Smallmouth Bass (Micropterus Dolomieu) ERSS
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

§4-71-6.5 LIST of CONDITIONALLY APPROVED ANIMALS November
§4-71-6.5 LIST OF CONDITIONALLY APPROVED ANIMALS November 28, 2006 SCIENTIFIC NAME COMMON NAME INVERTEBRATES PHYLUM Annelida CLASS Oligochaeta ORDER Plesiopora FAMILY Tubificidae Tubifex (all species in genus) worm, tubifex PHYLUM Arthropoda CLASS Crustacea ORDER Anostraca FAMILY Artemiidae Artemia (all species in genus) shrimp, brine ORDER Cladocera FAMILY Daphnidae Daphnia (all species in genus) flea, water ORDER Decapoda FAMILY Atelecyclidae Erimacrus isenbeckii crab, horsehair FAMILY Cancridae Cancer antennarius crab, California rock Cancer anthonyi crab, yellowstone Cancer borealis crab, Jonah Cancer magister crab, dungeness Cancer productus crab, rock (red) FAMILY Geryonidae Geryon affinis crab, golden FAMILY Lithodidae Paralithodes camtschatica crab, Alaskan king FAMILY Majidae Chionocetes bairdi crab, snow Chionocetes opilio crab, snow 1 CONDITIONAL ANIMAL LIST §4-71-6.5 SCIENTIFIC NAME COMMON NAME Chionocetes tanneri crab, snow FAMILY Nephropidae Homarus (all species in genus) lobster, true FAMILY Palaemonidae Macrobrachium lar shrimp, freshwater Macrobrachium rosenbergi prawn, giant long-legged FAMILY Palinuridae Jasus (all species in genus) crayfish, saltwater; lobster Panulirus argus lobster, Atlantic spiny Panulirus longipes femoristriga crayfish, saltwater Panulirus pencillatus lobster, spiny FAMILY Portunidae Callinectes sapidus crab, blue Scylla serrata crab, Samoan; serrate, swimming FAMILY Raninidae Ranina ranina crab, spanner; red frog, Hawaiian CLASS Insecta ORDER Coleoptera FAMILY Tenebrionidae Tenebrio molitor mealworm, -

2008 Annual Report
Document ID #P113359 Report covers work performed under BPA contract #26763 Report was completed under BPA contract #34772 REPORT ON THE PREDATION INDEX, PREDATOR CONTROL FISHERIES, AND PROGRAM EVALUATION FOR THE COLUMBIA RIVER BASIN EXPERIMENTAL NORTHERN PIKEMINNOW MANAGEMENT PROGRAM 2008 ANNUAL REPORT Prepared by: Russell Porter Pacific States Marine Fisheries Commission In Cooperation with: Oregon Department of Fish and Wildlife Washington Department of Fish and Wildlife U.S. Department of Agriculture Prepared for: U.S. Department of Energy Bonneville Power Administration Environment, Fish and Wildlife P.O. Box 3621 Portland, OR 97208-3621 Project Number 199007700 Contract Number 00026763 Table of Contents Executive Summary 5 Report A – Sport Reward Fishery in the Columbia and Snake Rivers 10 Abstract 11 Introduction 12 Methods of Operation 13 Fishery Operation 13 Boundaries and Season 13 Registration Stations 14 Reward System 14 Angler Sampling 15 Returning Anglers 16 Non-Returning Anglers 16 Northern Pikeminnow Handling Procedures 17 Biological Sampling 17 PIT Tag Detection 17 Northern Pikeminnow Processing 18 Results and Discussion 18 Northern Pikeminnow Harvest 18 Harvest by Week 19 Harvest by Fishing Location 20 Harvest by Registration Station 21 Harvest by Species/Incidental Catch 22 Angler Effort 24 Effort by Week 25 Effort by Fishing Location 27 Effort by Registration Station 27 Catch Per Angler Day (CPUE) 28 CPUE by Week 29 CPUE by Fishing Location 30 CPUE by Registration Station 30 Angler Totals 31 Tag Recovery 34 Northern -
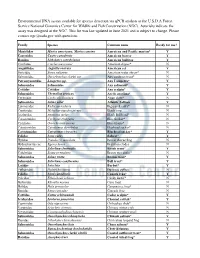
Edna Assay Development
Environmental DNA assays available for species detection via qPCR analysis at the U.S.D.A Forest Service National Genomics Center for Wildlife and Fish Conservation (NGC). Asterisks indicate the assay was designed at the NGC. This list was last updated in June 2021 and is subject to change. Please contact [email protected] with questions. Family Species Common name Ready for use? Mustelidae Martes americana, Martes caurina American and Pacific marten* Y Castoridae Castor canadensis American beaver Y Ranidae Lithobates catesbeianus American bullfrog Y Cinclidae Cinclus mexicanus American dipper* N Anguillidae Anguilla rostrata American eel Y Soricidae Sorex palustris American water shrew* N Salmonidae Oncorhynchus clarkii ssp Any cutthroat trout* N Petromyzontidae Lampetra spp. Any Lampetra* Y Salmonidae Salmonidae Any salmonid* Y Cottidae Cottidae Any sculpin* Y Salmonidae Thymallus arcticus Arctic grayling* Y Cyrenidae Corbicula fluminea Asian clam* N Salmonidae Salmo salar Atlantic Salmon Y Lymnaeidae Radix auricularia Big-eared radix* N Cyprinidae Mylopharyngodon piceus Black carp N Ictaluridae Ameiurus melas Black Bullhead* N Catostomidae Cycleptus elongatus Blue Sucker* N Cichlidae Oreochromis aureus Blue tilapia* N Catostomidae Catostomus discobolus Bluehead sucker* N Catostomidae Catostomus virescens Bluehead sucker* Y Felidae Lynx rufus Bobcat* Y Hylidae Pseudocris maculata Boreal chorus frog N Hydrocharitaceae Egeria densa Brazilian elodea N Salmonidae Salvelinus fontinalis Brook trout* Y Colubridae Boiga irregularis Brown tree snake* -

