Biology of Marine Fungi
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Chlamydospore Agar M113 Chlamydospore Agar Is Used for Differentiating Candida Albicans from Other Species of Candida on the Basis of Chlamydospore Formation
HiMedia Laboratories Technical Data Chlamydospore Agar M113 Chlamydospore Agar is used for differentiating Candida albicans from other species of Candida on the basis of chlamydospore formation. Composition** Ingredients Gms / Litre Ammonium sulphate 1.000 Monopotassium phosphate 1.000 Biotin 0.000005 Trypan blue 0.100 Purified polysaccharide 20.000 Agar 15.000 Final pH ( at 25°C) 5.1±0.2 **Formula adjusted, standardized to suit performance parameters Directions Suspend 37.1 grams in 1000 ml distilled water. Heat to boiling to dissolve the medium completely. Sterilize by autoclaving at 15 lbs pressure (121°C) for 15 minutes. Mix well and pour into sterile Petri plates. Principle And Interpretation Candida albicans is a diploid sexual fungus (a form of yeast), and the causitive agent of opportunistic oral and vaginal infections in humans (1). C. albicans is a commensal of skin, gastrointestinal and genitourinary tract. However, under certain conditions overgrowth of this results into oesopharyngeal candidiasis, vulvovaginal candidiasis and candidemia. Chlamydospores formation is the most differential characteristic of C. albicans (1). Chlamydospore Agar was specially designed for the differentiation of C. albicans from other species on the basis of chlamydospores formation. It is prepared according to the formula of Nickerson and Mankowshi (2). Ammonium sulphate acts as sources of ions that simulate metabolism. Monopotassium phosphate provides buffering to the medium. Biotin provides the necessary vitamins required for metabolism. Purified polysaccharide acts as a source of carbon. Trypan blue is a vital dye absorbed selectively by the chlamydospores and imparts blue colour to chlamydospores, whereas the filaments are colourless. Test for chlamydospores: Scratch cut mark like X onto the agar surface with inoculum using sterile needle. -

Introduction to Mycology
INTRODUCTION TO MYCOLOGY The term "mycology" is derived from Greek word "mykes" meaning mushroom. Therefore mycology is the study of fungi. The ability of fungi to invade plant and animal tissue was observed in early 19th century but the first documented animal infection by any fungus was made by Bassi, who in 1835 studied the muscardine disease of silkworm and proved the that the infection was caused by a fungus Beauveria bassiana. In 1910 Raymond Sabouraud published his book Les Teignes, which was a comprehensive study of dermatophytic fungi. He is also regarded as father of medical mycology. Importance of fungi: Fungi inhabit almost every niche in the environment and humans are exposed to these organisms in various fields of life. Beneficial Effects of Fungi: 1. Decomposition - nutrient and carbon recycling. 2. Biosynthetic factories. The fermentation property is used for the industrial production of alcohols, fats, citric, oxalic and gluconic acids. 3. Important sources of antibiotics, such as Penicillin. 4. Model organisms for biochemical and genetic studies. Eg: Neurospora crassa 5. Saccharomyces cerviciae is extensively used in recombinant DNA technology, which includes the Hepatitis B Vaccine. 6. Some fungi are edible (mushrooms). 7. Yeasts provide nutritional supplements such as vitamins and cofactors. 8. Penicillium is used to flavour Roquefort and Camembert cheeses. 9. Ergot produced by Claviceps purpurea contains medically important alkaloids that help in inducing uterine contractions, controlling bleeding and treating migraine. 10. Fungi (Leptolegnia caudate and Aphanomyces laevis) are used to trap mosquito larvae in paddy fields and thus help in malaria control. Harmful Effects of Fungi: 1. -

