Characterization of CD109
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Human and Mouse CD Marker Handbook Human and Mouse CD Marker Key Markers - Human Key Markers - Mouse
Welcome to More Choice CD Marker Handbook For more information, please visit: Human bdbiosciences.com/eu/go/humancdmarkers Mouse bdbiosciences.com/eu/go/mousecdmarkers Human and Mouse CD Marker Handbook Human and Mouse CD Marker Key Markers - Human Key Markers - Mouse CD3 CD3 CD (cluster of differentiation) molecules are cell surface markers T Cell CD4 CD4 useful for the identification and characterization of leukocytes. The CD CD8 CD8 nomenclature was developed and is maintained through the HLDA (Human Leukocyte Differentiation Antigens) workshop started in 1982. CD45R/B220 CD19 CD19 The goal is to provide standardization of monoclonal antibodies to B Cell CD20 CD22 (B cell activation marker) human antigens across laboratories. To characterize or “workshop” the antibodies, multiple laboratories carry out blind analyses of antibodies. These results independently validate antibody specificity. CD11c CD11c Dendritic Cell CD123 CD123 While the CD nomenclature has been developed for use with human antigens, it is applied to corresponding mouse antigens as well as antigens from other species. However, the mouse and other species NK Cell CD56 CD335 (NKp46) antibodies are not tested by HLDA. Human CD markers were reviewed by the HLDA. New CD markers Stem Cell/ CD34 CD34 were established at the HLDA9 meeting held in Barcelona in 2010. For Precursor hematopoetic stem cell only hematopoetic stem cell only additional information and CD markers please visit www.hcdm.org. Macrophage/ CD14 CD11b/ Mac-1 Monocyte CD33 Ly-71 (F4/80) CD66b Granulocyte CD66b Gr-1/Ly6G Ly6C CD41 CD41 CD61 (Integrin b3) CD61 Platelet CD9 CD62 CD62P (activated platelets) CD235a CD235a Erythrocyte Ter-119 CD146 MECA-32 CD106 CD146 Endothelial Cell CD31 CD62E (activated endothelial cells) Epithelial Cell CD236 CD326 (EPCAM1) For Research Use Only. -
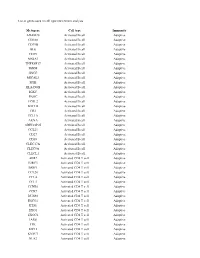
List of Genes Used in Cell Type Enrichment Analysis
List of genes used in cell type enrichment analysis Metagene Cell type Immunity ADAM28 Activated B cell Adaptive CD180 Activated B cell Adaptive CD79B Activated B cell Adaptive BLK Activated B cell Adaptive CD19 Activated B cell Adaptive MS4A1 Activated B cell Adaptive TNFRSF17 Activated B cell Adaptive IGHM Activated B cell Adaptive GNG7 Activated B cell Adaptive MICAL3 Activated B cell Adaptive SPIB Activated B cell Adaptive HLA-DOB Activated B cell Adaptive IGKC Activated B cell Adaptive PNOC Activated B cell Adaptive FCRL2 Activated B cell Adaptive BACH2 Activated B cell Adaptive CR2 Activated B cell Adaptive TCL1A Activated B cell Adaptive AKNA Activated B cell Adaptive ARHGAP25 Activated B cell Adaptive CCL21 Activated B cell Adaptive CD27 Activated B cell Adaptive CD38 Activated B cell Adaptive CLEC17A Activated B cell Adaptive CLEC9A Activated B cell Adaptive CLECL1 Activated B cell Adaptive AIM2 Activated CD4 T cell Adaptive BIRC3 Activated CD4 T cell Adaptive BRIP1 Activated CD4 T cell Adaptive CCL20 Activated CD4 T cell Adaptive CCL4 Activated CD4 T cell Adaptive CCL5 Activated CD4 T cell Adaptive CCNB1 Activated CD4 T cell Adaptive CCR7 Activated CD4 T cell Adaptive DUSP2 Activated CD4 T cell Adaptive ESCO2 Activated CD4 T cell Adaptive ETS1 Activated CD4 T cell Adaptive EXO1 Activated CD4 T cell Adaptive EXOC6 Activated CD4 T cell Adaptive IARS Activated CD4 T cell Adaptive ITK Activated CD4 T cell Adaptive KIF11 Activated CD4 T cell Adaptive KNTC1 Activated CD4 T cell Adaptive NUF2 Activated CD4 T cell Adaptive PRC1 Activated -

