Bacterial Assembly and Temporal Dynamics in Activated Sludge of a Full-Scale Municipal Wastewater Treatment Plant
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Spatio-Temporal Study of Microbiology in the Stratified Oxic-Hypoxic-Euxinic, Freshwater- To-Hypersaline Ursu Lake
Spatio-temporal insights into microbiology of the freshwater-to- hypersaline, oxic-hypoxic-euxinic waters of Ursu Lake Baricz, A., Chiriac, C. M., Andrei, A-., Bulzu, P-A., Levei, E. A., Cadar, O., Battes, K. P., Cîmpean, M., enila, M., Cristea, A., Muntean, V., Alexe, M., Coman, C., Szekeres, E. K., Sicora, C. I., Ionescu, A., Blain, D., O’Neill, W. K., Edwards, J., ... Banciu, H. L. (2020). Spatio-temporal insights into microbiology of the freshwater-to- hypersaline, oxic-hypoxic-euxinic waters of Ursu Lake. Environmental Microbiology. https://doi.org/10.1111/1462-2920.14909, https://doi.org/10.1111/1462-2920.14909 Published in: Environmental Microbiology Document Version: Peer reviewed version Queen's University Belfast - Research Portal: Link to publication record in Queen's University Belfast Research Portal Publisher rights Copyright 2019 Wiley. This work is made available online in accordance with the publisher’s policies. Please refer to any applicable terms of use of the publisher. General rights Copyright for the publications made accessible via the Queen's University Belfast Research Portal is retained by the author(s) and / or other copyright owners and it is a condition of accessing these publications that users recognise and abide by the legal requirements associated with these rights. Take down policy The Research Portal is Queen's institutional repository that provides access to Queen's research output. Every effort has been made to ensure that content in the Research Portal does not infringe any person's rights, or applicable UK laws. If you discover content in the Research Portal that you believe breaches copyright or violates any law, please contact [email protected]. -

Meiothermus Ruber Type Strain (21T)
Standards in Genomic Sciences (2010) 3:26-36 DOI:10.4056/sigs.1032748 Complete genome sequence of Meiothermus ruber type strain (21T) Brian J Tindall1, Johannes Sikorski1, Susan Lucas2, Eugene Goltsman2, Alex Copeland2, Tijana Glavina Del Rio2, Matt Nolan2, Hope Tice2, Jan-Fang Cheng2, Cliff Han2,3, Sam Pitluck2, Konstantinos Liolios2, Natalia Ivanova2, Konstantinos Mavromatis2, Galina Ovchinnikova2, Amrita Pati2, Regine Fähnrich1, Lynne Goodwin2,3, Amy Chen4, Krishna Palaniappan4, Miriam Land2,5, Loren Hauser2,5, Yun-Juan Chang2,5, Cynthia D. Jeffries2,5, Manfred Rohde6, Markus Göker1, Tanja Woyke2, James Bristow2, Jonathan A. Eisen2,7, Victor Markowitz4, Philip Hugenholtz2, Nikos C. Kyrpides2, Hans-Peter Klenk1, and Alla Lapidus2* 1 DSMZ - German Collection of Microorganisms and Cell Cultures GmbH, Braunschweig, Germany 2 DOE Joint Genome Institute, Walnut Creek, California, USA 3 Los Alamos National Laboratory, Bioscience Division, Los Alamos, New Mexico, USA 4 Biological Data Management and Technology Center, Lawrence Berkeley National Laboratory, Berkeley, California, USA 5 Oak Ridge National Laboratory, Oak Ridge, Tennessee, USA 6 HZI – Helmholtz Centre for Infection Research, Braunschweig, Germany 7 University of California Davis Genome Center, Davis, California, USA *Corresponding author: Alla Lapidus Keywords: thermophilic, aerobic, non-motile, free-living, Gram-negative, Thermales, Deino- cocci, GEBA Meiothermus ruber (Loginova et al. 1984) Nobre et al. 1996 is the type species of the genus Meiothermus. This thermophilic genus is of special interest, as its members share relatively low degrees of 16S rRNA gene sequence similarity and constitute a separate evolutionary li- neage from members of the genus Thermus, from which they can generally be distinguished by their slightly lower temperature optima. -

NC10 Phylum Anaerobs? Methanotrophs? Where Can I Find Them?
