The Changing Faces of Vitamin B12 and Folic Acid – Part Two: Integration of Symptoms, and Genetic Testing to Select the Best Drugs for Your Patient
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Methionine Synthase Supports Tumor Tetrahydrofolate Pools
bioRxiv preprint doi: https://doi.org/10.1101/2020.09.05.284521; this version posted September 7, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Methionine synthase supports tumor tetrahydrofolate pools Joshua Z. Wang1,2,#, Jonathan M. Ghergurovich1,3,#, Lifeng Yang1,2, and Joshua D. Rabinowitz1,2,* 1 Lewis-Sigler Institute for Integrative Genomics, Princeton University, Princeton, New Jersey, USA 2 Department of Chemistry, Princeton University, Princeton, New Jersey, USA 3 Department of Molecular Biology, Princeton University, Princeton, New Jersey, USA # These authors contributed equally to this work. *Corresponding author: Joshua Rabinowitz Department of Chemistry and the Lewis-Sigler Institute for Integrative Genomics, Princeton University, Washington Rd, Princeton, NJ 08544, USA Phone: (609) 258-8985; e-mail: [email protected] Conflict of Interest Disclosure: J.D.R. is a paid advisor and stockholder in Kadmon Pharmaceuticals, L.E.A.F. Pharmaceuticals, and Rafael Pharmaceuticals; a paid consultant of Pfizer; a founder, director, and stockholder of Farber Partners and Serien Therapeutics. JDR and JMG are inventors of patents in the area of folate metabolism held by Princeton University. 1 bioRxiv preprint doi: https://doi.org/10.1101/2020.09.05.284521; this version posted September 7, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Abstract Mammalian cells require activated folates to generate nucleotides for growth and division. The most abundant circulating folate species is 5-methyl tetrahydrofolate (5-methyl- THF), which is used to synthesize methionine from homocysteine via the cobalamin-dependent enzyme methionine synthase (MTR). -
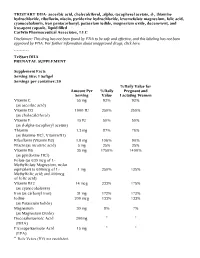
TRISTART DHA- Ascorbic Acid, Cholecalciferol, .Alpha
TRISTART DHA- ascorbic acid, cholecalciferol, .alpha.-tocopherol acetate, d-, thiamine hydrochloride, riboflavin, niacin, pyridoxine hydrochloride, levomefolate magnesium, folic acid, cyanocobalamin, iron pentacarbonyl, potassium iodide, magnesium oxide, doconexent, and icosapent capsule, liquid filled CarWin Pharmaceutical Associates, LLC Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here. ---------- TriStart DHA PRENATAL SUPPLEMENT Supplement Facts Serving Size: 1 Softgel Servings per container: 30 %Daily Value for Amount Per %Daily Pregnant and Serving Value Lactating Women Vitamin C 55 mg 92% 92% (as ascorbic acid) Vitamin D3 1000 IU 250% 250% (as cholecalciferol) Vitamin E 15 IU 50% 50% (as d-alpha-tocopheryl acetate) Thiamin 1.3 mg 87% 76% (as thiamine HCl, Vitamin B1) Riboflavin (Vitamin B2) 1.8 mg 106% 90% Niacin (as nicotinic acid) 5 mg 25% 25% Vitamin B6 35 mg 1750% 1400% (as pyridoxine HCl) Folate (as 630 mcg of L- Methylfolate Magnesium, molar equivalent to 600mcg of L- 1 mg 250% 125% Methylfolic acid; and 400mcg of folic acid) Vitamin B12 14 mcg 233% 175% (as cyanocobalamin) Iron (as carbonyl iron) 31 mg 172% 172% Iodine 200 mcg 133% 133% (as Potassium Iodide) Magnesium 30 mg 8% 7% (as Magnesium Oxide) Docosahexaenoic Acid 200mg * * (DHA) Eicosapentaenoate Acid 15 mg * * (EPA) * Daily Values (DV) not established. OTHER INGREDIENTS: Gelatin (bovine), Glycerin, Purified Water, Yellow Bees Wax, Caramel Powder, Soy Lecithin, Natural Orange Flavor, Ethyl Vanillin. Contains: Soy and Fish TriStart DHA™ Softgel capsules are dye free, lactose, gluten and sugar free. -

