2013 Thesis Small Mammals
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Special Publications Museum of Texas Tech University Number 63 18 September 2014
Special Publications Museum of Texas Tech University Number 63 18 September 2014 List of Recent Land Mammals of Mexico, 2014 José Ramírez-Pulido, Noé González-Ruiz, Alfred L. Gardner, and Joaquín Arroyo-Cabrales.0 Front cover: Image of the cover of Nova Plantarvm, Animalivm et Mineralivm Mexicanorvm Historia, by Francisci Hernández et al. (1651), which included the first list of the mammals found in Mexico. Cover image courtesy of the John Carter Brown Library at Brown University. SPECIAL PUBLICATIONS Museum of Texas Tech University Number 63 List of Recent Land Mammals of Mexico, 2014 JOSÉ RAMÍREZ-PULIDO, NOÉ GONZÁLEZ-RUIZ, ALFRED L. GARDNER, AND JOAQUÍN ARROYO-CABRALES Layout and Design: Lisa Bradley Cover Design: Image courtesy of the John Carter Brown Library at Brown University Production Editor: Lisa Bradley Copyright 2014, Museum of Texas Tech University This publication is available free of charge in PDF format from the website of the Natural Sciences Research Laboratory, Museum of Texas Tech University (nsrl.ttu.edu). The authors and the Museum of Texas Tech University hereby grant permission to interested parties to download or print this publication for personal or educational (not for profit) use. Re-publication of any part of this paper in other works is not permitted without prior written permission of the Museum of Texas Tech University. This book was set in Times New Roman and printed on acid-free paper that meets the guidelines for per- manence and durability of the Committee on Production Guidelines for Book Longevity of the Council on Library Resources. Printed: 18 September 2014 Library of Congress Cataloging-in-Publication Data Special Publications of the Museum of Texas Tech University, Number 63 Series Editor: Robert J. -

Virginia Journal of Science Official Publication of the Virginia Academy of Science
VIRGINIA JOURNAL OF SCIENCE OFFICIAL PUBLICATION OF THE VIRGINIA ACADEMY OF SCIENCE Vol. 62 No. 3 Fall 2011 TABLE OF CONTENTS ARTICLES PAGE Breeding Biology of Oryzomys Palustris, the Marsh Rice Rat, in Eastern Virginia. Robert K. Rose and Erin A. Dreelin. 113 Abstracts missing from Volume 62 Number 1 & 2 123 Academy Minutes 127 The Horsley Award paper for 2011 135 Virginia Journal of Science Volume 62, Number 3 Fall 2011 Breeding Biology of Oryzomys Palustris, the Marsh Rice Rat, in Eastern Virginia Robert K. Rose1 and Erin A. Dreelin2, Department of Biological Sciences, Old Dominion University, Norfolk, Virginia 23529-0266 ABSTRACT The objectives of our study were to determine the age of maturity, litter size, and the timing of the breeding season of marsh rice rats (Oryzomys palustris) of coastal Virginia. From May 1995 to May 1996, monthly samples of rice rats were live-trapped in two coastal tidal marshes of eastern Virginia, and then necropsied. Sexual maturity was attained at 30-40 g for both sexes. Mean litter size of 4.63 (n = 16) did not differ among months or in mass or parity classes. Data from two other studies conducted in the same county, one of them contemporaneous, also were examined. Based on necropsy, rice rats bred from March to October; breeding did not occur in December-February. By contrast, rice rats observed during monthly trapping on nearby live-trap grids were judged, using external indicators, to be breeding year-round except January. Compared to internal examinations, external indicators of reproductive condition were not reliable for either sex in predicting breeding status in the marsh rice rat. -

March Rice Rat, <I>Oryzomys Palustris</I>