NC10 phylum Anaerobs? Methanotrophs? Where can I find them? Beate Kraft Microbial Diversity 2012 Abstract Methylomirabilis oxyfera, the only cultured member of the NC10 phylum performs the newly discovered pathway of NO-dismutation. In this study different habitats were screened for the presence of member of this phylum and the diversity of the sequences obtained was analyzed. Furthermore enrichment cultures for nitrite reduction coupled to methane oxidation were set up. Indeed, the presence of NC10 seems to be associated with nitrite and methane rich fresh water environments. Introduction Recently a new nitrite reduction pathway, NO-dismutation, coupled to methane oxidation has been discovered (Ettwig et al. 2010). In NO-dismutation nitrite is oxidized to NO as in denitrification but then NO dimutated into N2 and O2 instead of being further reduced to N2O. The generated oxygen is then used for methane oxidation. The organism responsible for this process is Methylomirabils oxfera. It is the only cultured member of the candidate phylum NC10. Fig 1: Pathway of NO-dismutation (from Ettwig et al. 2010) It remains open if other members of the NC10 phylum share the same metabolism and respiratory pathway or if they are metabolically more diverse. 16S sequences that fall into the NC10 phylum have been found in mainly oxygen limited freshwater habitats such as lakes including lake sediments, rice paddy soils, wastewater sludge and ditches (Deutzmann and Schink 2011, Luesken et a.l 2011). Their occurrence apparently seem to be correlated with the presence of methane and nitrite in the habitat. This would suggest a similar respiratory pathway as in Methylomirabilis oxyfera. -

Yu-Chen Ling and John W. Moreau
Microbial Distribution and Activity in a Coastal Acid Sulfate Soil System Introduction: Bioremediation in Yu-Chen Ling and John W. Moreau coastal acid sulfate soil systems Method A Coastal acid sulfate soil (CASS) systems were School of Earth Sciences, University of Melbourne, Melbourne, VIC 3010, Australia formed when people drained the coastal area Microbial distribution controlled by environmental parameters Microbial activity showed two patterns exposing the soil to the air. Drainage makes iron Microbial structures can be grouped into three zones based on the highest similarity between samples (Fig. 4). Abundant populations, such as Deltaproteobacteria, kept constant activity across tidal cycling, whereas rare sulfides oxidize and release acidity to the These three zones were consistent with their geological background (Fig. 5). Zone 1: Organic horizon, had the populations changed activity response to environmental variations. Activity = cDNA/DNA environment, low pH pore water further dissolved lowest pH value. Zone 2: surface tidal zone, was influenced the most by tidal activity. Zone 3: Sulfuric zone, Abundant populations: the heavy metals. The acidity and toxic metals then Method A Deltaproteobacteria Deltaproteobacteria this area got neutralized the most. contaminate coastal and nearby ecosystems and Method B 1.5 cause environmental problems, such as fish kills, 1.5 decreased rice yields, release of greenhouse gases, Chloroflexi and construction damage. In Australia, there is Gammaproteobacteria Gammaproteobacteria about a $10 billion “legacy” from acid sulfate soils, Chloroflexi even though Australia is only occupied by around 1.0 1.0 Cyanobacteria,@ Acidobacteria Acidobacteria Alphaproteobacteria 18% of the global acid sulfate soils. Chloroplast Zetaproteobacteria Rare populations: Alphaproteobacteria Method A log(RNA(%)+1) Zetaproteobacteria log(RNA(%)+1) Method C Method B 0.5 0.5 Cyanobacteria,@ Bacteroidetes Chloroplast Firmicutes Firmicutes Bacteroidetes Planctomycetes Planctomycetes Ac8nobacteria Fig. -