The Vitamin B12 Coenzyme
THE VITAMIN B12 COENZYME D. DoLPHIN, A. W. JoHNSON, R. RoDRIGO and N. SHAW Department of Chemistry, University of Nottingham, U.K. INTRODUCTION In 19•58 Barker and his associatesl-3 recognized a new coenzyme which controlled the conversion of glutamate into ß-methylaspartate by Clostridium tetanomorpkim. The coenzyme was shown4 to be related to !f;-vitamin B12, i.e. contair ing an adenine nucleotide grouping in place of the 5,6-dimethyl benziminawle nucleotide of vitamin B12, although similar coenzymes con taining btnziminazole or 5,6-dimethylbenziminazole were produced by growing C. tetanomorphum in the presence of the a ppropriate base5. Other variations of the nucleotide base have been achieved using Propionibacterium arabinosum in the presence of other purines and benziminazoles6• The pres ence of;:he coenzymes in a wide variety of micro-organisms such as several species of Actinomycetes including Streptomyces olivaceus and S. griseus has been dem( mstrated by the glutamate isomerase assay7 or by isolation. I t appears thü Vitamin B12 and its analogues are always biosynthesized in the form of their coenzymes. Preliminary physical and chemical studies sug gested that in the 5,6-dimethylbenzirninazolyl cobamide coenzyme the cyanide gr )up of vitamin B12, cyanocobalamin, was replaced by an adenine nucleoside':, 5, 8 and the determination9 of the complete structure (I; R = 5'-de·)xyadenosyl) of the coenzyme by X-ray analysis revealed the existence c f an essentially covalent bond between the cobalt atom and the S'.. carbon üom of the additional 5'-deoxyadenosine group. The molecule Me CH 2• CO· NH2 In the vitamin 8 12 coenzyme R =5' - deoxyadenosyl = Me Me 539 D. -

L-Carnitine, Mecobalamin and Folic Acid Tablets) TRINERVE-LC
For the use of a Registered Medical Practitioner or a Hospital or a Laboratory only (L-Carnitine, Mecobalamin and Folic acid Tablets) TRINERVE-LC 1. Name of the medicinal product Trinerve-LC Tablets 2. Qualitative and quantitative composition Each film- coated tablets contains L-Carnitine…………………….500 mg Mecobalamin……………….1500 mcg Folic acid IP…………………..1.5mg 3. Pharmaceutical form Film- coated tablets 4. Clinical particulars 4.1 Therapeutic indications Vitamin and micronutrient supplementation in the management of chronic disease. 4.2 Posology and method of administration For oral administration only. One tablet daily or as directed by physician. 4.3 Contraindications Hypersensitivity to any constituent of the product. 4.4 Special warnings and precautions for use L-Carnitine The safety and efficacy of oral L-Carnitine has not been evaluated in patients with renal insufficiency. Chronic administration of high doses of oral L-Carnitine in patients with severely compromised renal function or in ESRD patients on dialysis may result in accumulation of the potentially toxic metabolites, trimethylamine (TMA) and trimethylamine-N-oxide (TMAO), since these metabolites are normally excreted in the urine. Mecobalamin Should be given with caution in patients suffering from folate deficiency. The following warnings and precautions suggested with parent form – vitamin B12 The treatment of vitamin B12 deficiency can unmask the symptoms of polycythemia vera. Megaloblastic anemia is sometimes corrected by treatment with vitamin B12. But this can have very serious side effects. Don’t attempt vitamin B12 therapy without close supervision by healthcare provider. Do not take vitamin B12 if Leber’s disease, a hereditary eye disease. -

Nutrition 102 – Class 3
Nutrition 102 – Class 3 Angel Woolever, RD, CD 1 Nutrition 102 “Introduction to Human Nutrition” second edition Edited by Michael J. Gibney, Susan A. Lanham-New, Aedin Cassidy, and Hester H. Vorster May be purchased online but is not required for the class. 2 Technical Difficulties Contact: Erin Deichman 574.753.1706 [email protected] 3 Questions You may raise your hand and type your question. All questions will be answered at the end of the webinar to save time. 4 Review from Last Week Vitamins E, K, and C What it is Source Function Requirement Absorption Deficiency Toxicity Non-essential compounds Bioflavonoids: Carnitine, Choline, Inositol, Taurine, and Ubiquinone Phytoceuticals 5 Priorities for Today’s Session B Vitamins What they are Source Function Requirement Absorption Deficiency Toxicity 6 7 What Is Vitamin B1 First B Vitamin to be discovered 8 Vitamin B1 Sources Pork – rich source Potatoes Whole-grain cereals Meat Fish 9 Functions of Vitamin B1 Converts carbohydrates into glucose for energy metabolism Strengthens immune system Improves body’s ability to withstand stressful conditions 10 Thiamine Requirements Groups: RDA (mg/day): Infants 0.4 Children 0.7-1.2 Males 1.5 Females 1 Pregnancy 2 Lactation 2 11 Thiamine Absorption Absorbed in the duodenum and proximal jejunum Alcoholics are especially susceptible to thiamine deficiency Excreted in urine, diuresis, and sweat Little storage of thiamine in the body 12 Barriers to Thiamine Absorption Lost into cooking water Unstable to light Exposure to sunlight Destroyed -

