , UPC# scales GAUGES & HOSES 42004 RITCHIE: YELLOW 2SWAGING TOOL Swaging Set ‐ 1/4", 5/16", 3/8", 1/2" and 5/8" JACKET , UPC# 60476 3FLARING TOOL For 3/16”, 1/4”, 5/16”, 3/8”, 7/16”, 1/2” & 5/8” O.D. tubing IMPERIAL: 195‐FC for manually operating schrader valves (this is NOT a valve core C&D VALVE: having two of these would be 4VALVE CORE DEPRESSOR remover) CD5050 more useful AC Amperes: Range 0A‐300A, Resolution 0.2A, Clamp‐on AC/DC Microamperes: Range 0µA‐100µA, Resolution 0.1µA AC Volts: Range 0V‐600V, Resolution 1mV FLUKE: MODEL DC Volts: Range 0V‐600V, Resolution .1V 5DIGITAL MULTIMETER(S) 116/323 HVAC Ohms: Range 0.1Ω‐10MΩ, Resolution 0.2Ω COMBO KIT Microfarads: Range 0µF‐1000µF, Resolution 1µF "Type K" thermocouple probe receptacle or adapter True RMS MTC HVACR PROGRAM STUDENT TOOL LIST revised 12/15/2017 REQUIRED OPTIONAL REPRESENTATIVE # DESCRIPTION REQUIRED MINIMUM SPECIFICATIONS MODEL SPECIFICATIONS PHOTO PIPE CLAMP 6 TYPE K thermocouple, ribbon style element FLUKE: 80PK‐8 TEMPERTURE PROBE 7POCKET PSYCHROMETER must measure WB, DB, DP, %RH FIELDPIECE: PRH2 REFRIGERATION VALVE ratcheting type, for 3/16, 1/4, 5/16, & 3/8 ‐ inch square valve 8 STEM WRENCH stems with flare cut‐off groove, built‐in reamer and spare cutting wheel 9TUBING CUTTERS suitable -

Your Ultimate RV Toolbox
Your Ultimate RV Toolbox Most RV enthusiasts start with a basic assortment of tools such as screwdrivers, pliers, and sockets. The more you travel, and the more comfortable you get with doing some of your own maintenance and repairs, the more you “customize” your toolbox for your rig and level of experience. Just as there are hundreds of different types of RVs, there are a wide assortment of tools that can be used to do the job so there is not one “standard” list of tools that will be right for every RVer. However, here is a list that is a good reference to get you started on your journey to the “Ultimate Toolbox”. Basics • Screwdrivers – Philips/flat head (three different sizes but • Cordless Screw Gun/Charger definitely #2 phillips head) • Electric Screw Gun (Just In Case!) • Pliers – Large and small • Blow Hammer • Channel Lock Pliers • Regular Hammer • Needle Nose Pliers • Crescent Wrench – 2 sizes • Side Cutter • Razor Knife • Tin Snips • Vise Grips • Wire Stripper/cutter • Tire Gauge • Socket Sets – ¼” – 3/8” – Some ½” for larger projects like lug • Bubble Level nuts – SAE & Metric ( I also carry a set of deep well sockets – • Plumbers Tape 3/8”) • Gloves • Multi-bit set with Torx, #2 Philips, and others • LED Flashlight • Assorted Drill Bits • Shop Towels • Wrench Set – Open End/Box End – SAE & Metric • Hand Cleaner/Sanitizer Electrical • Duct Tape • Assorted Fuse • Black Electrical Tape • Extra 120-volt Outlet/GFCI • Rescue Tape • Battery Filler/Distilled Water • 12-volt Test Light • Zip Ties • Digital MultimeterDigital Multimeter -

Product Catalog February 1, 2019
MADE IN ® U.S.A. product CaTAlog february 1, 2019 www.eklindtool.com America’s Leading Manufacturer of High Quality Hex Keys since 1950 INDEX TORX® HEX Ball / Tamper Standard Ball Tamper Standard Page Contents ® Torx® Resistant Torx Hex Resistant Hex INDEX 3 Combination Sets ì   4 Ergo-Fold™ Keys Sets ì ì   5 Classic Fold-Up Sets ì   6 Ball-Hex-L™ Key Sets in Molded Holders ì  7 Hex-L® Key Sets in Molded Holders ì ì  8 Hex-L® Keys in Metal Boxes ì   9 Hex-L® Keys in Vinyl Pouches ì ì ì   10 Cushion & Standard Grip T-Key Sets  11 Power-T™ Key Sets ì   12 PSD™Precision Screwdriver Sets ì   12 Ball-Hex Drivers™  13-14 Individual Ball-Hex-L™ Keys ì  15-17 Individual Hex-L® Keys   18 Individual Torx® L-Keys ì ì 19 Individual Power-T™ Keys ì   20-21 Individual Cushion & Standard GripT-Keys  22 Individual PSD™ Precision Screwdrivers ì   23 Individual Ball-Hex Drivers™  WWW.EKLINDTOOL.COM TORX® tools drive all TORX®, TORX PLUS® and “star” fasteners. 2 TORX® and TORX PLUS® are registered trademarks of Acument Intellectual Properties, LLC. COMBINATION SETS Eklind Combo Sets are a great choice when both inch and metric Hex Keys are needed. These sets offer convenience as well as value. Combo Sets include popular inch and SETS COMBINATION metric sizes in Ball-Hex,Standard Hex and Torx® style tips. -