CD109 Released from Human Bone Marrow Mesenchymal Stem Cells Attenuates TGF-Β-Induced Epithelial to Mesenchymal Transition and Stemness of Squamous Cell Carcinoma
www.impactjournals.com/oncotarget/ Oncotarget, 2017, Vol. 8, (No. 56), pp: 95632-95647 Research Paper CD109 released from human bone marrow mesenchymal stem cells attenuates TGF-β-induced epithelial to mesenchymal transition and stemness of squamous cell carcinoma Shufeng Zhou1, Renzo Cecere2 and Anie Philip1 1Division of Plastic Surgery, Department of Surgery, McGill University, Montreal, QC, Canada 2Division of Cardiac Surgery, Department of Surgery, McGill University, Montreal, QC, Canada Correspondence to: Anie Philip, email: [email protected] Keywords: human bone marrow mesenchymal stem cells; squamous cell carcinoma; CD109; TGF-β; epithelial to mesenchymal transition Abbreviations: hBM-MSC-CM: human bone marrow mesenchymal stem cells conditioned medium; hFibro-CM: human fibroblast cells conditioned medium; EMT: epithelial to mesenchymal transition; SCC: squamous cell carcinoma Received: March 31, 2017 Accepted: July 06, 2017 Published: September 16, 2017 Copyright: Zhou et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License 3.0 (CC BY 3.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. ABSTRACT Although there is increasing evidence that human bone marrow mesenchymal stem cells (hBM-MSCs) play an important role in cancer progression, the underlying mechanisms are poorly understood. Transforming growth factor β (TGF-β) is an important pro-metastatic cytokine. We have previously shown that CD109, a glycosylphosphatidylinositol-anchored protein, is a TGF-β co-receptor and a strong inhibitor of TGF-β signalling. Moreover, CD109 can be released from the cell surface. In the current study, we examined whether hBM-MSCs regulate the malignant properties of squamous cell carcinoma cells, and whether CD109 plays a role in mediating the effect of hBM-MSCs on cancer cells. -

Supplementary Table 1: Adhesion Genes Data Set
Supplementary Table 1: Adhesion genes data set PROBE Entrez Gene ID Celera Gene ID Gene_Symbol Gene_Name 160832 1 hCG201364.3 A1BG alpha-1-B glycoprotein 223658 1 hCG201364.3 A1BG alpha-1-B glycoprotein 212988 102 hCG40040.3 ADAM10 ADAM metallopeptidase domain 10 133411 4185 hCG28232.2 ADAM11 ADAM metallopeptidase domain 11 110695 8038 hCG40937.4 ADAM12 ADAM metallopeptidase domain 12 (meltrin alpha) 195222 8038 hCG40937.4 ADAM12 ADAM metallopeptidase domain 12 (meltrin alpha) 165344 8751 hCG20021.3 ADAM15 ADAM metallopeptidase domain 15 (metargidin) 189065 6868 null ADAM17 ADAM metallopeptidase domain 17 (tumor necrosis factor, alpha, converting enzyme) 108119 8728 hCG15398.4 ADAM19 ADAM metallopeptidase domain 19 (meltrin beta) 117763 8748 hCG20675.3 ADAM20 ADAM metallopeptidase domain 20 126448 8747 hCG1785634.2 ADAM21 ADAM metallopeptidase domain 21 208981 8747 hCG1785634.2|hCG2042897 ADAM21 ADAM metallopeptidase domain 21 180903 53616 hCG17212.4 ADAM22 ADAM metallopeptidase domain 22 177272 8745 hCG1811623.1 ADAM23 ADAM metallopeptidase domain 23 102384 10863 hCG1818505.1 ADAM28 ADAM metallopeptidase domain 28 119968 11086 hCG1786734.2 ADAM29 ADAM metallopeptidase domain 29 205542 11085 hCG1997196.1 ADAM30 ADAM metallopeptidase domain 30 148417 80332 hCG39255.4 ADAM33 ADAM metallopeptidase domain 33 140492 8756 hCG1789002.2 ADAM7 ADAM metallopeptidase domain 7 122603 101 hCG1816947.1 ADAM8 ADAM metallopeptidase domain 8 183965 8754 hCG1996391 ADAM9 ADAM metallopeptidase domain 9 (meltrin gamma) 129974 27299 hCG15447.3 ADAMDEC1 ADAM-like, -

