Reduction of Side Effects from Aromatase Inhibitors
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

CHAPTER 29 ORGANIC CHEMICALS VI 29-1 Notes 1
)&f1y3X CHAPTER 29 ORGANIC CHEMICALS VI 29-1 Notes 1. Except where the context otherwise requires, the headings of this chapter apply only to: (a) Separate chemically defined organic compounds, whether or not containing impurities; (b) Mixtures of two or more isomers of the same organic compound (whether or not containing impurities), except mixtures of acyclic hydrocarbon isomers (other than stereoisomers), whether or not saturated (chapter 27); (c) The products of headings 2936 to 2939 or the sugar ethers and sugar esters, and their salts, of heading 2940, or the products of heading 2941, whether or not chemically defined; (d) Products mentioned in (a), (b) or (c) above dissolved in water; (e) Products mentioned in (a), (b) or (c) above dissolved in other solvents provided that the solution constitutes a normal and necessary method of putting up these products adopted solely for reasons of safety or for transport and that the solvent does not render the product particularly suitable for specific use rather than for general use; (f) The products mentioned in (a), (b), (c), (d) or (e) above with an added stabilizer (including an anticaking agent) necessary for their preservation or transport; (g) The products mentioned in (a), (b), (c), (d), (e) or (f) above with an added antidusting agent or a coloring or odoriferous substance added to facilitate their identification or for safety reasons, provided that the additions do not render the product particularly suitable for specific use rather than for general use; (h) The following products, diluted to standard strengths, for the production of azo dyes: diazonium salts, couplers used for these salts and diazotizable amines and their salts. -

(12) United States Patent (10) Patent No.: US 8,552,057 B2 Brinton Et Al
US008552057B2 (12) United States Patent (10) Patent No.: US 8,552,057 B2 Brinton et al. (45) Date of Patent: *Oct. 8, 2013 (54) PHYTOESTROGENIC FORMULATIONS FOR OTHER PUBLICATIONS ALLEVATION OR PREVENTION OF Morito et al. Interaction of Phytoestrogens with Estrogen Receptors NEURODEGENERATIVE DISEASES a and b (II). Biol. Pharm. Bull. 25(1), pp. 48-52 (2002).* Kinjo et al. Interactions of Phytoestrogens with Estrogen Receptors a (75) Inventors: Roberta Diaz, Brinton, Rancho Palos and b (III). Biol. Pharm. Bull. 27(2) pp. 185-188 (2004).* Verdes, CA (US); Liqin Zhao, Los An, et al., "Estrogen receptor -selective transcriptional activity and Angeles, CA (US) recruitment of coregulators by phytoestrogens', J Biol. Chem. 276(21): 17808-14 (2001). (73) Assignee: University of Southern California, Los Avis, et al. Is there a menopausal syndrome'? Menopausal status and symptoms across racial/ethnic groups', Soc. Sci. Med., 52(3):345-56 Angeles, CA (US) (2001). Brinton, et al. Impact of estrogen therapy on Alzheimer's disease: a (*) Notice: Subject to any disclaimer, the term of this fork in the road?’ CNS Drugs, 18(7):405-422 (2004). patent is extended or adjusted under 35 Bromberger, et al., Psychologic distress and natural menopause: a U.S.C. 154(b) by 0 days. multiethnic community study’. Am. J. Public Health, 91 (9) 1435-42 (2001). This patent is Subject to a terminal dis Brookmeyer, et al., “Projections of Alzheimer's disease in the United claimer. States and the public health impact of delaying disease onset'. Am. J. Public Health, 88(9): 1337-42 (1998). (21) Appl. -

Ricini Oleum
PHARMACOGNOSY II PHAR306 6th Semester 5th Lecture Prof. Dr. Müberra Koşar Ass. Prof. Dr. Aybike Yektaoğlu Eastern Mediterranean University Faculty of Pharmacy Department of Pharmacognosy PHARMACEUTICAL FIXED OILS AND ANIMAL FATS FIXED OILS & ANIMAL FATS Amygdalae oleum • “Almond oil” • obtained by crushing of the seeds of two varieties Prunus dulcis var. dulcis or P. dulcis var. amara (Rosaceae) in the cold • Almond oil is obtained in the Mediterranean countries (Italy, France, Spain and North Africa) where its culture is obtained • The only difference between the two varieties is the cyanogenic glycoside content of the var. amara FIXED OILS&ANIMAL FATS Amygdalae oleum • seeds carries 40-55% fixed oil • the refined oil mainly contains oleic acid (62-86%), linoleic (20- 30%), palmitic (4-9%) • Amydalae oleum raffinatum (Almond oil, refined) (Eur.Pu.) • Amydalae oleum virginale (Almond oil, virgin) (Eur.Ph.) • major used in cosmetology and dermatology • used as a carrier in oily injectable preparations FIXED OILS&ANIMAL FATS Arachidis oleum • “Arachis oil, Peanut oil” – “Peanut butter” • Arachis hypogaea (Fabaceae) • cultivated in South America, China, India, Australia, and West Africa • due to various genotypes they vary in fatty acid content • the seeds are cold-pressed • they have similar properties as olive oil • most suitable oil for added for embedding purposes into other oils (e.g. olive oil) FIXED OILS&ANIMAL FATS Arachidis oleum - content • seeds carries 40-50% fixed oil • 50-65% oleic acid • 18-30% linoleic acid • 8-10% palmitic -