Utility Catalog Edition 2
For Professionals... Since 1857® Utility Catalog Edition 2 www.kleintools.com Product Index Company History ... Page 1 W E A R E Y E P R OTECTI ON M Cable & Lineman Buckets Bolt Cutters .........Pages 2-6 & Accessories ........Pages 20-24 Klein Pliers ........ Pages 7-8 Tool Bags ............Pages 25-27 Tool Pouches ...........Pages 9-12 Wrenches & Holders ...........Pages 28-29 Lineman’s Screwdrivers ........Page 13 Climbing Equipment ......... Pages 30-37 33_Linemans_ System Pole & Tree Hammers ............Page 14 Climbers .......... Pages 38-40 Knives & ..... Pages 41-44 Cutting Tools ........Pages 15-18 Block & Tackle CL200 Measuring Tools .....Page 19 Test & Measurement. .Pages 45-48 AUTO A AC Personal Protection Warranty ......... Page 49 Equipment ..........Page 19 Please visit www.kleintools.com for downloadable color product images and PDF formatted Klein Tools Catalogs. Company History Klein Tools was founded in 1857 by an industrious German immigrant, Mathias Klein, who began in the hand tool business when a broken side-cutting pliers was brought to his forge shop by a telegraph lineman. Mathias repaired the pliers by forging and finishing a new half for the tool and riveting it to the old half. Soon the lineman returned because the other original half of the pliers had broken and needed replacement. Mathias forged and finished the second half of the pliers and riveted it to the other replacement half – creating the first complete Klein pliers. To this day, Klein Tools is proudly owned and managed by the Klein family. Over 150 years of hard work and dedication has earned Klein the reputation of supplying only the finest quality products for users of professional hand tools History Company and occupational protective equipment. -

What's in Your Toolbox? You'll Find Lots of Online Recommendations About the 20 Tools You Should Have on Hand in Your
What’s In Your Toolbox? You’ll find lots of online recommendations about the 20 tools you should have on hand in your RV. Or, you can let us do the research and download our list. No need to feel you have to have every item on the list before you leave home. It’s something you may want to build up to. Feel free to make your own additions and corrections, and to share this or your list with others. Without further ado, here’s our version of the “RV Toolbox Top 20.” 1. Slip-Joint Pliers - Handy for tightening almost anything you can’t get a wrench on. 2. Multi-Tip Screwdriver - w/2 or more blade tips, Phillips head tips, torx or other tips 3. Claw Hammer - For traditional ‘pounding, leverage 4. Socket Wrench Set - Some sets include both English and Metric sizes - You might need both 5. Utility Knife - Standard, w/sliding blade, extra blades 6. Tape - Duct Tape, Electrical Tape, Velcro-style hook-and-loop patches 7. Adhesives - Gorilla Glue or similar, Super Glue 8. Tire Pressure Gauge 9. Adjustable Voltmeter 10. Vise-Grips 11. Abrasives - Sand paper, metal file 12. Battery-Powered Drill w/charger, basic drill bits 13. Loose Parts: Assorted Screws, Bolts, Nuts, & Washers, Wire Nuts, Zip Ties, Spare Bulbs, Automotive Fuses etc. 14. Wood Putty 15. Bottle Jack w/Tire Wrench - to change tires, brace the frame, etc. 16. Tape Measure 17. Hacksaw w/spare blades 18. LED Flashlight w/spare batteries 19. C-Clamps 20. Lubricants - WD 40, 3-in-1 oil, lock de-icer/lubricant When possible and practical, it’s good to make use of the molded plastic cases that come with many tools - Keeps them organized, dry, and quiet. -

Safety Alert from the International Association of Drilling Contractors
Safety Alert From the International Association of Drilling Contractors ALERT 14 – 20 HAND TOOL LEFT ON CCU RESULTS IN POTENTIAL DROPPED OBJECT WHAT HAPPENED: When receiving baskets and containers from the vessel, the flagman on the platform observed a sledgehammer lying loose on top of a cargo carrying unit (CCU). The weight of the sledgehammer is 2.24 kg (~5 lbs). The possible drop height from the basket to the sea/vessel was estimated to be 60 meters (~197 feet), Impact=1318 J. The landing area on the platform was secured from entry during the cargo operation. The Master of the vessel received confirmation from the crew of the vessel that they were missing one sledgehammer. The last time the sledgehammer had been used was during preparations for loading water based mud at the quay (dock) when the vessel was in port and the bulk hose was connected-up by the vessel crew. During that operation, an object was discovered in the hose by the crew, who informed the bridge. The bridge then requested base personnel come onboard to clear the hose. The operator from the base had borrowed a sledge hammer from one of the vessel’s crew and removed the object from the hose. After the cargo hose had been connected, the sledgehammer was inadvertently placed onto the CCU. The deck crew then connected the hose and opened the loading valve on orders from the bridge. One crew member was appointed as hose guard ensuring that there were no leaks or deviations in the operation. The sledgehammer had not been noticed by the vessel crew before the CCU was sent up to the platform offshore.