Evaluating TNA Stability Under Simulated Physiological Conditions
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

(12) Patent Application Publication (10) Pub. No.: US 2013/0203610 A1 Meller Et Al
US 20130203610A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2013/0203610 A1 Meller et al. (43) Pub. Date: Aug. 8, 2013 (54) TOOLS AND METHOD FOR NANOPORES Related U.S. Application Data UNZIPPING-DEPENDENT NUCLECACID (60) Provisional application No. 61/318,872, filed on Mar. SEQUENCING 30, 2010. (75) Inventors: Amit Meller, Brookline, MA (US); Alon Publication Classification Singer, Brighton, MA (US) (51) Int. Cl. (73) Assignee: TRUSTEES OF BOSTON CI2O I/68 (2006.01) UNIVERSITY, Boston, MA (US) (52) U.S. Cl. CPC .................................... CI2O I/6874 (2013.01) (21) Appl. No.: 13/638,455 USPC ................................................. 506/6:506/16 (57) ABSTRACT (22) PCT Filed: Mar. 30, 2011 Provided herein is a library that comprises a plurality of molecular beacons (MBs), each MB having a detectable (86). PCT No.: PCT/US2O11AO3O430 label, a detectable label blocker and a modifier group. The S371 (c)(1), library is used in conjunction with nanopore unzipping-de (2), (4) Date: Apr. 17, 2013 pendent sequencing of nucleic acids. Patent Application Publication Aug. 8, 2013 Sheet 1 of 18 US 2013/020361.0 A1 . N s Patent Application Publication Aug. 8, 2013 Sheet 2 of 18 US 2013/020361.0 A1 I’9IAI ::::::::::: Dº3.modoueN Patent Application Publication Aug. 8, 2013 Sheet 3 of 18 US 2013/020361.0 A1 Z’9IAI ~~~~~~~~~);........ Patent Application Publication Aug. 8, 2013 Sheet 4 of 18 US 2013/020361.0 A1 s :·.{-zzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzzz iii. 9 iii.338 lii &S Patent Application Publication Aug. 8, 2013 Sheet 5 of 18 US 2013/020361.0 A1 s r Patent Application Publication Aug. -

Alternative Biochemistries for Alien Life: Basic Concepts and Requirements for the Design of a Robust Biocontainment System in Genetic Isolation
G C A T T A C G G C A T genes Review Alternative Biochemistries for Alien Life: Basic Concepts and Requirements for the Design of a Robust Biocontainment System in Genetic Isolation Christian Diwo 1 and Nediljko Budisa 1,2,* 1 Institut für Chemie, Technische Universität Berlin Müller-Breslau-Straße 10, 10623 Berlin, Germany; [email protected] 2 Department of Chemistry, University of Manitoba, 144 Dysart Rd, 360 Parker Building, Winnipeg, MB R3T 2N2, Canada * Correspondence: [email protected] or [email protected]; Tel.: +49-30-314-28821 or +1-204-474-9178 Received: 27 November 2018; Accepted: 21 December 2018; Published: 28 December 2018 Abstract: The universal genetic code, which is the foundation of cellular organization for almost all organisms, has fostered the exchange of genetic information from very different paths of evolution. The result of this communication network of potentially beneficial traits can be observed as modern biodiversity. Today, the genetic modification techniques of synthetic biology allow for the design of specialized organisms and their employment as tools, creating an artificial biodiversity based on the same universal genetic code. As there is no natural barrier towards the proliferation of genetic information which confers an advantage for a certain species, the naturally evolved genetic pool could be irreversibly altered if modified genetic information is exchanged. We argue that an alien genetic code which is incompatible with nature is likely to assure the inhibition of all mechanisms of genetic information transfer in an open environment. The two conceivable routes to synthetic life are either de novo cellular design or the successive alienation of a complex biological organism through laboratory evolution. -