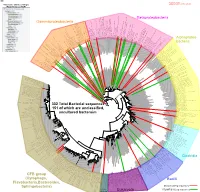
Bacteria Clostridia Bacilli Eukaryota CFB Group
AM935842.1.1361 uncultured Burkholderiales bacterium Class Betaproteobacteria AY283260.1.1552 Alcaligenes sp. PCNB−2 Class Betaproteobacteria AM934953.1.1374 uncultured Burkholderiales bacterium Class Betaproteobacteria AJ581593.1.1460 uncultured betaAM936569.1.1351 proteobacterium uncultured Class Betaproteobacteria Derxia sp. Class Betaproteobacteria AJ581621.1.1418 uncultured beta proteobacterium Class Betaproteobacteria DQ248272.1.1498 uncultured soil bacterium soil uncultured DQ248272.1.1498 DQ248235.1.1498 uncultured soil bacterium RS49 DQ248270.1.1496 uncultured soil bacterium DQ256489.1.1211 Variovorax paradoxus Class Betaproteobacteria Class paradoxus Variovorax DQ256489.1.1211 AF523053.1.1486 uncultured Comamonadaceae bacterium Class Betaproteobacteria AY706442.1.1396 uncultured bacterium uncultured AY706442.1.1396 AJ536763.1.1422 uncultured bacterium CS000359.1.1530 Variovorax paradoxus Class Betaproteobacteria Class paradoxus Variovorax CS000359.1.1530 AY168733.1.1411 uncultured bacterium AJ009470.1.1526 uncultured bacterium SJA−62 Class Betaproteobacteria Class SJA−62 bacterium uncultured AJ009470.1.1526 AY212561.1.1433 uncultured bacterium D16212.1.1457 Rhodoferax fermentans Class Betaproteobacteria Class fermentans Rhodoferax D16212.1.1457 AY957894.1.1546 uncultured bacterium AJ581620.1.1452 uncultured beta proteobacterium Class Betaproteobacteria RS76 AY625146.1.1498 uncultured bacterium RS65 DQ316832.1.1269 uncultured beta proteobacterium Class Betaproteobacteria DQ404909.1.1513 uncultured bacterium uncultured DQ404909.1.1513 AB021341.1.1466 bacterium rM6 AJ487020.1.1500 uncultured bacterium uncultured AJ487020.1.1500 RS7 RS86RC AF364862.1.1425 bacterium BA128 Class Betaproteobacteria AY957931.1.1529 uncultured bacterium uncultured AY957931.1.1529 CP000884.723807.725332 Delftia acidovorans SPH−1 Class Betaproteobacteria AY957923.1.1520 uncultured bacterium uncultured AY957923.1.1520 RS18 AY957918.1.1527 uncultured bacterium uncultured AY957918.1.1527 AY945883.1.1500 uncultured bacterium AF526940.1.1489 uncultured Ralstonia sp. -

The Phylogenetic Composition and Structure of Soil Microbial Communities Shifts in Response to Elevated Carbon Dioxide
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by University of Minnesota Digital Conservancy The ISME Journal (2012) 6, 259–272 & 2012 International Society for Microbial Ecology All rights reserved 1751-7362/12 www.nature.com/ismej ORIGINAL ARTICLE The phylogenetic composition and structure of soil microbial communities shifts in response to elevated carbon dioxide Zhili He1, Yvette Piceno2, Ye Deng1, Meiying Xu1,3, Zhenmei Lu1,4, Todd DeSantis2, Gary Andersen2, Sarah E Hobbie5, Peter B Reich6 and Jizhong Zhou1,2 1Institute for Environmental Genomics, Department of Botany and Microbiology, University of Oklahoma, Norman, OK, USA; 2Ecology Department, Earth Sciences Division, Lawrence Berkeley National Laboratory, Berkeley, CA, USA; 3Guangdong Provincial Key Laboratory of Microbial Culture Collection and Application, Guangdong Institute of Microbiology, Guangzhou, China; 4College of Life Sciences, Zhejiang University, Hangzhou, China; 5Department of Ecology, Evolution, and Behavior, St Paul, MN, USA and 6Department of Forest Resources, University of Minnesota, St Paul, MN, USA One of the major factors associated with global change is the ever-increasing concentration of atmospheric CO2. Although the stimulating effects of elevated CO2 (eCO2) on plant growth and primary productivity have been established, its impacts on the diversity and function of soil microbial communities are poorly understood. In this study, phylogenetic microarrays (PhyloChip) were used to comprehensively survey the richness, composition and structure of soil microbial communities in a grassland experiment subjected to two CO2 conditions (ambient, 368 p.p.m., versus elevated, 560 p.p.m.) for 10 years. The richness based on the detected number of operational taxonomic units (OTUs) significantly decreased under eCO2. -