University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Mammalogy Papers: University of Nebraska State Museum, University of Nebraska State Museum 1-25-1985 March Rice Rat, Oryzomys palustris Hugh H. Genoways University of Nebraska - Lincoln, [email protected] Follow this and additional works at: http://digitalcommons.unl.edu/museummammalogy Part of the Biodiversity Commons, Terrestrial and Aquatic Ecology Commons, and the Zoology Commons Genoways, Hugh H., "March Rice Rat, Oryzomys palustris" (1985). Mammalogy Papers: University of Nebraska State Museum. 227. http://digitalcommons.unl.edu/museummammalogy/227 This Article is brought to you for free and open access by the Museum, University of Nebraska State at DigitalCommons@University of Nebraska - Lincoln. It has been accepted for inclusion in Mammalogy Papers: University of Nebraska State Museum by an authorized administrator of DigitalCommons@University of Nebraska - Lincoln. Genoways in Species of Special Concern in Pennsylvania (Genoways & Brenner, editors). Special Publication, Carnegie Museum of Natural History (1985) no. 11. Copyright 1985, Carnegie Museum of Natural History. Used by permission. 402 SPECIAL PUBLICATION CARNEGIE MUSEUM OF NATURAL HISTORY NO. 11 "'-" "~_MARSH RICE RAT (Oryzomys pa!ustris) Status Undetermined MARSH RICE RAT Oryzomys palustris Family Cricetidae Order Rodentia River Valley and in the areas surrounding its prin OTHER NAMES: Rice rat, swamp rice rat, north cipal tributaries (Hall, 1981). ern rice rat. HABITAT: The marsh rice rat is a semi-aquatic DESCRIPTION: A medium-sized rat that would species that is found in greatest abundance in the be most easily confused with smaller individuals of marshes and swamps and other wetlands ofthe Gulf the introduced Norway rat (Rattus norvegicus). -

B a N I S T E R I A
B A N I S T E R I A A JOURNAL DEVOTED TO THE NATURAL HISTORY OF VIRGINIA ISSN 1066-0712 Published by the Virginia Natural History Society The Virginia Natural History Society (VNHS) is a nonprofit organization dedicated to the dissemination of scientific information on all aspects of natural history in the Commonwealth of Virginia, including botany, zoology, ecology, archaeology, anthropology, paleontology, geology, geography, and climatology. The society’s periodical Banisteria is a peer-reviewed, open access, online-only journal. Submitted manuscripts are published individually immediately after acceptance. A single volume is compiled at the end of each year and published online. The Editor will consider manuscripts on any aspect of natural history in Virginia or neighboring states if the information concerns a species native to Virginia or if the topic is directly related to regional natural history (as defined above). Biographies and historical accounts of relevance to natural history in Virginia also are suitable for publication in Banisteria. Membership dues and inquiries about back issues should be directed to the Co-Treasurers, and correspondence regarding Banisteria to the Editor. For additional information regarding the VNHS, including other membership categories, annual meetings, field events, pdf copies of papers from past issues of Banisteria, and instructions for prospective authors visit http://virginianaturalhistorysociety.com/ Editorial Staff: Banisteria Editor Todd Fredericksen, Ferrum College 215 Ferrum Mountain Road Ferrum, Virginia 24088 Associate Editors Philip Coulling, Nature Camp Incorporated Clyde Kessler, Virginia Tech Nancy Moncrief, Virginia Museum of Natural History Karen Powers, Radford University Stephen Powers, Roanoke College C. L. Staines, Smithsonian Environmental Research Center Copy Editor Kal Ivanov, Virginia Museum of Natural History Copyright held by the author(s). -