And Wildlife, 1928-72
Bibliography of Research Publications of the U.S. Bureau of Sport Fisheries and Wildlife, 1928-72 UNITED STATES DEPARTMENT OF THE INTERIOR BUREAU OF SPORT FISHERIES AND WILDLIFE RESOURCE PUBLICATION 120 BIBLIOGRAPHY OF RESEARCH PUBLICATIONS OF THE U.S. BUREAU OF SPORT FISHERIES AND WILDLIFE, 1928-72 Edited by Paul H. Eschmeyer, Division of Fishery Research Van T. Harris, Division of Wildlife Research Resource Publication 120 Published by the Bureau of Sport Fisheries and Wildlife Washington, B.C. 1974 Library of Congress Cataloging in Publication Data Eschmeyer, Paul Henry, 1916 Bibliography of research publications of the U.S. Bureau of Sport Fisheries and Wildlife, 1928-72. (Bureau of Sport Fisheries and Wildlife. Kesource publication 120) Supt. of Docs. no.: 1.49.66:120 1. Fishes Bibliography. 2. Game and game-birds Bibliography. 3. Fish-culture Bibliography. 4. Fishery management Bibliogra phy. 5. Wildlife management Bibliography. I. Harris, Van Thomas, 1915- joint author. II. United States. Bureau of Sport Fisheries and Wildlife. III. Title. IV. Series: United States Bureau of Sport Fisheries and Wildlife. Resource publication 120. S914.A3 no. 120 [Z7996.F5] 639'.9'08s [016.639*9] 74-8411 For sale by the Superintendent of Documents, U.S. Government Printing OfTie Washington, D.C. Price $2.30 Stock Number 2410-00366 BIBLIOGRAPHY OF RESEARCH PUBLICATIONS OF THE U.S. BUREAU OF SPORT FISHERIES AND WILDLIFE, 1928-72 INTRODUCTION This bibliography comprises publications in fishery and wildlife research au thored or coauthored by research scientists of the Bureau of Sport Fisheries and Wildlife and certain predecessor agencies. Separate lists, arranged alphabetically by author, are given for each of 17 fishery research and 6 wildlife research labora tories, stations, investigations, or centers. -

Zootaxa,Crustacean Classification
Zootaxa 1668: 313–325 (2007) ISSN 1175-5326 (print edition) www.mapress.com/zootaxa/ ZOOTAXA Copyright © 2007 · Magnolia Press ISSN 1175-5334 (online edition) Crustacean classification: on-going controversies and unresolved problems* GEOFF A. BOXSHALL Department of Zoology, The Natural History Museum, Cromwell Road, London SW7 5BD, United Kingdom E-mail: [email protected] *In: Zhang, Z.-Q. & Shear, W.A. (Eds) (2007) Linnaeus Tercentenary: Progress in Invertebrate Taxonomy. Zootaxa, 1668, 1–766. Table of contents Abstract . 313 Introduction . 313 Treatment of parasitic Crustacea . 315 Affinities of the Remipedia . 316 Validity of the Entomostraca . 318 Exopodites and epipodites . 319 Using of larval characters in estimating phylogenetic relationships . 320 Fossils and the crustacean stem lineage . 321 Acknowledgements . 322 References . 322 Abstract The journey from Linnaeus’s original treatment to modern crustacean systematics is briefly characterised. Progress in our understanding of phylogenetic relationships within the Crustacea is linked to continuing discoveries of new taxa, to advances in theory and to improvements in methodology. Six themes are discussed that serve to illustrate some of the major on-going controversies and unresolved problems in the field as well as to illustrate changes that have taken place since the time of Linnaeus. These themes are: 1. the treatment of parasitic Crustacea, 2. the affinities of the Remipedia, 3. the validity of the Entomostraca, 4. exopodites and epipodites, 5. using larval characters in estimating phylogenetic rela- tionships, and 6. fossils and the crustacean stem-lineage. It is concluded that the development of the stem lineage concept for the Crustacea has been dominated by consideration of taxa known only from larval or immature stages. -