Threose Nucleic Acid
2/23/11! Something else…! •" Neither the chicken nor the egg came first! Alternative Ideas! •" Transitional forms that were later discarded! Or was it the “egkin”?! Some experiments with peptide nucleic acid! Threose Nucleic Acid (TNA)! (PNA).! PNA: Peptide backbone with bases! •" Threose is one of two sugars with a four- sided ring! Can act as template for polymerization of RNA! •" Fewer issues with incorrect linkages, From activated nucleotides! selection of correct handedness! (Böhler, et al., Nature, 376, 578! •" Replace ribose sugar in RNA with threose! & comments by Piccirilli, pg. 548 17 Aug. 1995! •" Can base pair with RNA! •" Could have preceded RNA! PNA could be simpler to form under prebiotic conditions! Main point is that a simpler thing (not necessarily PNA) ! could have preceded RNA! 1! 2/23/11! Focus on Energy! Membranes! G. Wächtershäuser! Inorganic - organic connection! •" Membranes provide enclosure! FeS2 ! (Iron pyrite)! –"Also fundamental for metabolism! Attracts negatively charged molecules ! •" Membranes never arise from scratch! Surface catalysis provides energy via formation from! –"Always passed down and added to! FeS + H2S! –"All derived from ancestral cell! •" T. Cavalier-Smith proposes membranes! Scene is hot sulfur vents on sea floor! –"Plus nucleic acid formed “ob-cell”! Some successes in simulations! Amino acids formed peptide bonds! –"Merger of 2 ob-cells formed first cell! Thioester World! H O 1." Need precursor to RNA world! H H S H O Thioester! S C. de Duve! C C O 2. !Need energy conversion! In Vital Dust! O !Protometabolism! Thiol + Carboxyl! Background:! H O H H O H O Ester! O Thiols involved in metabolism, particularly in ancient ! C C pathways! O O Hydroxyl + Carboxyl! Also can catalyze ester formation by group transfer! Reactions! e.g. -

Darwinian Evolution of an Alternative Genetic System Provides Support for TNA As an RNA Progenitor Hanyang Yu†,Suzhang† and John C
ARTICLES PUBLISHED ONLINE: 10 JANUARY 2012 | DOI: 10.1038/NCHEM.1241 Darwinian evolution of an alternative genetic system provides support for TNA as an RNA progenitor Hanyang Yu†,SuZhang† and John C. Chaput* The pre-RNA world hypothesis postulates that RNA was preceded in the evolution of life by a simpler genetic material, but it is not known if such systems can fold into structures capable of eliciting a desired function. Presumably, whatever chemistry gave rise to RNA would have produced other RNA analogues, some of which may have preceded or competed directly with RNA. Threose nucleic acid (TNA), a potentially natural derivative of RNA, has received considerable interest as a possible RNA progenitor due to its chemical simplicity and ability to exchange genetic information with itself and RNA. Here, we have applied Darwinian evolution methods to evolve, in vitro, a TNA receptor that binds to an arbitrary target with high affinity and specificity. This demonstration shows that TNA has the ability to fold into tertiary structures with sophisticated chemical functions, which provides evidence that TNA could have served as an ancestral genetic system during an early stage of life. he question of why nature chose ribofuranosyl nucleic acids as like DNA and RNA, can fold into structures with complicated func- the molecular basis of life’s genetic material has prompted a tions—a prerequisite for any RNA progenitor in a hypothetical Tsystematic analysis of nucleic acid structure with regard to pre-RNA world. the chemical etiology of RNA1. Results from these studies reveal that several potentially natural RNA alternatives are capable of Results and discussion Watson–Crick base pairing, demonstrating the capacity for these The discovery by Eschenmoser and colleagues that TNA can molecules to store genetic information. -

Dna Enzymes for Peptide-Nucleic Acid Conjugation and for Lysine Methylation
DNA ENZYMES FOR PEPTIDE-NUCLEIC ACID CONJUGATION AND FOR LYSINE METHYLATION BY CHIH-CHI CHU DISSERTATION Submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy in Chemistry in the Graduate College of the University of Illinois at Urbana-Champaign, 2017 Urbana, Illinois Doctoral Committee: Professor Scott K. Silverman, Chair Professor Paul J. Hergenrother Associate Professor Douglas A. Mitchell Professor Steven C. Zimmerman Abstract Proteins and RNA are known to be enzymes in nature. These biopolymers have complex secondary and tertiary structures that can enable substrate binding and catalysis. DNA is primarily double-stranded and is not known to be catalytic in nature. However, given the similarities in chemical structure between DNA and RNA, it is reasonable to think that single- stranded DNA can also form complex structures. In fact, artificial DNA enzymes have been identified in laboratories by in vitro selection. The identification of new enzymes favors the use of nucleic acids over proteins for several reasons. First, nucleic acids can be amplified by natural enzymes whereas proteins cannot be amplified in any way. Second, the number of possible sequences is smaller for nucleic acids (4n, where n is the length of the biopolymer) than for proteins (20n). Therefore, selection experiments for identifying nucleic acid enzymes will cover a larger fraction of total sequence space. Furthermore, of the sequence space that is covered, a large portion of nucleic acid sequences can fold into secondary or tertiary structures, whereas most random sequences of proteins often will not fold and thus will aggregate. Between the two nucleic acid polymers, DNA offers additional advantages over RNA because DNA can be directly amplified by polymerases whereas RNA requires an extra reverse transcription step. -