The Natural Compounds Atraric Acid and N-Butylbenzene-Sulfonamide
The natural compounds atraric acid and N-butylbenzene-sulfonamide as antagonists of the human androgen receptor and inhibitors of prostate cancer cell growth Daniela Roell, Aria Baniahmad To cite this version: Daniela Roell, Aria Baniahmad. The natural compounds atraric acid and N-butylbenzene-sulfonamide as antagonists of the human androgen receptor and inhibitors of prostate cancer cell growth. Molec- ular and Cellular Endocrinology, Elsevier, 2010, 332 (1-2), pp.1. 10.1016/j.mce.2010.09.013. hal- 00654968 HAL Id: hal-00654968 https://hal.archives-ouvertes.fr/hal-00654968 Submitted on 24 Dec 2011 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Accepted Manuscript Title: The natural compounds atraric acid and N-butylbenzene-sulfonamide as antagonists of the human androgen receptor and inhibitors of prostate cancer cell growth Authors: Daniela Roell, Aria Baniahmad PII: S0303-7207(10)00474-0 DOI: doi:10.1016/j.mce.2010.09.013 Reference: MCE 7644 To appear in: Molecular and Cellular Endocrinology Received date: 29-6-2010 Revised date: 3-9-2010 Accepted date: 27-9-2010 Please cite this article as: Roell, D., Baniahmad, A., The natural compounds atraric acid and N-butylbenzene-sulfonamide as antagonists of the human androgen receptor and inhibitors of prostate cancer cell growth, Molecular and Cellular Endocrinology (2010), doi:10.1016/j.mce.2010.09.013 This is a PDF file of an unedited manuscript that has been accepted for publication. -

(12) United States Patent (10) Patent No.: US 9,616,072 B2 Birrell (45) Date of Patent: *Apr
USOO961. 6072B2 (12) United States Patent (10) Patent No.: US 9,616,072 B2 Birrell (45) Date of Patent: *Apr. 11, 2017 (54) REDUCTION OF SIDE EFFECTS FROM 6,200,593 B1 3, 2001 Place AROMATASE INHIBITORS USED FOR 6,241,529 B1 6, 2001 Place 6,569,896 B2 5/2003 Dalton et al. TREATING BREAST CANCER 6,593,313 B2 7/2003 Place et al. 6,696,432 B1 2/2004 Elliesen et al. (71) Applicant: Chavah Pty Ltd, Medindie, South 6,995,284 B2 2/2006 Dalton et al. Australia (AU) 7,772.433 B2 8, 2010 Dalton et al. 8,003,689 B2 8, 2011 Veverka 8,008,348 B2 8, 2011 Steiner et al. (72) Inventor: Stephen Nigel Birrell, Medindie (AU) 8,980,569 B2 3/2015 Weinberg et al. 8,980,840 B2 3/2015 Truitt, III et al. (73) Assignee: CHAVAH PTY LTD., Stirling, South 9,150,501 B2 10/2015 Dalton et al. Australia (AU) 2003/0O87885 A1 5/2003 Masini-Eteve et al. 2004/O191311 A1 9/2004 Liang et al. *) Notice: Subject to anyy disclaimer, the term of this 2005/OO32750 A1 2/2005 Steiner et al. 2005/0176692 A1 8/2005 Amory et al. patent is extended or adjusted under 35 2005/0233970 A1 10/2005 Garnick U.S.C. 154(b) by 0 days. 2006, OO69067 A1 3/2006 Bhatnagar et al. 2007/0066568 A1 3/2007 Dalton et al. This patent is Subject to a terminal dis 2009,0264534 A1 10/2009 Dalton et al. claimer. 2010, O144687 A1 6, 2010 Glaser 2014, OO18433 A1 1/2014 Dalton et al. -