Helmintos Parásitos De Fauna Silvestre En Las Costas De Guerrero, Oaxaca
University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Estudios en Biodiversidad Parasitology, Harold W. Manter Laboratory of 2015 Helmintos parásitos de fauna silvestre en las costas de Guerrero, Oaxaca y Chiapas, México Griselda Pulido-Flores Universidad Autónoma del Estado de Hidalgo, [email protected] Scott onkM s Universidad Autónoma del Estado de Hidalgo, [email protected] Jorge Falcón-Ordaz Universidad Autónoma del Estado de Hidalgo Juan Violante-González Universidad Autónoma de Guerrero Follow this and additional works at: http://digitalcommons.unl.edu/biodiversidad Part of the Biodiversity Commons, Botany Commons, and the Terrestrial and Aquatic Ecology Commons Pulido-Flores, Griselda; Monks, Scott; Falcón-Ordaz, Jorge; and Violante-González, Juan, "Helmintos parásitos de fauna silvestre en las costas de Guerrero, Oaxaca y Chiapas, México" (2015). Estudios en Biodiversidad. 6. http://digitalcommons.unl.edu/biodiversidad/6 This Article is brought to you for free and open access by the Parasitology, Harold W. Manter Laboratory of at DigitalCommons@University of Nebraska - Lincoln. It has been accepted for inclusion in Estudios en Biodiversidad by an authorized administrator of DigitalCommons@University of Nebraska - Lincoln. Helmintos parásitos de fauna silvestre en las costas de Guerrero, Oaxaca y Chiapas, México Griselda Pulido-Flores, Scott Monks, Jorge Falcón-Ordaz, y Juan Violante-González Resumen La costa sureste del Pacífico en México es rica en biodiversidad, en parte por la posición en la intersección de las corrientes oceánicas ecuatoriales. Sin embargo, los helmintos son un grupo de organismos que ha sido poco estudiado en la región y los registros están en diversas fuentes de información. -

Development of a System-Wide Predator Control Program: Northern Squawfish Management Program
Development of a System-wide Predator Control Program Northern Squawfish Management Program - Implementation Annual Report 1997 October 1998 DOE/BP-24514-8 This Document should be cited as follows: Young, Franklin, "Development of a System-wide Predator Control Program; Northern Squawfish Management Program - Implementation", 1997 Annual Report, Project No. 199007700, 100 electronic pages, (BPA Report DOE/BP-24514-8) Bonneville Power Administration P.O. Box 3621 Portland, OR 97208 This report was funded by the Bonneville Power Administration (BPA), U.S. Department of Energy, as part of BPA's program to protect, mitigate, and enhance fish and wildlife affected by the development and operation of hydroelectric facilities on the Columbia River and its tributaries. The views in this report are the author's and do not necessarily represent the views of BPA. DEVELOPMENT OF A SYSTEMWIDE PREDATOR CONTROL PROGRAM: STEPWISE IMPLEMENTATION OF A PREDATION INDEX, PREDATOR CONTROL FISHERIES, AND EVALUATION PLAN IN THE COLUMBIA RIVER BASIN SECTION I: IMPLEMENTATION 1997 ANNUAL REPORT Prepared by: Franklin R. Young Columbia Basin Fish and Wildlife Authority In Cooperation With Nez Perce Tribe of Idaho Oregon Department of Fish and Wildlife Washington Department of Fish and Wildlife Pacific States Marine Fisheries Commission Columbia River Inter-tribal Fish Commission Confederated Tribes of the Umatilla Indian Reservation Confederated Tribes of the Warm Springs Reservation of Oregon Confederated Tribes and Bands of the Yakama Indian Reservation Prepared -