Design, Synthesis, and Characterization of Functional Click Nucleic
DESIGN, SYNTHESIS, AND CHARACTERIZATION OF FUNCTIONAL CLICK NUCLEIC ACID POLYMERS AND CONJUGATES FOR BIOLOGICAL APPLICATIONS By ALEX J. ANDERSON B.S., Vanderbilt University, 2015 M.S., University of Colorado, 2017 A thesis submitted to the Faculty of the Graduate School of the University of Colorado in partial fulfillment of the requirements for the degree of Doctor of Philosophy Department of Chemical and Biological Engineering 2020 Thesis committee: Dr. Christopher N. Bowman, Chair Dr. Stephanie J. Bryant Dr. Virginia L. Ferguson Dr. Jennifer N. Cha Dr. Frank Vernerey Abstract Anderson, Alex J. (PhD. Chemical and Biological Engineering) Design, Synthesis, and Characterization of Functional Click Nucleic Acid Polymers and Conjugates for Biological Applications Thesis directed by Christopher N. Bowman and Stephanie J. Bryant Oligonucleotides are a powerful class of biopolymer which, through Watson-and-Crick hydrogen bonding, are capable of directing the self-assembly of materials as well as recognizing and binding to very specific targets. These features have made oligonucleotides an attractive tool for uses in materials science and biotechnology. However, there are several drawbacks to the implementation of oligonucleotides with a natural backbone (i.e. DNA or RNA) including susceptibility to degradation and a limited scale of production. Click Nucleic Acids (CNAs) represent a new class of oligonucleotide that was designed to alleviate these issues. The distinguishing feature of CNAs is that they are polymerized via radical mediated thiol-ene click chemistry, which vastly increases the speed and scale of oligonucleotide synthesis. This thesis focuses on developing CNA polymers and conjugates and demonstrating the utility of CNAs as an alternative oligonucleotide for various biological applications. -

12.12.2017 1 CHAPTER 1 OLIGONUCLEOTIDES Part 2
12.12.2017 Canonical nucleobase pairing and alternative base pairs CHAPTER 1 OLIGONUCLEOTIDES Part 2 – noncanonical nucleobases Natural and non-natural base pairs that function in polymerase reactions Self-assembly of whole genes and DNA nanostructures The technology tested by assembly of the kanamycin-resistance gene and growing the bacteria in the environment containing kanamycin after assembly and conversion of that gene. S. Benner et al., Beilstein J. Org. Chem. 2014, 10, 2348–2360. doi:10.3762/bjoc.10.245 1 12.12.2017 AEGIS – Artificially Expanded Genetic Information System AEGIS – Artificially Expanded Genetic Information System ZP CG S. Benner et al., Beilstein J. Org. Chem. 2014, 10, 2348–2360. doi:10.3762/bjoc.10.245 S. Benner et al., J. Am. Chem. Soc., 2011 , 133 (38), pp 15105–15112 AEGIS – Permanent orthogonal nucleobases surviving PCR ACGTZP-DNA crystal structures Electron density presented to the minor groove recognition site by polymerases „ minor groove scanning hypothesis” Error rate 0,2% per a PCR cycle – both removal and incorporation of Z and P the artificial genetic system capable to evolve. Pol: Deep Vent – 2 Z/P, Taq/Phu – 3-4 Z/P 18-mers: 2+2 Z:P pairs B-DNA dZTP ( deprotonated ) at higher pH pairs slightly with G 6 consecutive Z:P A-DNA loss of some Z, but also gain of some new Z mutants. 0,1 nm wider, but otherwise alike G:C pairs S. Benner et al., J. Am. Chem. Soc., 2011 , 133 (38), pp 15105–15112 S. Benner et al., J. Am. Chem. Soc., 2015, 137, pp 6947–6955 2 12.12.2017 Unnatural aminoacid incorporation using a noncanonical base pair (A) The coupled transcription–translation system using the nonstandard codon– anticodon interaction for the site-specific incorporation of 3-chlorotyrosine into the Ras protein. -