Population Genetics of the Native Rodents of the Galápagos Islands, Ecuador
Population Genetics of the Native Rodents of the Galápagos Islands, Ecuador A dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy at George Mason University By Sarah Johnson Master of Science Stephen F. Austin State University, 2005 Bachelor of Science Texas A&M University, 2003 Director: Dr. Cody W. Edwards, Assistant Professor Department of Environmental Science and Public Policy Summer Semester 2009 George Mason University Fairfax, VA Copyright 2009 Sarah Johnson All Rights Reserved ii ACKNOWLEDGMENTS I would like to thank my parents (Michael and Kay Johnson) and my sisters (Kris and Faith) for their unwavering support throughout my academic career. This dissertation is lovingly dedicated to my parents. I would like to thank my Aggie Family (Brad and Kristin Atchison, Reece and Erin Flood, Samir Moussa, Doug Fuentes, and the rest of the IV Horsemen). They have always lovingly provided a shoulder to lean on and kind ear willing to listen. I would like to thank my fellow graduate students at GMU (Jeff Streicher, Mike Jarcho, Kat Bryant, Tammy Henry, Geoff Cook, Ryan Peters, Kristin Wolf, Trishna Dutta, Sandeep Sharma, and Jolanda Luksenburg) for their help in the field, lab, classroom, and all aspects of student life. I am eternally indebted to Dr. Pat Gillevet and Masi Sikaroodi for their invaluable assistance in the lab, and to Dr. Jesús Maldonado for his assistance in writing the dissertation. They are infinite sources of help and support for which I am forever grateful. The project would not have been possible without Dr. Cody W. Edwards and Dr. -

List of 28 Orders, 129 Families, 598 Genera and 1121 Species in Mammal Images Library 31 December 2013
What the American Society of Mammalogists has in the images library LIST OF 28 ORDERS, 129 FAMILIES, 598 GENERA AND 1121 SPECIES IN MAMMAL IMAGES LIBRARY 31 DECEMBER 2013 AFROSORICIDA (5 genera, 5 species) – golden moles and tenrecs CHRYSOCHLORIDAE - golden moles Chrysospalax villosus - Rough-haired Golden Mole TENRECIDAE - tenrecs 1. Echinops telfairi - Lesser Hedgehog Tenrec 2. Hemicentetes semispinosus – Lowland Streaked Tenrec 3. Microgale dobsoni - Dobson’s Shrew Tenrec 4. Tenrec ecaudatus – Tailless Tenrec ARTIODACTYLA (83 genera, 142 species) – paraxonic (mostly even-toed) ungulates ANTILOCAPRIDAE - pronghorns Antilocapra americana - Pronghorn BOVIDAE (46 genera) - cattle, sheep, goats, and antelopes 1. Addax nasomaculatus - Addax 2. Aepyceros melampus - Impala 3. Alcelaphus buselaphus - Hartebeest 4. Alcelaphus caama – Red Hartebeest 5. Ammotragus lervia - Barbary Sheep 6. Antidorcas marsupialis - Springbok 7. Antilope cervicapra – Blackbuck 8. Beatragus hunter – Hunter’s Hartebeest 9. Bison bison - American Bison 10. Bison bonasus - European Bison 11. Bos frontalis - Gaur 12. Bos javanicus - Banteng 13. Bos taurus -Auroch 14. Boselaphus tragocamelus - Nilgai 15. Bubalus bubalis - Water Buffalo 16. Bubalus depressicornis - Anoa 17. Bubalus quarlesi - Mountain Anoa 18. Budorcas taxicolor - Takin 19. Capra caucasica - Tur 20. Capra falconeri - Markhor 21. Capra hircus - Goat 22. Capra nubiana – Nubian Ibex 23. Capra pyrenaica – Spanish Ibex 24. Capricornis crispus – Japanese Serow 25. Cephalophus jentinki - Jentink's Duiker 26. Cephalophus natalensis – Red Duiker 1 What the American Society of Mammalogists has in the images library 27. Cephalophus niger – Black Duiker 28. Cephalophus rufilatus – Red-flanked Duiker 29. Cephalophus silvicultor - Yellow-backed Duiker 30. Cephalophus zebra - Zebra Duiker 31. Connochaetes gnou - Black Wildebeest 32. Connochaetes taurinus - Blue Wildebeest 33. Damaliscus korrigum – Topi 34. -