1 Differential Morphological Changes in Response to Environmental Stimuli in A
bioRxiv preprint doi: https://doi.org/10.1101/372078; this version posted July 19, 2018. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. 1 Differential morphological changes in response to environmental stimuli in a 2 fungal plant pathogen 3 4 Carolina Sardinha Franciscoa, Xin Maa, Maria Manuela Zwyssiga, Bruce A. McDonalda, 5 Javier Palma-Guerreroa 6 7 aPlant Pathology Group, Institute of Integrative Biology, ETH Zürich, 8092 Zürich, 8 Switzerland 9 10 Running Title: Pleomorphism in Zymoseptoria tritici 11 12 #Address correspondence to Javier Palma-Guerrero, [email protected] 13 1 bioRxiv preprint doi: https://doi.org/10.1101/372078; this version posted July 19, 2018. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. 14 ABSTRACT 15 During their life cycles, pathogens have to adapt to many biotic and abiotic 16 environmental constraints to maximize their overall fitness. Morphological transitions are 17 one of the least understood of the many strategies employed by fungal plant pathogens 18 to adapt to constantly changing environments. We characterized the responses of the 19 wheat pathogen Zymoseptoria tritici to a series of environmental stimuli using 20 microscopy, transcriptomic analyses, and survival assays to explore the effects of 21 changing environments on morphology and adaptation. We found that all tested stimuli 22 induced morphological changes, with distinct responses observed among four different 23 strains. The transcription analyses indicated a co-regulation of morphogenesis and 24 virulence factors in Z. -

Escovopsis Trichodermoides Sp. Nov., Isolated from a Nest of the Lower Attine Ant Mycocepurus Goeldii
Antonie van Leeuwenhoek (2015) 107:731–740 DOI 10.1007/s10482-014-0367-1 ORIGINAL PAPER Escovopsis trichodermoides sp. nov., isolated from a nest of the lower attine ant Mycocepurus goeldii Virginia E. Masiulionis • Marta N. Cabello • Keith A. Seifert • Andre Rodrigues • Fernando C. Pagnocca Received: 30 April 2014 / Accepted: 18 December 2014 / Published online: 10 January 2015 Ó Springer International Publishing Switzerland 2015 Abstract Currently, five species are formally other species by highly branched, trichoderma-like described in Escovopsis, a specialized mycoparasitic conidiophores lacking swollen vesicles, with reduced genus of fungus gardens of attine ants (Hymenoptera: conidiogenous cells and distinctive conidia morphol- Formicidae: tribe Attini). Four species were isolated ogy. Phylogenetic analyses based on partial tef1 gene from leaf-cutting ants in Brazil, including Escovopsis sequences support the distinctiveness of this species. moelleri and Escovopsis microspora from nests of A portion of the internal transcribed spacers of the Acromyrmex subterraneus molestans, Escovopsis nuclear rDNA was sequenced to serve as a DNA weberi from a nest of Atta sp. and Escovopsis barcode. Future molecular and morphological studies lentecrescens from a nest of Acromyrmex subterran- in this group of fungi will certainly unravel the eus subterraneus. The fifth species, Escovopsis taxonomic diversity of Escovopsis associated with aspergilloides was isolated from a nest of the higher fungus-growing ants. attine ant Trachymyrmex ruthae from Trinidad. Here, we describe a new species, Escovopsis trichodermo- Keywords Attini Á Fungus-growing ant Á ides isolated from a fungus garden of the lower attine Hypocreales Á Mycoparasitism ant Mycocepurus goeldii, which differs from the five V. -

Homarus Gammarus ) Reveals Its Generalist Parasitic Strategy in Marine Invertebrates
Title Halioticida noduliformans infection in eggs of lobster ( Homarus gammarus ) reveals its generalist parasitic strategy in marine invertebrates Authors Holt, C; Foster, R; Daniels, CL; van der Giezen, M; Feist, SW; Stentiford, GD; Bass, D Description publisher: Elsevier articletitle: Halioticida noduliformans infection in eggs of lobster (Homarus gammarus) reveals its generalist parasitic strategy in marine invertebrates journaltitle: Journal of Invertebrate Pathology articlelink: http://dx.doi.org/10.1016/ j.jip.2018.03.002 content_type: article copyright: © 2018 The Authors. Published by Elsevier Inc.; 0000-0002-6719-5565 Date Submitted 2018-06-02 Journal of Invertebrate Pathology xxx (xxxx) xxx–xxx Contents lists available at ScienceDirect Journal of Invertebrate Pathology journal homepage: www.elsevier.com/locate/jip Halioticida noduliformans infection in eggs of lobster (Homarus gammarus) reveals its generalist parasitic strategy in marine invertebrates ⁎ Corey Holta,b,c, , Rachel Fosterd, Carly L. Danielsc, Mark van der Giezenb, Stephen W. Feista, Grant D. Stentiforda, David Bassa,d a Pathology and Microbial Systematics, Centre for Environment, Fisheries and Aquaculture Science (Cefas), Barrack Road, Weymouth, Dorset DT4 8UB, United Kingdom b Biosciences, University of Exeter, Stocker Road, Exeter EX4 4QD, United Kingdom c The National Lobster Hatchery, South Quay, Padstow PL28 9BL, United Kingdom d The Natural History Museum, Cromwell Road, Kensington, London SW7 5BD, United Kingdom ARTICLE INFO ABSTRACT Keywords: A parasite exhibiting Oomycete-like morphology and pathogenesis was isolated from discoloured eggs of the Halioticida noduliformans European lobster (Homarus gammarus) and later found in gill tissues of adults. Group-specific Oomycete primers Homarus gammarus were designed to amplify the 18S ribosomal small subunit (SSU), which initially identified the organism as the Haliphthoros same as the ‘Haliphthoros’ sp. -