Being Aquifex Aeolicus: Untangling a Hyperthermophile's Checkered Past
GBE Being Aquifex aeolicus: Untangling a Hyperthermophile’s Checkered Past Robert J.M. Eveleigh1,2, Conor J. Meehan1,2,JohnM.Archibald1, and Robert G. Beiko2,* 1Department of Biochemistry and Molecular Biology, Dalhousie University, Halifax, Nova Scotia, Canada 2Faculty of Computer Science, Dalhousie University, Halifax, Nova Scotia, Canada *Corresponding author: E-mail: [email protected]. Accepted: November 22, 2013 Abstract Lateral gene transfer (LGT) is an important factor contributing to the evolution of prokaryotic genomes. The Aquificae are a hyper- thermophilic bacterial group whose genes show affiliations to many other lineages, including the hyperthermophilic Thermotogae, the Proteobacteria, and the Archaea. Previous phylogenomic analyses focused on Aquifex aeolicus identified Thermotogae and Downloaded from Aquificae either as successive early branches or sisters in a rooted bacterial phylogeny, but many phylogenies and cellular traits have suggested a stronger affiliation with the Epsilonproteobacteria. Different scenarios for the evolution of the Aquificae yield different phylogenetic predictions. Here, we outline these scenarios and consider the fit of the available data, including three sequenced Aquificae genomes, to different sets of predictions. Evidence from phylogenetic profiles and trees suggests that the Epsilonproteobacteria have the strongest affinities with the three Aquificae analyzed. However, this pattern is shown by only a http://gbe.oxfordjournals.org/ minority of encoded proteins, and the Archaea, many lineages of thermophilic bacteria, and members of genus Clostridium and class Deltaproteobacteria also show strong connections to the Aquificae. The phylogenetic affiliations of different functional subsystems showed strong biases: Most but not all genes implicated in the core translational apparatus tended to group Aquificae with Thermotogae, whereas a wide range of metabolic and cellular processes strongly supported the link between Aquificae and Epsilonproteobacteria. -

Research Article Review Jmb
J. Microbiol. Biotechnol. (2017), 27(0), 1–7 https://doi.org/10.4014/jmb.1707.07027 Research Article Review jmb Methods 20,546 sequences and all the archaeal datasets were normalized to 21,154 sequences by the “sub.sample” Bioinformatics Analysis command. The filtered sequences were classified against The raw read1 and read2 datasets was demultiplexed by the SILVA 16S reference database (Release 119) using a trimming the barcode sequences with no more than 1 naïve Bayesian classifier built in Mothur with an 80% mismatch. Then the sequences with the same ID were confidence score [5]. Sequences passing through all the picked from the remaining read1 and read2 datasets by a filtration were also clustered into OTUs at 6% dissimilarity self-written python script. Bases with average quality score level. Then a “classify.otu” function was utilized to assign lower than 25 over a 25 bases sliding window were the phylogenetic information to each OTU. excluded and sequences which contained any ambiguous base or had a final length shorter than 200 bases were Reference abandoned using Sickle [1]. The paired reads were assembled into contigs and any contigs with an ambiguous 1. Joshi NA, FJ. 2011. Sickle: A sliding-window, adaptive, base, more than 8 homopolymeric bases and fewer than 10 quality-based trimming tool for FastQ files (Version 1.33) bp overlaps were culled. After that, the contigs were [Software]. further trimmed to get rid of the contigs that have more 2. Schloss PD. 2010. The Effects of Alignment Quality, than 1 forward primer mismatch and 2 reverse primer Distance Calculation Method, Sequence Filtering, and Region on the Analysis of 16S rRNA Gene-Based Studies. -