Annex 2B Tariff Schedule of the United States See General Notes to Annex 2B for Staging Explanation HTSUS No
Annex 2B Tariff Schedule of the United States See General Notes to Annex 2B for Staging Explanation HTSUS No. Description Base Rate Staging 0101 Live horses, asses, mules and hinnies: 0101.10.00 -Purebred breeding animals Free E 0101.90 -Other: 0101.90.10 --Horses Free E 0101.90.20 --Asses 6.8% B --Mules and hinnies: 0101.90.30 ---Imported for immediate slaughter Free E 0101.90.40 ---Other 4.5% A 0102 Live bovine animals: 0102.10.00 -Purebred breeding animals Free E 0102.90 -Other: 0102.90.20 --Cows imported specially for dairy purposes Free E 0102.90.40 --Other 1 cent/kg A 0103 Live swine: 0103.10.00 -Purebred breeding animals Free E -Other: 0103.91.00 --Weighing less than 50 kg each Free E 0103.92.00 --Weighing 50 kg or more each Free E 0104 Live sheep and goats: 0104.10.00 -Sheep Free E 0104.20.00 -Goats 68 cents/head A 0105 Live poultry of the following kinds: Chickens, ducks, geese, turkeys and guineas: -Weighing not more than 185 g: 0105.11.00 --Chickens 0.9 cents each A 0105.12.00 --Turkeys 0.9 cents each A 0105.19.00 --Other 0.9 cents each A -Other: 0105.92.00 --Chickens, weighing not more than 2,000 g 2 cents/kg A 0105.93.00 --Chickens, weighing more than 2,000 g 2 cents/kg A 0105.99.00 --Other 2 cents/kg A 0106 Other live animals: -Mammals: 0106.11.00 --Primates Free E 0106.12.00 --Whales, dolphins and porpoises (mammals of the order Cetacea); manatees and dugongs (mammals of the order Sirenia) Free E 0106.19 --Other: 2B-Schedule-1 HTSUS No. -
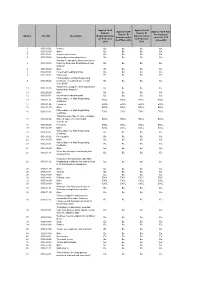
Number HS 2007 Description Applied Tariff Rate for Guatemala Under The
Applied Tariff Applied Tariff Applied Tariff Applied Tariff Rate Rate for Rate for El Rate for El for Honduras Number HS 2007 Description Guatemala under Salvador under Salvador under under the FTA the FTA since the FTA since the FTA in 2010 since 2010 2010 2011 1 0101.10.01 Horses Ex. Ex. Ex. Ex. 2 0101.10.99 Other Ex. Ex. Ex. Ex. 3 0101.90.01 Jump or race horses Ex. Ex. Ex. Ex. 4 0101.90.02 Non pedigreed breeding horses Ex. Ex. Ex. Ex. "Horses for slaughter, when imported by 5 0101.90.03 Federally Inspected Establishment type Ex. Ex. Ex. Ex. packers." 6 0101.90.99 Other Ex. Ex. Ex. Ex. 7 0102.10.01 Pure-bred breeding animals Ex. Ex. Ex. Ex. 8 0102.90.01 Dairy cows Ex. Ex. Ex. Ex. "With pedigree or High Registrating 9 0102.90.02 Certificate, excluding those of code Ex. Ex. Ex. Ex. 0102.90.01" "Bovines for slaughter, when imported by 10 0102.90.03 Ex. Ex. Ex. Ex. Industrial de Abastos." 11 0102.90.99 Other Ex. Ex. Ex. Ex. 12 0103.10.01 Pure-bred breeding animals Ex. Ex. Ex. Ex. With pedigree or High Registrating 13 0103.91.01 EXCL. EXCL. EXCL. EXCL. Certificate. 14 0103.91.02 Peccaries EXCL. EXCL. EXCL. EXCL. 15 0103.91.99 Other EXCL. EXCL. EXCL. EXCL. With pedigree or High Registrating 16 0103.92.01 EXCL. EXCL. EXCL. EXCL. Certificate. "Weighing more than 110 kg., excluding 17 0103.92.02 those of codes 0103.92.01 and EXCL. -