Inoculum Size Effect in Dimorphic Fungi: Extracellular Control of Yeast-Mycelium Dimorphism in Ceratocystis Ulmi
University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Papers in Microbiology Papers in the Biological Sciences 3-1-2004 Inoculum Size Effect in Dimorphic Fungi: Extracellular Control of Yeast-Mycelium Dimorphism in Ceratocystis ulmi Jacob M. Hornby University of Nebraska-Lincoln, [email protected] Sarah M. Jacobitz-Kizzier University of Nebraska-Lincoln Donna J. McNeel University of Nebraska-Lincoln Ellen C. Jensen University of Nebraska-Lincoln, [email protected] David S. Treves University of Nebraska-Lincoln See next page for additional authors Follow this and additional works at: https://digitalcommons.unl.edu/bioscimicro Part of the Microbiology Commons Hornby, Jacob M.; Jacobitz-Kizzier, Sarah M.; McNeel, Donna J.; Jensen, Ellen C.; Treves, David S.; and Nickerson, Kenneth W., "Inoculum Size Effect in Dimorphic Fungi: Extracellular Control of Yeast-Mycelium Dimorphism in Ceratocystis ulmi" (2004). Papers in Microbiology. 37. https://digitalcommons.unl.edu/bioscimicro/37 This Article is brought to you for free and open access by the Papers in the Biological Sciences at DigitalCommons@University of Nebraska - Lincoln. It has been accepted for inclusion in Papers in Microbiology by an authorized administrator of DigitalCommons@University of Nebraska - Lincoln. Authors Jacob M. Hornby, Sarah M. Jacobitz-Kizzier, Donna J. McNeel, Ellen C. Jensen, David S. Treves, and Kenneth W. Nickerson This article is available at DigitalCommons@University of Nebraska - Lincoln: https://digitalcommons.unl.edu/ bioscimicro/37 APPLIED AND ENVIRONMENTAL MICROBIOLOGY, Mar. 2004, p. 1356–1359 Vol. 70, No. 3 0099-2240/04/$08.00ϩ0 DOI: 10.1128/AEM.70.3.1356–1359.2004 Copyright © 2004, American Society for Microbiology. All Rights Reserved. -

The Crustacean Society Mid-Year Meeting 2019
THE CRUSTACEAN SOCIETY MID-YEAR MEETING 2019 ABSTRACT BOOKLET Table of Contents PLENARY LECTURES ........................................................................................................... 1 ORAL PRESENTATIONS ...................................................................................................... 7 SYMPOSIUM 1: Frontiers in Crustacean Biology: Asian Perspectives ................................ 43 SYMPOSIUM 2: Recent Advances in Caridean Systematics ............................................... 53 SYMPOSIUM 3: Evolution and Ecology of Parasitic and Symbiotic Crustaceans ................ 59 SYMPOSIUM 4: Biology of Freshwater Crayfish ................................................................ 69 SYMPOSIUM 5: Deep-sea Biodiversity: A Crustacean Perspective .................................... 77 SYMPOSIUM 6: Comparative Endocrinology and Genomics in Arthropods ....................... 87 SYMPOSIUM 7: Fossil and Modern Clam Shrimp .............................................................. 97 SYMPOSIUM 8: Aquaculture Biotechnology of Crabs ..................................................... 108 POSTER PRESENTATIONS ............................................................................................... 114 PLENARY LECTURES PL1 Effects of temperature variations on reproduction: Transduction of physiological stress through species interactions between two porcelain crabs B. TSUKIMURA1, ALEX GUNDERSON2, JONATHON STILLMAN3 1. California State University, Fresno, USA 2. Tulane University, USA 3. -