The Environment, Diversity and Activity of Microbial Communities in Submarine Freshwater Springs in the Dead Sea
The environment, diversity and activity of microbial communities in submarine freshwater springs in the Dead Sea Dissertation Zur Erlangung des Grades eines Doktors der Naturwissenschaften - Dr. rer. nat. - dem Fachbereich Geowissenschaften der Universität Bremen vorgelegt an Stefan Häusler Bremen, April 2014 Die vorliegende Arbeit wurde in der Zeit von November 2010 bis April 2014 am Max- Planck-Institut für Marine Mikrobiologie in Bremen angefertigt. 1. Gutachter: Prof. Dr. Marcel M.M. Kuypers 2. Gutachter: Prof. Dr. Thorsten Dittmar Datum des Promotionskolloquiums: 16. Mai 2014 Contents Summary 1 Zusammenfassung 3 Chapter 1 7 Introduction 7 Motivation and objectives 23 Overview of enclosed manuscripts 35 Chapter 2 39 Microbial and chemical characterization of underwater fresh water springs in the Dead Sea Chapter 3 105 Microenvironments of reduced salinity harbor biofilms in Dead Sea underwater springs Chapter 4 125 Spatial distribution of diatom and cyanobacterial microbial mats in the Dead Sea is determined by response to rapid salinity fluctuations Chapter 5 153 Sulfate reduction and sulfide oxidation in extremely steep salinity gradients formed by freshwater springs emerging into the Dead Sea Chapter 6 189 Conclusions and perspectives Contributed work 201 Acknowledgements 211 Summary Summary The Dead Sea, located at the border between Jordan, Israel and the Palestinian authority is one of the most hypersaline lakes on earth. Its waters contain a total dissolved salt concentration of up to 348 g L-1, which is about 10 times higher than regular sea water. The lake is characterized by elevated concentrations of divalent cations (~2 M Mg2+ and ~0.5 M Ca2+), which, in addition to the high salinity, form an extreme environment where only highly adapted microorganisms can survive. -

The Ecology of the Chloroflexi in Full-Scale Activated Sludge 2 Wastewater Treatment Plants
bioRxiv preprint doi: https://doi.org/10.1101/335752; this version posted May 31, 2018. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. 1 The ecology of the Chloroflexi in full-scale activated sludge 2 wastewater treatment plants 3 Marta Nierychlo1, Aleksandra Miłobędzka2,3, Francesca Petriglieri1, Bianca 4 McIlroy1, Per Halkjær Nielsen1, and Simon Jon McIlroy1§* 5 1Center for Microbial Communities, Department of Chemistry and Bioscience, 6 Aalborg University, Aalborg, Denmark 7 2Microbial Ecology and Environmental Biotechnology Department, Institute of 8 Botany, Faculty of Biology, University of Warsaw; Biological and Chemical 9 Research Centre, Żwirki i Wigury 101, Warsaw 02-089, Poland 10 3Department of Biology, Faculty of Building Services, Hydro and Environmental 11 Engineering, Warsaw University of Technology, 00-653 Warsaw, Poland 12 * Corresponding author: Simon Jon McIlroy, Center for Microbial Communities, 13 Department of Chemistry and Bioscience, Aalborg University, Fredrik Bajers Vej 7H, 14 DK-9220 Aalborg, Denmark; Tel.: +45 9940 3573; Fax: +45 9814 1808; Email: 15 [email protected] 16 § Present address: Australian Centre for Ecogenomics, University of Queensland, 17 Australia 1 bioRxiv preprint doi: https://doi.org/10.1101/335752; this version posted May 31, 2018. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. -