Beyond Rna and Dna: In-Situ Sequencing of Informational Polymers
International Workshop on Instrumentation for Planetary Missions (2012) 1136.pdf BEYOND RNA AND DNA: IN-SITU SEQUENCING OF INFORMATIONAL POLYMERS. C. E. Carr1-3,*, G. Ruvkun2-3 and M. T. Zuber1. 1Department of Earth, Atmospheric and Planetary Sciences, MIT, Cambridge MA. 2Department of Molecular Biology, Massachusetts General Hospital, Boston, MA. 3Department of Genetics, Har- vard Medical School, Boston, MA. *Correspondence to [email protected]. Dust, Introduction: Nucleic acid sequencing provides a Comets, Earth GNA/TNA?? RNA DNA powerful approach to search for life beyond Earth, as Meteors Meteoritic Exchange well as to monitor levels of forward contamination [1, Mars Common ancestor / 2nd genesis / no life 2]. A strong case can be made to look for RNA or Sun Enceladeus 2nd genesis / no life DNA-based life on Mars [3, 4]: any life there could Europa 2nd genesis / no life Complex organics Others 2nd genesis / no life have a common ancestor with life on Earth due to ex- produced around all stars Delivered to Life evolved one or tensive meteoritic transfer between our two planets [5- multiple potentially more times 9] and the potential for transfer of viable microbes. habitable zones Here we argue that in-situ sequencing has a role in Figure 1. Delivery of similar organic material to mul- life detection even beyond the context of meteoritic tiple habitable zones: if life arose beyond Earth, what transfer, such as searching for life in potentially habit- informational polymers would it use? able environments on Enceladus [10, 11], or Europa [12]. This is because 1) sequencing of non standard nucleic acids is now possible, and 2) it may also be possible to sequence even more generic informational As a result, similar organic material may be delivered polymers (IPs). -

DNA Before Proteins? Recent Discoveries in Nucleic Acid Catalysis Strengthen the Case
Portland State University PDXScholar Chemistry Faculty Publications and Presentations Chemistry 1-2009 DNA Before Proteins? Recent Discoveries in Nucleic Acid Catalysis Strengthen the Case Aaron Steven Burton Portland State University Niles Lehman Portland State University Follow this and additional works at: https://pdxscholar.library.pdx.edu/chem_fac Part of the Biology Commons, and the Genetics Commons Let us know how access to this document benefits ou.y Citation Details Burton, A.S.; Lehman, N. (2009). DNA before proteins? Recent discoveries in nucleic acid catalysis strengthen the case. Astrobiology 9:125-130. This Article is brought to you for free and open access. It has been accepted for inclusion in Chemistry Faculty Publications and Presentations by an authorized administrator of PDXScholar. Please contact us if we can make this document more accessible: [email protected]. ASTROBIOLOGY Volume 9, Number 1, 2009 © Mary Ann Liebert, Inc. DOI: 10.1089/ast.2008.0240 Hypothesis Paper DNA Before Proteins? Recent Discoveries in Nucleic Acid Catalysis Strengthen the Case Aaron S. Burton and Niles Lehman Abstract An RNA-DNA World could arise from an all-RNA system with the development of as few as three ribozymes— a DNA-dependent RNA polymerase, an RNA-dependent DNA polymerase, and a catalyst for the production of DNA nucleotides. A significant objection to DNA preceding proteins is that RNA has not been shown to cat- alyze the production of DNA. However, RNA- and DNAzymes have been recently discovered that catalyze chemical reactions capable of forming deoxyribose, such as mixed aldol condensation of 5’-glyceryl- and 3’- glycoaldehyde-terminated DNA strands. -

Theranostics Molecular Beacons of Xeno-Nucleic Acid for Detecting
Theranostics 2013, Vol. 3, Issue 6 395 Ivyspring International Publisher Theranostics 2013; 3(6):395-408. doi: 10.7150/thno.5935 Review Molecular Beacons of Xeno-Nucleic Acid for Detecting Nucleic Acid Qi Wang2, Lei Chen1, Yitao Long1, He Tian1 and Junchen Wu1, 1. Key Lab for Advanced Materials and Institute of Fine Chemicals, East China University of Science and Technology, China. 2. College of Public Health, Nantong University, China. Corresponding author: Junchen Wu, Meilong 130, Shanghai 200237, China. Telephone: (+86) 21-6425-3674, fax: (+86) 21-6425-2258, E-mail: [email protected]. © Ivyspring International Publisher. This is an open-access article distributed under the terms of the Creative Commons License (http://creativecommons.org/ licenses/by-nc-nd/3.0/). Reproduction is permitted for personal, noncommercial use, provided that the article is in whole, unmodified, and properly cited. Received: 2013.01.23; Accepted: 2013.04.10; Published: 2013.05.05 Abstract Molecular beacons (MBs) of DNA and RNA have aroused increasing interest because they allow a continuous readout, excellent spatial and temporal resolution to observe in real time. This kind of dual-labeled oligonucleotide probes can differentiate between bound and unbound DNA/RNA in homogenous hybridization with a high signal-to-background ratio in living cells. This review briefly summarizes the different unnatural sugar backbones of oligonucleotides combined with fluoro- phores that have been employed to sense DNA/RNA. With different probes, we epitomize the fundamental understanding of driving forces and these recognition processes. Moreover, we will introduce a few novel and attractive emerging applications and discuss their advantages and dis- advantages. -