Marsh Rice Rat Oryzomys Palustris ILLINOIS RANGE Adult
marsh rice rat Oryzomys palustris Kingdom: Animalia FEATURES Phylum: Chordata The marsh rice rat is about four and three-fourths Class: Mammalia inches in head-body length with a tail that is about Order: Rodentia as long as or longer than the head-body length. It has a slender body with gray-brown fur and white Family: Cricetidae feet. The belly fur is gray. The tail is gray-brown ILLINOIS STATUS above and gray below. The ears are hairy. common, native BEHAVIORS The marsh rice rat may be found in the southern one-fourth of Illinois living in marshes and swamps. Its diet usually includes seeds and leaves of grasses and aquatic plants, but it may also eat turtles, fishes, bird eggs, small mammals, insects and snails. This rodent is mainly nocturnal and is active all year. It is a good swimmer. While on land, it makes and uses runways. Mating occurs throughout the spring and summer. Young are raised in a nest of grasses and vines that is placed above flood level. Litter size varies from three to five. Young marsh rice rats reach maturity in about two months. adult ILLINOIS RANGE © Illinois Department of Natural Resources. 2021. Biodiversity of Illinois. Unless otherwise noted, photos and images © Illinois Department of Natural Resources. Aquatic Habitats marshes; swamps Woodland Habitats none Prairie and Edge Habitats none © Illinois Department of Natural Resources. 2021. Biodiversity of Illinois. Unless otherwise noted, photos and images © Illinois Department of Natural Resources.. -

Redalyc.RIQUEZA, ENDEMISMO Y CONSERVACIÓN DE LOS
Mastozoología Neotropical ISSN: 0327-9383 [email protected] Sociedad Argentina para el Estudio de los Mamíferos Argentina Solari, Sergio; Muñoz-Saba, Yaneth; Rodríguez-Mahecha, José V.; Defler, Thomas R.; Ramírez- Chaves, Héctor E.; Trujillo, Fernando RIQUEZA, ENDEMISMO Y CONSERVACIÓN DE LOS MAMÍFEROS DE COLOMBIA Mastozoología Neotropical, vol. 20, núm. 2, julio-diciembre, 2013, pp. 301-365 Sociedad Argentina para el Estudio de los Mamíferos Tucumán, Argentina Disponible en: http://www.redalyc.org/articulo.oa?id=45729294008 Cómo citar el artículo Número completo Sistema de Información Científica Más información del artículo Red de Revistas Científicas de América Latina, el Caribe, España y Portugal Página de la revista en redalyc.org Proyecto académico sin fines de lucro, desarrollado bajo la iniciativa de acceso abierto Mastozoología Neotropical, 20(2):301-365, Mendoza, 2013 Copyright ©SAREM, 2013 Versión impresa ISSN 0327-9383 http://www.sarem.org.ar Versión on-line ISSN 1666-0536 Artículo RIQUEZA, ENDEMISMO Y CONSERVACIÓN DE LOS MAMÍFEROS DE COLOMBIA Sergio Solari1, Yaneth Muñoz-Saba2, José V. Rodríguez-Mahecha3, Thomas R. Defler4, Héctor E. Ramírez-Chaves5 y Fernando Trujillo6 1 Instituto de Biología, Grupo Mastozoología & Colección Teriológica, Universidad de Antioquia, Medellín, Colombia. [Correspondencia: <[email protected]>]. 2 Instituto de Ciencias Naturales, Universidad Nacional de Colombia, Bogotá D.C., Colombia. 3 Director Científico, Conservación Internacional Colombia, Bogotá D.C., Colombia. 4 Departamento de Biología, Universidad Nacional de Colombia, Bogotá, D.C., Colombia. 5 School of Biological Sciences, University of Queensland, Queensland, Australia. 6 Director Científico, Fundación Omacha, Bogotá D.C., Colombia. RESUMEN. Se actualiza la diversidad de especies de mamíferos de Colombia con base en una nueva revisión de especímenes en las mayores colecciones del país y el extranjero y la compilación de cambios taxonómicos recientes que involucran especies presentes en el país. -