(12) Patent Application Publication (10) Pub. No.: US 2005/0196469 A1 Thys-Jacobs (43) Pub
US 2005O196469A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2005/0196469 A1 Thys-Jacobs (43) Pub. Date: Sep. 8, 2005 (54) MICRONUTRIENT SUPPLEMENT (22) Filed: Mar. 4, 2004 COMBINATION FOR ACNE TREATMENT AND PREVENTION Publication Classification (76) Inventor: Susan Thys-Jacobs, Larchmont, NY (51) Int. Cl.' ....................... A61 K 31/59; A61 K 31/525; (US) A61K 33/10; A61K 31/19 (52) U.S. Cl. ......................... 424/687; 514/168; 514/251; Correspondence Address: 514/574 GOTTLEB RACKMAN & REISMAN PC 27O MADSON AVENUE 8TH FLOOR (57) ABSTRACT NEW YORK, NY 100160601 A micronutrient Supplement comprising effective amounts (21) Appl. No.: 10/794,729 of calcium, Vitamin D, and folate treats and prevents acne. US 2005/0196469 A1 Sep. 8, 2005 MICRONUTRIENT SUPPLEMENT COMBINATION therapies include benzoyl peroxide which has comedolytic FOR ACNE TREATMENT AND PREVENTION and antibacterial effects, topical antibacterials Such as eryth romycin or clindamycin, azelaic acid, tazaroc, and topical FIELD OF THE INVENTION retinoids. Acne that is resistant to topical treatment requires oral antibiotics or isotretinoin. Indications for isotretinoin 0001. This invention relates to a micronutrient supple include Severe Scarring, acne that is resistant to oral antibi ment in the treatment of acne Vulgaris and inflammation. In otics and acne present for many years that quickly relapses particular, this invention relates to a multi-vitamin and when an oral antibiotic therapy is discontinued. Of note, oral mineral Supplement for improving skin and hair health. isotretinoin is a potent teratogen. Current Standards of acne therapy include the topical descquarnative drugs and antibac BACKGROUND OF THE INVENTION terial agents. -

The Potential Protective Role of Vitamin K in Diabetic Neuropathy
VITAMINS The potential protective role of vitamin K in diabetic neuropathy DILIP MEHTA Viridis Biopharma 6/10 Jogani Industrial Complex ew cases of diabetes are symptomatic pain relief (3-5). V. N. Purav Marg, Chunabhatti increasing worldwide at a rapid Mumbai 400022, India The etiopathology of peripheral pace, with the total number of neuropathy is poorly understood and many [email protected] people with diabetes was projected factors, including dietary deficiencies, may www.viridisbiopharma.com Nto rise from 171 million in 2000 to 366 million contribute to the clinical manifestation of the in 2030 – an increase of nearly 200 million in condition. Deficiency of vitamin B12 (also only three decades. There are more cases of known as cobalamin), which results in a lack diabetes in women and urban populations, of a related compound, methylcobalamin, is with diabetes in developing countries projected manifested by megaloblastic anemia, and to double in the coming years (1). has been associated with significant Based on reports from the Centers for neurological pathology, especially peripheral Disease Control and Prevention, type 2 neuropathy (6-8). Vitamin B12 is also diabetes dult onset diabetes affects associated with the onset of diabetic approximately 9.3% of the general neuropathy. In patients with diabetic population in the United States in contrast to neuropathy, vitamin B12 deficiency may be 25.9% among those 65 years or older (2). aggravated by the use of antidiabetic agents Diabetes mellitus accounts for 90% of the such as metformin (9-11). Even short-term cases of diabetes patients (3,4). treatment with metformin causes a decrease The prevalence of type 2 diabetes in serum cobalamin, folic acid and an increases with age, higher then 25 body increase in homocysteine, which leads to mass index and the presence of the disease peripheral neuropathy in patients with in family history. -