Aromatase and Its Inhibitors: Significance for Breast Cancer Therapy † EVAN R
Aromatase and Its Inhibitors: Significance for Breast Cancer Therapy † EVAN R. SIMPSON* AND MITCH DOWSETT *Prince Henry’s Institute of Medical Research, Monash Medical Centre, Clayton, Victoria 3168, Australia; †Department of Biochemistry, Royal Marsden Hospital, London SW3 6JJ, United Kingdom ABSTRACT Endocrine adjuvant therapy for breast cancer in recent years has focussed primarily on the use of tamoxifen to inhibit the action of estrogen in the breast. The use of aromatase inhibitors has found much less favor due to poor efficacy and unsustainable side effects. Now, however, the situation is changing rapidly with the introduction of the so-called phase III inhibitors, which display high affinity and specificity towards aromatase. These compounds have been tested in a number of clinical settings and, almost without exception, are proving to be more effective than tamoxifen. They are being approved as first-line therapy for elderly women with advanced disease. In the future, they may well be used not only to treat young, postmenopausal women with early-onset disease but also in the chemoprevention setting. However, since these compounds inhibit the catalytic activity of aromatase, in principle, they will inhibit estrogen biosynthesis in every tissue location of aromatase, leading to fears of bone loss and possibly loss of cognitive function in these younger women. The concept of tissue-specific inhibition of aromatase expression is made possible by the fact that, in postmenopausal women when the ovaries cease to produce estrogen, estrogen functions primarily as a local paracrine and intracrine factor. Furthermore, due to the unique organization of tissue-specific promoters, regulation in each tissue site of expression is controlled by a unique set of regulatory factors. -

• Lipids: a Heterogeneous Class of Naturally Occurring Organic
Organic Lecture Series 1 Organic Lecture Series •• Lipids:Lipids: a heterogeneous class of naturally occurring organic compounds classified together on the basis of common solubility properties –they are insoluble in water but soluble in aprotic organic solvents, including diethyl ether, methylene chloride, and acetone 2 Organic Lecture Series • Lipids include –triglycerides, phospholipids, prostaglandins, prostacyclins, and fat-soluble vitamins –cholesterol, steroid hormones, and bile acids 3 Organic Lecture Series TriglyceridesTriglycerides •• Triglyceride:Triglyceride: an ester of glycerol with three fatty acids O CH OCR O 2 CH 2 OH RCOOH 1. NaOH, H2 O R' COCH O HOCH + R'COOH 2 . HCl, H O 2 CH OH R' ' COOH CH 2 OCR'' 2 A triglyceride 1,2,3-Propanetriol Fatty acids (Glycerol, glycerin) 4 FattyFatty acidacid Organic Lecture Series • Fatty acid: a carboxylic acid derived from hydrolysis of animal fats, vegetable oils, or membrane phospholipids – nearly all have an even number of carbon atoms, most between 12 and 20, in an unbranched chain – the three most abundant are palmitic (16:0), stearic acid (18:0), and oleic acid (18:1) 5 FattyFatty acidacid Organic Lecture Series –the three most abundant are palmitic (16:0), stearic acid (18:0), and oleic acid (18:1) –In this labeling system: (carbon #:alkene #) palmitate (16:0) oleate (18:1) O O CH 2 OC(CH2 ) 14CH3 stearate (18:0) CH3 (CH2 )7 CH= CH( CH2 )7 COCH O CH 2 OC(CH2 ) 16CH3 6 FattyFatty acidacid Organic Lecture Series –in most unsaturated fatty acids, the cis isomer predominates; the -
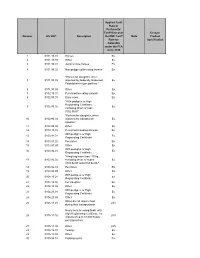
Number HS 2007 Description Applied Tariff Rate Or Preferential Tariff Rate
Applied Tariff Rate or Preferential Tariff Rate over Ex-out / Number HS 2007 Description the NMF Tariff Note Product Rate for Specification Colombia under the FTA since 2010 1 0101.10.01 Horses Ex. 2 0101.10.99 Other Ex. 3 0101.90.01 Jump or race horses Ex. 4 0101.90.02 Non pedigreed breeding horses Ex. "Horses for slaughter, when 5 0101.90.03 imported by Federally Inspected Ex. Establishment type packers." 6 0101.90.99 Other Ex. 7 0102.10.01 Pure-bred breeding animals Ex. 8 0102.90.01 Dairy cows Ex. "With pedigree or High Registrating Certificate, 9 0102.90.02 Ex. excluding those of code 0102.90.01" "Bovines for slaughter, when 10 0102.90.03 imported by Industrial de Ex. Abastos." 11 0102.90.99 Other Ex. 12 0103.10.01 Pure-bred breeding animals Ex. With pedigree or High 13 0103.91.01 Ex. Registrating Certificate. 14 0103.91.02 Peccaries Ex. 15 0103.91.99 Other Ex. With pedigree or High 16 0103.92.01 Ex. Registrating Certificate. "Weighing more than 110 kg., 17 0103.92.02 excluding those of codes Ex. 0103.92.01 and 0103.92.03." 18 0103.92.03 Peccaries Ex. 19 0103.92.99 Other Ex. With pedigree or High 20 0104.10.01 Ex. Registrating Certificate. 21 0104.10.02 For slaughter. Ex. 22 0104.10.99 Other Ex. With pedigree or High 23 0104.20.01 Ex. Registrating Certificate. 24 0104.20.99 Other Ex. When do not require food 25 0105.11.01 28% during their transportation Newly born breeding birds, with High Registrating Certificate, for 26 0105.11.02 28% imports of up to 18,000 heads per transaction. -