Population Genetics of Rice Rats (Oryzomys Palustris) at the Northern Edge of the Species Range
Southern Illinois University Carbondale OpenSIUC Theses Theses and Dissertations 8-1-2019 Population Genetics of Rice Rats (Oryzomys palustris) at the Northern Edge of the Species Range Phillip Conrad Williams Southern Illinois University Carbondale, [email protected] Follow this and additional works at: https://opensiuc.lib.siu.edu/theses Recommended Citation Williams, Phillip Conrad, "Population Genetics of Rice Rats (Oryzomys palustris) at the Northern Edge of the Species Range" (2019). Theses. 2602. https://opensiuc.lib.siu.edu/theses/2602 This Open Access Thesis is brought to you for free and open access by the Theses and Dissertations at OpenSIUC. It has been accepted for inclusion in Theses by an authorized administrator of OpenSIUC. For more information, please contact [email protected]. POPULATION GENETICS OF RICE RATS (ORYZOMYS PALUSTRIS) AT THE NORTHERN EDGE OF THE SPECIES RANGE by Conrad Williams B.S. University of Texas at Austin, 2011 A Thesis Submitted in Partial Fulfillment of the Requirements for the Master of Science Degree Department of Zoology in the Graduate School Southern Illinois University Carbondale August 2019 THESIS APPROVAL POPULATION GENETICS OF RICE RATS (ORYZOMYS PALUSTRIS) AT THE NORTHERN EDGE OF THE SPECIES RANGE by Conrad Williams A Thesis Submitted in Partial Fulfillment of the Requirements For the Degree of Master of Science in the Field of Zoology Approved by: Dr. Kamal M. Ibrahim, Chair Dr. Edward Heist Dr. Carey Krajewski Graduate School Southern Illinois University Carbondale July 3, 2019 AN ABSTRACT OF THE THESIS OF Conrad Williams, for the Master of Science degree in ZOOLOGY, presented on July 3, 2019, at Southern Illinois University Carbondale. -

List of Taxa for Which MIL Has Images
LIST OF 27 ORDERS, 163 FAMILIES, 887 GENERA, AND 2064 SPECIES IN MAMMAL IMAGES LIBRARY 31 JULY 2021 AFROSORICIDA (9 genera, 12 species) CHRYSOCHLORIDAE - golden moles 1. Amblysomus hottentotus - Hottentot Golden Mole 2. Chrysospalax villosus - Rough-haired Golden Mole 3. Eremitalpa granti - Grant’s Golden Mole TENRECIDAE - tenrecs 1. Echinops telfairi - Lesser Hedgehog Tenrec 2. Hemicentetes semispinosus - Lowland Streaked Tenrec 3. Microgale cf. longicaudata - Lesser Long-tailed Shrew Tenrec 4. Microgale cowani - Cowan’s Shrew Tenrec 5. Microgale mergulus - Web-footed Tenrec 6. Nesogale cf. talazaci - Talazac’s Shrew Tenrec 7. Nesogale dobsoni - Dobson’s Shrew Tenrec 8. Setifer setosus - Greater Hedgehog Tenrec 9. Tenrec ecaudatus - Tailless Tenrec ARTIODACTYLA (127 genera, 308 species) ANTILOCAPRIDAE - pronghorns Antilocapra americana - Pronghorn BALAENIDAE - bowheads and right whales 1. Balaena mysticetus – Bowhead Whale 2. Eubalaena australis - Southern Right Whale 3. Eubalaena glacialis – North Atlantic Right Whale 4. Eubalaena japonica - North Pacific Right Whale BALAENOPTERIDAE -rorqual whales 1. Balaenoptera acutorostrata – Common Minke Whale 2. Balaenoptera borealis - Sei Whale 3. Balaenoptera brydei – Bryde’s Whale 4. Balaenoptera musculus - Blue Whale 5. Balaenoptera physalus - Fin Whale 6. Balaenoptera ricei - Rice’s Whale 7. Eschrichtius robustus - Gray Whale 8. Megaptera novaeangliae - Humpback Whale BOVIDAE (54 genera) - cattle, sheep, goats, and antelopes 1. Addax nasomaculatus - Addax 2. Aepyceros melampus - Common Impala 3. Aepyceros petersi - Black-faced Impala 4. Alcelaphus caama - Red Hartebeest 5. Alcelaphus cokii - Kongoni (Coke’s Hartebeest) 6. Alcelaphus lelwel - Lelwel Hartebeest 7. Alcelaphus swaynei - Swayne’s Hartebeest 8. Ammelaphus australis - Southern Lesser Kudu 9. Ammelaphus imberbis - Northern Lesser Kudu 10. Ammodorcas clarkei - Dibatag 11. Ammotragus lervia - Aoudad (Barbary Sheep) 12. -