Review and Meta-Analysis of the Environmental Biology and Potential Invasiveness of a Poorly-Studied Cyprinid, the Ide Leuciscus Idus
REVIEWS IN FISHERIES SCIENCE & AQUACULTURE https://doi.org/10.1080/23308249.2020.1822280 REVIEW Review and Meta-Analysis of the Environmental Biology and Potential Invasiveness of a Poorly-Studied Cyprinid, the Ide Leuciscus idus Mehis Rohtlaa,b, Lorenzo Vilizzic, Vladimır Kovacd, David Almeidae, Bernice Brewsterf, J. Robert Brittong, Łukasz Głowackic, Michael J. Godardh,i, Ruth Kirkf, Sarah Nienhuisj, Karin H. Olssonh,k, Jan Simonsenl, Michał E. Skora m, Saulius Stakenas_ n, Ali Serhan Tarkanc,o, Nildeniz Topo, Hugo Verreyckenp, Grzegorz ZieRbac, and Gordon H. Coppc,h,q aEstonian Marine Institute, University of Tartu, Tartu, Estonia; bInstitute of Marine Research, Austevoll Research Station, Storebø, Norway; cDepartment of Ecology and Vertebrate Zoology, Faculty of Biology and Environmental Protection, University of Lodz, Łod z, Poland; dDepartment of Ecology, Faculty of Natural Sciences, Comenius University, Bratislava, Slovakia; eDepartment of Basic Medical Sciences, USP-CEU University, Madrid, Spain; fMolecular Parasitology Laboratory, School of Life Sciences, Pharmacy and Chemistry, Kingston University, Kingston-upon-Thames, Surrey, UK; gDepartment of Life and Environmental Sciences, Bournemouth University, Dorset, UK; hCentre for Environment, Fisheries & Aquaculture Science, Lowestoft, Suffolk, UK; iAECOM, Kitchener, Ontario, Canada; jOntario Ministry of Natural Resources and Forestry, Peterborough, Ontario, Canada; kDepartment of Zoology, Tel Aviv University and Inter-University Institute for Marine Sciences in Eilat, Tel Aviv, -

Umpqua Bibliography by Groups I. Possible
Umpqua Bibliography by Groups Introduction This bibliography is about the Umpqua River Basin, particularly its hydrology and its fisheries. Some films, photographs and archival materials are included along with books, articles, maps and web pages. The bibliography was built using local, regional and national library catalogs, major online databases, theses and dissertations, watershed assessments and Federal endangered species reports. Its historical coverage begins with Joel Palmer’s 1847 Journal of Travels over the Rocky Mountains and ends in early 2008. I have attempted to cover major physical features of the Umpqua Basin, as well as important aquatic species and critical issues facing the area. Trying to build a comprehensive collection of references on any subject in the age of information is, to use an image Joel Palmer would understand, like trying to catch a greased pig. It is ever squirming out of your grasp. This collection of references is necessarily incomplete, but I’d be grateful to learn of any major omissions. --Susan Gilmont Updated September, 2008 I. Possible Candidates for Digitization: Grey Literature, Important Or Uncataloged Reports on the Umpqua River (Sorted by Date) Roth, A. R. A survey of the waters of the South Umpqua Ranger District : Umpqua National Forest . Portland, Or.: USDA Forest Service, Pacific Northwest Region; 1937. Call Number: OSU Libraries: Valley SH35.O7 U65 Notes: Baseline temperature data for the Umpqua Oregon State Game Commission. Lower Umpqua River Study : Summary : Annual Report. 1949. Call Number: Available from NMFS Environmental and Technical Svcs. Division, 525 NE Oregon St. Portland, OR 97232 (? dated reference). State Library or StreamNet should have this. -

Endangered Species
FEATURE: ENDANGERED SPECIES Conservation Status of Imperiled North American Freshwater and Diadromous Fishes ABSTRACT: This is the third compilation of imperiled (i.e., endangered, threatened, vulnerable) plus extinct freshwater and diadromous fishes of North America prepared by the American Fisheries Society’s Endangered Species Committee. Since the last revision in 1989, imperilment of inland fishes has increased substantially. This list includes 700 extant taxa representing 133 genera and 36 families, a 92% increase over the 364 listed in 1989. The increase reflects the addition of distinct populations, previously non-imperiled fishes, and recently described or discovered taxa. Approximately 39% of described fish species of the continent are imperiled. There are 230 vulnerable, 190 threatened, and 280 endangered extant taxa, and 61 taxa presumed extinct or extirpated from nature. Of those that were imperiled in 1989, most (89%) are the same or worse in conservation status; only 6% have improved in status, and 5% were delisted for various reasons. Habitat degradation and nonindigenous species are the main threats to at-risk fishes, many of which are restricted to small ranges. Documenting the diversity and status of rare fishes is a critical step in identifying and implementing appropriate actions necessary for their protection and management. Howard L. Jelks, Frank McCormick, Stephen J. Walsh, Joseph S. Nelson, Noel M. Burkhead, Steven P. Platania, Salvador Contreras-Balderas, Brady A. Porter, Edmundo Díaz-Pardo, Claude B. Renaud, Dean A. Hendrickson, Juan Jacobo Schmitter-Soto, John Lyons, Eric B. Taylor, and Nicholas E. Mandrak, Melvin L. Warren, Jr. Jelks, Walsh, and Burkhead are research McCormick is a biologist with the biologists with the U.S. -