Phytochemical Investigation of Turraea Robusta, Turraea Nilotica and Ekebergia Capensis for Antiplasmodial and Cytotoxic Compounds
UNIVERSITY OF NAIROBI PHYTOCHEMICAL INVESTIGATION OF TURRAEA ROBUSTA, TURRAEA NILOTICA AND EKEBERGIA CAPENSIS FOR ANTIPLASMODIAL AND CYTOTOXIC COMPOUNDS. BY BEATRICE NJERI IRUNGU I80/80299/2010 A Thesis Submitted in Fulfilment of the Requirements for Award of the Degree of Doctor of Philosophy in Chemistry of the University of Nairobi 2014 i DECLARATION I declare tha t this thesis is my original work and has not been submitted elsewhere for examination or award of a degree. Signature.................................................... Date......................................... Beatrice Njeri Irungu I80/80299/2010 Department of Chemistry School of Physical Sciences University of Nairobi This thesis is submitted for examination with our approval as research supervisors Signature Date Professor Jacob Midiwo ................... ................. Department of Chemistry University of Nairobi P.O Box 30197-00100, Nairobi-Kenya [email protected] Professor Abiy Yenesew ……………… …………… Department of Chemistry University of Nairobi P.O Box 30197-00100, Nairobi-Kenya [email protected] Dr. Jennifer Orwa ........................ ........................ Kenya Medical Research Institute Centre for Traditional Medicine and Drug Research P.O. Box 54840-00200, Nairobi- Kenya [email protected] ii DEDICATION This thesis is dedicated to my husband Kimani Maina for enduring this long process with me, always offering support and love. our children Maina Kimani and Nyawira Kimani Though too young to understand what mum was going through, you supported me in your own little ways. I pray that this will serve as an encouragement as you walk through your academic pathways. iii ACKNOWLEDGEMENT The road towards a doctorate degree is not walked alone. Thus, this project would not have been possible without the support of my supervisors/mentors, friends and colleagues, to only some of whom it is possible to give particular mention here. -

Aromtase Inhibitors in Breast Cancer
한국유방암학회지:제5권 제4호 Aromtase Inhibitors in Breast Cancer Department of Surgery, Gachon Medical School, Incheon, Korea Woo-Shin Shim, M.D. INTRODUCTION gressions and to be associated with minimal side effects and toxicity. The second strategy, blockade of estradiol biosyn- Breast cancer is now the second most cancer in women after thesis, was demonstrated to be feasible using the steroido- stomach cancer in Korea, and is increasing continuously. In the genesis inhibitor, aminoglutethimide, which produced tumor year 2000, the crude incidence of breast cancer in Korea was regressions equivalent to those observed with tamoxifen.(3) estimated about 23 per 100,000 people.(1) However, side effects from aminoglutethimide were consid- For the process of inducing breast cancer, estrogens appear erable and its effects on several steroidogenic enzymes required to play a predominant role. These sex steroids are believed to concomitant use of a glucocorticoid. Consequently, tamoxifen initiate and to promote the process of the breast carcinogenesis became the preferred, first line endocrine agent with which to by enhancing the rate of cell division and reducing time treat ER-positive advanced breast cancer. However, the clinical available for DNA repair. A new concept is that estrogens can efficacy of aminoglutethimide focused attention upon the need be metabolized to catechol-estrogens and then to quinines that to develop more potent, better tolerated, and more specific directly damage DNA. These two process-- estrogen receptor inhibitors of estrogen biosynthesis. mediated, genomic effects on proliferation and receptor independent, genotoxic effects of estrogen metabolites-- can act INHIBITION OF ESTRADIOL BIOSYNTHESIS in an additive or synergistic fashion to cause breast cancer.(2) Breast cancers that arise in patients can be divided into Multiple enzymatic steps are involved in the biosynthesis of hormone dependent and hormone independent subtypes.(3) The estradiol and could potentially be used as targets for inhibition.