Datos Personales Áreas De Actuación Formación Académica/Titulación
Datos Personales Nombre Robert Dale Owen Nombre en citaciones bibliográficas Robert D. Owen Áreas de actuación 1 Ciencias Naturales/Ciencias Biológicas/Biología y Biología de la Evolución/Mastozoologia, Biogeografia, Sistematica, Ecologia Formación académica/Titulación (concluida) 1977-1987 Doctorado - Department of Zoology University of Oklahoma, Estados Unidos Título: Multivariate morphometric analyses of the bat subfamily Stenodermatinae (Chiroptera: Phyllostomidae) Año de obtención: 1987 Tutor: Gary D. Schnell Becario de: National Science Foundation, Estados Unidos Palabras Clave: Murcielagos; Sistematica Áreas del conocimiento: Ciencias Naturales/Ciencias Biológicas/Biología y Biología de la Evolución/Mastozoologia, Biogeografia, Sistematica, Ecologia. 1966-1976 Grado - Department of Zoology University of Oklahoma, Estados Unidos Año de obtención: 1976 Becario de: Sigma Xi, Estados Unidos Palabras Clave: Zoologia Áreas del conocimiento: Ciencias Naturales/Ciencias Biológicas/Zoología, Ornitología, Entomología, Etología. Idiomas Entiende Inglés(Muy bien) Español(Bien) Francés(Regular) Portugués(Regular) Habla Inglés(Muy bien) Español(Bien) Lee Inglés(Muy bien) Español(Bien) Francés(Regular) Portugués(Bien) Escribe Inglés(Muy bien) Español(Bien) Actuación profesional Proyecto "Mamiferos del Paraguay" - PMP Vínculos con la institución 1995 - Actual Vínculo: Otro. Encuadramiento funcional: Investigador Principal. Carga horaria: 25. Otras informaciones Conseguir becas de investigacion; dirigir estudiantes en sus investigaciones; desarrollar lineas de investigacion en biologia, ecologia, y sistematica de mamiferos y otros taxa vertebrados. Actividades 03/2010 - Actual Líneas de Investigación Líneas de investigación 1. Investigaciones de la Estacion Biologica "Para La Tierra", Reserva Natural Laguna Blanca, Depto. San Pedro. 01/1995 - Actual Líneas de Investigación Líneas de investigación 1. Mamiferos del Paraguay. 01/2004 - 02/2008 Líneas de Investigación Líneas de investigación 1. Ecologia del Hantavirus en Paraguay. -
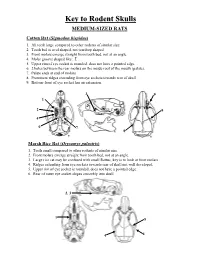
Key to Rodent Skulls MEDIUM-SIZED RATS Cotton Rat (Sigmodon Hispidus) 1
Key to Rodent Skulls MEDIUM-SIZED RATS Cotton Rat (Sigmodon hispidus) 1. All teeth large compared to other rodents of similar size. 2. Tooth bed is oval shaped, not teardrop shaped. 3. Front molars emerge straight from tooth bed, not at an angle. 4. Molar groove shaped like: Σ 5. Upper rim of eye socket is rounded, does not have a pointed edge. 6. 2 holes between the rear molars on the inside roof of the mouth (palate). 7. Palate ends at end of molars. 8. Prominent ridges extending from eye sockets towards rear of skull. 9. Bottom-front of eye socket has an extension. 3 9 1 2 5 4 6 7 8 Marsh Rice Rat (Orysomys palustris) 1. Teeth small compared to other rodents of similar size. 2. Front molars emerge straight from tooth bed, not at an angle. 3. Large rice rat may be confused with small Rattus, key is to look at front molars 4. Ridges extending from eye sockets towards rear of skull not well developed. 5. Upper rim of eye socket is rounded, does not have a pointed edge. 6. Rear of outer eye socket slopes smoothly into skull. 2, 3 1 5 4 6 LARGE-SIZED RATS Roof Rat (Rattus rattus) • Skull longer and pointer than cotton rat or rice rat. • Skull generally longer than 25 mm. 3. Front molars (and their tooth beds) strongly angled up towards front of skull. 4. Front molars large. 5. Tooth bed teardrop shaped, pointed towards rear. 6. Upper rim of eye sockets have a slight point into the sockets.