Modeling Prebiotic Catalysis with Nucleic Acid-Like Polymers and Its Implications for the Proposed RNA World*
Pure Appl. Chem., Vol. 76, No. 12, pp. 2085–2099, 2004. © 2004 IUPAC Modeling prebiotic catalysis with nucleic acid-like polymers and its implications for the proposed RNA world* S. G. Srivatsan‡ Department of Chemistry, Indian Institute of Technology-Kanpur, Kanpur-208016 (UP), India Abstract: The theory that RNA molecules played a pivotal role in the early evolution of life is now widely accepted. Studies related to this hypothetical “RNA world” include three major areas: the formation of precursors for the first RNA molecules, the polymerization process, and the potential of RNA to catalyze chemical and biochemical reactions. Several chemical and biochemical studies performed under simulated prebiotic conditions support the role of RNA as both genetic as well as catalytic material. However, owing to the lack of credible mechanism for de novo nucleic acid synthesis and the hydrolytic instability of RNA mole- cules, there has been some serious discussion of whether biopolymers that closely resembled nucleic acid preceded the “RNA world”. In this context, an overview of prebiotic chemistry, the role of mineral surface, and the significance of studies related to RNA-like polymers in the origin of life are presented here. INTRODUCTION Though life that exists today and in the geological record is based on DNA genomes and protein en- zymes, there is convincing evidence that hypothesizes the origin of life based on RNA [1]. This hypo- thetical primordial era is referred to as the “RNA world” [2]. The “RNA world” hypothesis assumes that all modern organisms are descendants of a prebiotic self-replicating moiety, which utilized RNA as both genetic as well as catalytic material. -
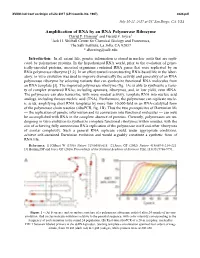
Amplification of RNA by an RNA Polymerase Ribozyme David P
XVIIIth Intl Conf on Origin of Life 2017 (LPI Contrib. No. 1967) 4024.pdf July 16-21, 2017 at UC San Diego, CA, USA Amplification of RNA by an RNA Polymerase Ribozyme David P. Horning1 and Gerald F. Joyce1 1Jack H. Skirball Center for Chemical Biology and Proteomics, The Salk Institute, La Jolla, CA 92037 * [email protected] Introduction: In all extant life, genetic information is stored in nucleic acids that are repli- cated by polymerase proteins. In the hypothesized RNA world, prior to the evolution of genet- ically-encoded proteins, ancestral organisms contained RNA genes that were replicated by an RNA polymerase ribozyme [1,2]. In an effort toward reconstructing RNA-based life in the labor- atory, in vitro evolution was used to improve dramatically the activity and generality of an RNA polymerase ribozyme by selecting variants that can synthesize functional RNA molecules from an RNA template [3]. The improved polymerase ribozyme (fig. 1A) is able to synthesize a varie- ty of complex structured RNAs, including aptamers, ribozymes, and, in low yield, even tRNA. The polymerase can also transcribe, with more modest activity, template RNA into nucleic acid analogs, including threose nucleic acid (TNA). Furthermore, the polymerase can replicate nucle- ic acids, amplifying short RNA templates by more than 10,000-fold in an RNA-catalyzed form of the polymerase chain reaction (riboPCR, fig. 1B). Thus the two prerequisites of Darwinian life — the replication of genetic information and its conversion into functional molecules — can now be accomplished with RNA in the complete absence of proteins. Currently, polymerases are un- dergoing in vitro evolution to synthesize complete functional ribozymes within minutes, with the aim of achieving fully autonomous RNA replication of the polymerase itself and other ribozymes of similar complexity.