Tall Man Lettering List REPORT DECEMBER 2013 1
Tall Man Lettering List REPORT DECEMBER 2013 1 TALL MAN LETTERING LIST REPORT WWW.HQSC.GOVT.NZ Published in December 2013 by the Health Quality & Safety Commission. This document is available on the Health Quality & Safety Commission website, www.hqsc.govt.nz ISBN: 978-0-478-38555-7 (online) Citation: Health Quality & Safety Commission. 2013. Tall Man Lettering List Report. Wellington: Health Quality & Safety Commission. Crown copyright ©. This copyright work is licensed under the Creative Commons Attribution-No Derivative Works 3.0 New Zealand licence. In essence, you are free to copy and distribute the work (including other media and formats), as long as you attribute the work to the Health Quality & Safety Commission. The work must not be adapted and other licence terms must be abided. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nd/3.0/nz/ Copyright enquiries If you are in doubt as to whether a proposed use is covered by this licence, please contact: National Medication Safety Programme Team Health Quality & Safety Commission PO Box 25496 Wellington 6146 ACKNOWLEDGEMENTS The Health Quality & Safety Commission acknowledges the following for their assistance in producing the New Zealand Tall Man lettering list: • The Australian Commission on Safety and Quality in Health Care for advice and support in allowing its original work to be either reproduced in whole or altered in part for New Zealand as per its copyright1 • The Medication Safety and Quality Program of Clinical Excellence Commission, New South -

Potential Benefits of Methylcobalamin: a Review
Open Access Austin Journal of Pharmacology and Therapeutics Review Article Potential Benefits of Methylcobalamin: A Review Gupta JK* and Qureshi Shaiba Sana Department of Pharmacology, GLA University Mathura, Abstract India Methylcobalamin is an active form of vitamin B12 that helps in synthesis *Corresponding author: Jeetendra Kumar Gupta, of methionine and S-adenosylmethionine. It is required for integrity of myelin, Department of Pharmacology, Institute of Pharmaceutical neuronal function, proper red blood cell formation and DNA synthesis. The largest Research, GLA University Mathura, India group of vitamin B12 deficiency is found in typical vegetarians all over the world, which can be alleviated with its analogue Methylcobalamin. It is a beneficial Received: August 17, 2015; Accepted: September 30, drug to most of the common disorders like cardiovascular disorders, diabetes, 2015; Published: October 08, 2015 anemia, hyperhomocysteinemia and degenerative disorders. Methylcobalamin helps in the synthesis of neuronal lipids, regeneration of axonal nerves and has neuroprotective activity, which promote neurons to function in proper way and thus improves Alzheimer disease, Parkinsonism, Dementia and neuropathic syndromes. It is an approved treatment for peripheral neuropathy. Keywords: Mecobalamin; Neuropathy; Anemia; Nootropic; Dietary supplement Abbreviations essential for cell growth and replication. Sometimes the liver cannot convert cyanocobalamin into adequate amount of methylcobalamin SAMe: S-Adenosyl Methionine; ERK: Extracellular Signal- needed for proper neuronal functioning. Through enhanced Regulated Kinases; PKB: Protein Kinase B; B-globulin: Beta Globulin; methylation, it exerts its nerve cell protective effect and accelerates ENFD: Epidermal Nerve Fiber Density; DPN: Diabetic Peripheral its growth. A lot of energy is required for cyanocobalamin to remove Neuropathy; NSAIDs: Non Steroidal Anti Inflammatory Drugs; THF: its cyanide and replaces it with methyl group [3]. -

Vitamin and Mineral Requirements in Human Nutrition
P000i-00xx 3/12/05 8:54 PM Page i Vitamin and mineral requirements in human nutrition Second edition VITPR 3/12/05 16:50 Page ii WHO Library Cataloguing-in-Publication Data Joint FAO/WHO Expert Consultation on Human Vitamin and Mineral Requirements (1998 : Bangkok, Thailand). Vitamin and mineral requirements in human nutrition : report of a joint FAO/WHO expert consultation, Bangkok, Thailand, 21–30 September 1998. 1.Vitamins — standards 2.Micronutrients — standards 3.Trace elements — standards 4.Deficiency diseases — diet therapy 5.Nutritional requirements I.Title. ISBN 92 4 154612 3 (LC/NLM Classification: QU 145) © World Health Organization and Food and Agriculture Organization of the United Nations 2004 All rights reserved. Publications of the World Health Organization can be obtained from Market- ing and Dissemination, World Health Organization, 20 Avenue Appia, 1211 Geneva 27, Switzerland (tel: +41 22 791 2476; fax: +41 22 791 4857; e-mail: [email protected]). Requests for permis- sion to reproduce or translate WHO publications — whether for sale or for noncommercial distri- bution — should be addressed to Publications, at the above address (fax: +41 22 791 4806; e-mail: [email protected]), or to Chief, Publishing and Multimedia Service, Information Division, Food and Agriculture Organization of the United Nations, 00100 Rome, Italy. The designations employed and the presentation of the material in this publication do not imply the expression of any opinion whatsoever on the part of the World Health Organization and the Food and Agriculture Organization of the United Nations concerning the legal status of any country, territory, city or area or of its authorities, or concerning the delimitation of its frontiers or boundaries. -