Comparison of Exploited and Unexploited Yellow Perch Perca ¯Avescens (Mitchill) Populations in Nebraska Sandhill Lakes
Fisheries Management and Ecology, 2001, 8, 533±542 Comparison of exploited and unexploited yellow perch Perca ¯avescens (Mitchill) populations in Nebraska Sandhill lakes C. P. PAUKERT & D. W. WILLIS Department of Wildlife and Fisheries Sciences, South Dakota State University, Brookings, SD, USA Abstract Exploitation can have a pronounced eect on ®sh populations. Yellow perch, Perca ¯avescens (Mitchill), populations in Nebraska Sandhill lakes were sampled in 1998 and 1999. Three of the 29 lakes containing yellow perch have been closed to ®shing for at least 10 years. Unexploited yellow perch populations had fast growth rates, but age structure was similar to exploited populations. For unexploited lakes combined, mortality and condition were not dierent from exploited lakes. However, one unexploited lake, Marsh Lake, had the fastest growth, highest proportion of older ®sh and highest condition of all populations sampled. This lake had low interspeci®c competition and high invertebrate abundance, which likely resulted in fast growth and high condition. However, size structure and growth were also related to lake productivity. Although exploitation may aect yellow perch populations, other factors (food availability, predators and lake productivity) also play an important role in structuring these populations. Regardless, these results indicate the potential of yellow perch in Nebraska Sandhill lakes given no exploitation. KEYWORDS: exploitation, Nebraska, Perca ¯avescens, productivity, yellow perch. Introduction Exploitation can substantially alter ®sh population characteristics. Unexploited populations typically have a high proportion of larger, older ®sh (Goedde & Coble 1981), which also may be in lower body condition (Van Den Avyle & Hayward 1999). Population size structure and abundance may be diminished soon after a lake is open to angling (Redmond 1974; Goedde & Coble 1981), which has been attributed to high harvest of naõÈ ve ®sh. -

Guadalupe Bass Micropterus Treculii (Vaillant & Bocourt, 1874)
American Fisheries Society Symposium 82:55–60, 2015 © 2015 by the American Fisheries Society Guadalupe Bass Micropterus treculii (Vaillant & Bocourt, 1874) STEPHEN G. CURTIS* Aquatic Station, Department of Biology, Texas State University 601 University Drive, San Marcos, Texas 78666, USA JOSHUAH S. PERKIN Division of Biology, Kansas State University 116 Ackert Hall, Manhattan, Kansas 66506, USA PRESTON T. BEAN Department of Natural Resources Management, Texas Tech University 254 Red Raider Lane, Junction, Texas 76849, USA MARIO L. SULLIVAN AND TIMOTHY H. BONNER Aquatic Station, Department of Biology, Texas State University 601 University Drive, San Marcos, Texas 78666, USA Taxonomic Status Guadalupe Bass Micropterus treculii diverged from northeastern ancestral Micropterus (Conner and Suttkus1986) approximately 4.1–5.7 million years ago during the late Miocene or early Pliocene (Near et al. 2003, 2005). The species was originally described by Cope (1880) as the Texas (Johnson Fork of the Llano River) version of Florida Bass M. floridanus, differing slightly in some morphometric and meristic counts from its Florida counterpart. Since that time, Guadalupe Bass have undergone sev- eral redescriptions, including Dioplites treculii (Vaillant and Bocourt 1883), M. nuecensis var. treculii (Vaillant and Bocourt 1883), M. salmoides (Jordan and Gilbert 1886, Evermann and Kendall 1894), M. pseudaplites (Hubbs 1927), M. punctulatus punctulatus (Hubbs and Bailey 1940), M. p. treculii (Hubbs and Bailey 1942), M. treculi (Jurgens and Hubbs 1953; Hubbs 1954), and its current nomenclature M. treculii (Nelson et al. 2004). Johnson et al. (2001) determined that the sister taxa of Guadalupe Bass is Spotted Bass M. punctulatus based on mitochondrial DNA analyses.