Four New Ophiostoma Species Associated with Conifer- and Hardwood-Infesting Bark and Ambrosia Beetles from the Czech Republic and Poland
Antonie van Leeuwenhoek (2019) 112:1501–1521 https://doi.org/10.1007/s10482-019-01277-5 (0123456789().,-volV)( 0123456789().,-volV) ORIGINAL PAPER Four new Ophiostoma species associated with conifer- and hardwood-infesting bark and ambrosia beetles from the Czech Republic and Poland Robert Jankowiak . Piotr Bilan´ski . Beata Strzałka . Riikka Linnakoski . Agnieszka Bosak . Georg Hausner Received: 30 November 2018 / Accepted: 14 May 2019 / Published online: 28 May 2019 Ó The Author(s) 2019 Abstract Fungi under the order Ophiostomatales growth rates, and their insect associations. Based on (Ascomycota) are known to associate with various this study four new taxa can be circumscribed and the species of bark beetles (Coleoptera: Curculionidae: following names are provided: Ophiostoma pityok- Scolytinae). In addition this group of fungi contains teinis sp. nov., Ophiostoma rufum sp. nov., Ophios- many taxa that can impart blue-stain on sapwood and toma solheimii sp. nov., and Ophiostoma taphrorychi some are important tree pathogens. A recent survey sp. nov. O. rufum sp. nov. is a member of the that focussed on the diversity of the Ophiostomatales Ophiostoma piceae species complex, while O. pityok- in the forest ecosystems of the Czech Republic and teinis sp. nov. resides in a discrete lineage within Poland uncovered four putative new species. Phylo- Ophiostoma s. stricto. O. taphrorychi sp. nov. together genetic analyses of four gene regions (ITS1-5.8S-ITS2 with O. distortum formed a well-supported clade in region, ß-tubulin, calmodulin, and translation elonga- Ophiostoma s. stricto close to O. pityokteinis sp. nov. tion factor 1-a) indicated that these four species are O. -
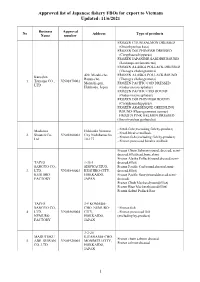
Approved List of Japanese Fishery Fbos for Export to Vietnam Updated: 11/6/2021
Approved list of Japanese fishery FBOs for export to Vietnam Updated: 11/6/2021 Business Approval No Address Type of products Name number FROZEN CHUM SALMON DRESSED (Oncorhynchus keta) FROZEN DOLPHINFISH DRESSED (Coryphaena hippurus) FROZEN JAPANESE SARDINE ROUND (Sardinops melanostictus) FROZEN ALASKA POLLACK DRESSED (Theragra chalcogramma) 420, Misaki-cho, FROZEN ALASKA POLLACK ROUND Kaneshin Rausu-cho, (Theragra chalcogramma) 1. Tsuyama CO., VN01870001 Menashi-gun, FROZEN PACIFIC COD DRESSED LTD Hokkaido, Japan (Gadus macrocephalus) FROZEN PACIFIC COD ROUND (Gadus macrocephalus) FROZEN DOLPHIN FISH ROUND (Coryphaena hippurus) FROZEN ARABESQUE GREENLING ROUND (Pleurogrammus azonus) FROZEN PINK SALMON DRESSED (Oncorhynchus gorbuscha) - Fresh fish (excluding fish by-product) Maekawa Hokkaido Nemuro - Fresh bivalve mollusk. 2. Shouten Co., VN01860002 City Nishihamacho - Frozen fish (excluding fish by-product) Ltd 10-177 - Frozen processed bivalve mollusk Frozen Chum Salmon (round, dressed, semi- dressed,fillet,head,bone,skin) Frozen Alaska Pollack(round,dressed,semi- TAIYO 1-35-1 dressed,fillet) SANGYO CO., SHOWACHUO, Frozen Pacific Cod(round,dressed,semi- 3. LTD. VN01840003 KUSHIRO-CITY, dressed,fillet) KUSHIRO HOKKAIDO, Frozen Pacific Saury(round,dressed,semi- FACTORY JAPAN dressed) Frozen Chub Mackerel(round,fillet) Frozen Blue Mackerel(round,fillet) Frozen Salted Pollack Roe TAIYO 3-9 KOMABA- SANGYO CO., CHO, NEMURO- - Frozen fish 4. LTD. VN01860004 CITY, - Frozen processed fish NEMURO HOKKAIDO, (excluding by-product) FACTORY JAPAN -