Structural Basis for the Inhibition of Human 5,10-Methenyltetrahydrofolate Synthetase by N10-Substituted Folate Analogues
Published OnlineFirst September 8, 2009; DOI: 10.1158/0008-5472.CAN-09-1927 Experimental Therapeutics, Molecular Targets, and Chemical Biology Structural Basis for the Inhibition of Human 5,10-Methenyltetrahydrofolate Synthetase by N10-Substituted Folate Analogues Dong Wu,1 Yang Li,1 Gaojie Song,1 Chongyun Cheng,1 Rongguang Zhang,2 Andrzej Joachimiak,2 NeilShaw, 1 and Zhi-Jie Liu1 1National Laboratory of Biomacromolecules, Institute of Biophysics, Chinese Academy of Sciences, Beijing, China and 2Structural Biology Center, Argonne National Laboratory, Argonne, Illinois Abstract of the one-carbon metabolic network, dihydrofolate reductase, 5,10-Methenyltetrahydrofolate synthetase (MTHFS) regulates which catalyzes the conversion of dihydrofolate to tetrahydrofolate. the flow of carbon through the one-carbon metabolic network, Similarly, colorectal and pancreatic cancers are routinely treated by which supplies essential components for the growth and administration of the pyrimidine analogue 5-fluorouracil (5-FU), proliferation of cells. Inhibition of MTHFS in human MCF-7 which inhibits thymidylate synthase (TS; ref. 6). TS converts breast cancer cells has been shown to arrest the growth of deoxyuridylate to deoxythymidylate using a carbon donated by cells. Absence of the three-dimensional structure of human 5,10-methylenetetrahydrofolate (Fig. 1). This conversion is essential MTHFS (hMTHFS) has hampered the rational design and for DNA synthesis (7). To increase the clinical efficiency of the optimization of drug candidates. Here, we report the treatment, 5-formyltetrahydrofolate is often coadministered along structures of native hMTHFS, a binary complex of hMTHFS with 5-FU. The beneficial effect of administering 5-formyltetrahy- with ADP, hMTHFS bound with the N5-iminium phosphate drofolate during the therapy with 5-FU is a consequence of the reaction intermediate, and an enzyme-product complex of 5,10-methenyltetrahydrofolate synthetase (MTHFS)–catalyzed hMTHFS. -

BC Cancer Benefit Drug List September 2021
Page 1 of 65 BC Cancer Benefit Drug List September 2021 DEFINITIONS Class I Reimbursed for active cancer or approved treatment or approved indication only. Reimbursed for approved indications only. Completion of the BC Cancer Compassionate Access Program Application (formerly Undesignated Indication Form) is necessary to Restricted Funding (R) provide the appropriate clinical information for each patient. NOTES 1. BC Cancer will reimburse, to the Communities Oncology Network hospital pharmacy, the actual acquisition cost of a Benefit Drug, up to the maximum price as determined by BC Cancer, based on the current brand and contract price. Please contact the OSCAR Hotline at 1-888-355-0355 if more information is required. 2. Not Otherwise Specified (NOS) code only applicable to Class I drugs where indicated. 3. Intrahepatic use of chemotherapy drugs is not reimbursable unless specified. 4. For queries regarding other indications not specified, please contact the BC Cancer Compassionate Access Program Office at 604.877.6000 x 6277 or [email protected] DOSAGE TUMOUR PROTOCOL DRUG APPROVED INDICATIONS CLASS NOTES FORM SITE CODES Therapy for Metastatic Castration-Sensitive Prostate Cancer using abiraterone tablet Genitourinary UGUMCSPABI* R Abiraterone and Prednisone Palliative Therapy for Metastatic Castration Resistant Prostate Cancer abiraterone tablet Genitourinary UGUPABI R Using Abiraterone and prednisone acitretin capsule Lymphoma reversal of early dysplastic and neoplastic stem changes LYNOS I first-line treatment of epidermal