1 Supplementary Material a Major Clade of Prokaryotes with Ancient
Supplementary Material A major clade of prokaryotes with ancient adaptations to life on land Fabia U. Battistuzzi and S. Blair Hedges Data assembly and phylogenetic analyses Protein data set: Amino acid sequences of 25 protein-coding genes (“proteins”) were concatenated in an alignment of 18,586 amino acid sites and 283 species. These proteins included: 15 ribosomal proteins (RPL1, 2, 3, 5, 6, 11, 13, 16; RPS2, 3, 4, 5, 7, 9, 11), four genes (RNA polymerase alpha, beta, and gamma subunits, Transcription antitermination factor NusG) from the functional category of Transcription, three proteins (Elongation factor G, Elongation factor Tu, Translation initiation factor IF2) of the Translation, Ribosomal Structure and Biogenesis functional category, one protein (DNA polymerase III, beta subunit) of the DNA Replication, Recombination and repair category, one protein (Preprotein translocase SecY) of the Cell Motility and Secretion category, and one protein (O-sialoglycoprotein endopeptidase) of the Posttranslational Modification, Protein Turnover, Chaperones category, as annotated in the Cluster of Orthologous Groups (COG) (Tatusov et al. 2001). After removal of multiple strains of the same species, GBlocks 0.91b (Castresana 2000) was applied to each protein in the concatenation to delete poorly aligned sites (i.e., sites with gaps in more than 50% of the species and conserved in less than 50% of the species) with the following parameters: minimum number of sequences for a conserved position: 110, minimum number of sequences for a flank position: 110, maximum number of contiguous non-conserved positions: 32000, allowed gap positions: with half. The signal-to-noise ratio was determined by altering the “minimum length of a block” parameter. -

Supplement of Biogeographical Distribution of Microbial Communities Along the Rajang River–South China Sea Continuum
Supplement of Biogeosciences, 16, 4243–4260, 2019 https://doi.org/10.5194/bg-16-4243-2019-supplement © Author(s) 2019. This work is distributed under the Creative Commons Attribution 4.0 License. Supplement of Biogeographical distribution of microbial communities along the Rajang River–South China Sea continuum Edwin Sien Aun Sia et al. Correspondence to: Moritz Müller ([email protected]) The copyright of individual parts of the supplement might differ from the CC BY 4.0 License. Supp. Fig. S1: Monthly Mean Precipitation (mm) from Jan 2016 to Sep 2017. Relevant months are highlighted red (Aug 2016), blue (Mar 2017) and dark blue (Sep 2017). Monthly precipitation for the period in between the cruises (August 2016 to September 2017) were obtained from the Tropical Rainfall Measuring Mission website (NASA 2019) in order to gauge the seasonality (wet or dry). As the rainfall data do not correlate with the monsoonal periods, the seasons in which the sampling cruises coincide with were classified based on the mean rainfall that occurred for each month. The August 2016 cruise (colored red) is classified as the dry season based on the lower mean rainfall value as compared to the other two (March 2017 and September 2017), in which the both are classified as the wet season. Supp. Fig. S2: Non-metric Multi-dimensional Scaling (NMDS) diagram of seasonal (August 2016, March 2017 and September 2017) and particle association (particle-attached or free-living) Seasonality was observed within the three cruises irrespective of the particle association (Supp. Fig. 3). The August 2016 cruise was found to cluster with the September 2017 whereas the March 2017 cruise clustered separately from the other two cruises.