Chinese Red Swimming Crab (Portunus Haanii) Fishery Improvement Project (FIP) in Dongshan, China (August-December 2018)
Chinese Red Swimming Crab (Portunus haanii) Fishery Improvement Project (FIP) in Dongshan, China (August-December 2018) Prepared by Min Liu & Bai-an Lin Fish Biology Laboratory College of Ocean and Earth Sciences, Xiamen University March 2019 Contents 1. Introduction........................................................................................................ 5 2. Materials and Methods ...................................................................................... 6 2.1. Study site and survey frequency .................................................................... 6 2.2. Sample collection .......................................................................................... 7 2.3. Species identification................................................................................... 10 2.4. Sample measurement ................................................................................... 11 2.5. Interviews.................................................................................................... 13 2.6. Estimation of annual capture volume of Portunus haanii ............................. 15 3. Results .............................................................................................................. 15 3.1. Species diversity.......................................................................................... 15 3.1.1. Species composition .............................................................................. 15 3.1.2. ETP species ......................................................................................... -

Entomopathogenic Fungal Identification
Entomopathogenic Fungal Identification updated November 2005 RICHARD A. HUMBER USDA-ARS Plant Protection Research Unit US Plant, Soil & Nutrition Laboratory Tower Road Ithaca, NY 14853-2901 Phone: 607-255-1276 / Fax: 607-255-1132 Email: Richard [email protected] or [email protected] http://arsef.fpsnl.cornell.edu Originally prepared for a workshop jointly sponsored by the American Phytopathological Society and Entomological Society of America Las Vegas, Nevada – 7 November 1998 - 2 - CONTENTS Foreword ......................................................................................................... 4 Important Techniques for Working with Entomopathogenic Fungi Compound micrscopes and Köhler illumination ................................... 5 Slide mounts ........................................................................................ 5 Key to Major Genera of Fungal Entomopathogens ........................................... 7 Brief Glossary of Mycological Terms ................................................................. 12 Fungal Genera Zygomycota: Entomophthorales Batkoa (Entomophthoraceae) ............................................................... 13 Conidiobolus (Ancylistaceae) .............................................................. 14 Entomophaga (Entomophthoraceae) .................................................. 15 Entomophthora (Entomophthoraceae) ............................................... 16 Neozygites (Neozygitaceae) ................................................................. 17 Pandora -

Transcriptional Profiling of Spiny Lobster Metamorphosis Reveals Three New Additions to the Nuclear Receptor Superfamily Cameron J
Hyde et al. BMC Genomics (2019) 20:531 https://doi.org/10.1186/s12864-019-5925-5 RESEARCH ARTICLE Open Access Transcriptional profiling of spiny lobster metamorphosis reveals three new additions to the nuclear receptor superfamily Cameron J. Hyde1, Quinn P. Fitzgibbon2, Abigail Elizur1, Gregory G. Smith2 and Tomer Ventura1* Abstract Background: The Crustacea are an evolutionarily diverse taxon which underpins marine food webs and contributes significantly to the global economy. However, our knowledge of crustacean endocrinology and development is far behind that of terrestrial arthropods. Here we present a unique insight into the molecular pathways coordinating crustacean metamorphosis, by reconciling nuclear receptor (NR) gene activity from a 12-stage, 3-replicate transcriptome in the ornate spiny lobster (Panulirus ornatus) during larval development. Results: We annotated 18 distinct nuclear receptor genes, including three novel NRs which are upregulated prior to metamorphosis and have hence been named the “molt-associated receptors” (MARs). We also demonstrate the ecdysone-responsive expression of several known molt-related NRs including ecdysone receptor, fushi-tarazu-F1 and E75. Phylogenetic analysis of the curated NR family confirmed gene annotations and suggested that the MARs are a recent addition to the crustacean superfamily, occurring across the Malacostraca from the Stomatopoda to the Decapoda. The ligand-binding domain of these receptors appears to be less conserved than that of typical group-1 NRs. Expression data from two other crustacean species was utilized to examine MAR expression. The Y-organ of the tropical land crab showed a decline in expression of all MARs from intermolt to post-molt. Tissue distributions showed gonad-enriched expression in the Eastern rock lobster and antennal gland-enriched expression in the tropical land crab, although expression was evident across most tissues.