Integrin Alpha 2 Antibody
Product Datasheet Integrin alpha 2 Antibody Catalog No: #49071 Package Size: #49071-1 50ul #49071-2 100ul Orders: [email protected] Support: [email protected] Description Product Name Integrin alpha 2 Antibody Host Species Rabbit Clonality Monoclonal Clone No. SN0752 Purification ProA affinity purified Applications WB, ICC/IF, IHC, IP, FC Species Reactivity Hu, Ms, Rt Immunogen Description recombinant protein Other Names BR antibody CD 49b antibody CD49 antigen like family member B antibody CD49 antigen-like family member B antibody CD49b antibody CD49b antigen antibody Collagen receptor antibody DX5 antibody Glycoprotein Ia deficiency included antibody GP Ia antibody GP Ia deficiency, included antibody GPIa antibody HPA 5 included antibody HPA5 included antibody Human platelet alloantigen system 5 antibody Integrin alpha 2 antibody Integrin alpha-2 antibody Integrin, alpha 2 (CD49B alpha 2 subunit of VLA 2 receptor) antibody ITA2_HUMAN antibody ITGA2 antibody Platelet alloantigen Br(a), included antibody Platelet antigen Br antibody Platelet glycoprotein GPIa antibody Platelet glycoprotein Ia antibody Platelet glycoprotein Ia/IIa antibody Platelet membrane glycoprotein Ia antibody Platelet receptor for collagen, deficiency of, included antibody Very late activation protein 2 receptor alpha 2 subunit antibody VLA 2 alpha chain antibody VLA 2 antibody VLA 2 subunit alpha antibody VLA-2 subunit alpha antibody VLA2 antibody VLA2 receptor alpha 2 subunit antibody VLAA2 antibody Accession No. Swiss-Prot#:P17301 Calculated MW 150 kDa Formulation 1*TBS (pH7.4), 1%BSA, 40%Glycerol. Preservative: 0.05% Sodium Azide. Storage Store at -20°C Application Details WB: 1:1,000-5,000 IHC: 1:50-1:200 ICC: 1:100-1:500 FC: 1:50-1:100 Images Address: 8400 Baltimore Ave., Suite 302, College Park, MD 20740, USA http://www.sabbiotech.com 1 Western blot analysis of ITGA2 on A431 cells lysates using anti-ITGA2 antibody at 1/1,000 dilution. -

Integrin Α2β1 in Nonactivated Conformation Can Induce Focal
www.nature.com/scientificreports OPEN Integrin α2β1 in nonactivated conformation can induce focal adhesion kinase signaling Received: 17 February 2017 Maria Salmela1, Johanna Jokinen1,2, Silja Tiitta1, Pekka Rappu1, R. Holland Cheng 2 & Jyrki Accepted: 2 May 2017 Heino1 Published: xx xx xxxx Conformational activation of integrins is generally required for ligand binding and cellular signalling. However, we have previously reported that the nonactivated conformation of α2β1 integrin can also bind to large ligands, such as human echovirus 1. In this study, we show that the interaction between the nonactivated integrin and a ligand resulted in the activation of focal adhesion kinase (FAK) in a protein kinase C dependent manner. A loss-of-function mutation, α2E336A, in the α2-integrin did not prevent the activation of FAK, nor did EDTA-mediated inactivation of the integrin. Full FAK activation was observed, since phosphorylation was not only confirmed in residue Y397, but also in residues Y576/7. Furthermore, initiation of downstream signaling by paxillin phosphorylation in residue Y118 was evident, even though this activation was transient by nature, probably due to the lack of talin involvement in FAK activation and the absence of vinculin in the adhesion complexes formed by the nonactivated integrins. Altogether these results indicate that the nonactivated integrins can induce cellular signaling, but the outcome of the signaling differs from conventional integrin signaling. The well-established model of integrin function stresses the importance of the conformational change from a nonactivated, bent conformation to an active, extended state prior to ligand binding and outside-in signaling1. Despite the fact that the conformational activation of integrins is often required for ligand binding, nonactivated integrins may have specific biological functions as well. -

Development of Novel Monoclonal Antibodies Against CD109 Overexpressed in Human Pancreatic Cancer
www.oncotarget.com Oncotarget, 2018, Vol. 9, (No. 28), pp: 19994-20007 Research Paper Development of novel monoclonal antibodies against CD109 overexpressed in human pancreatic cancer Gustavo A. Arias-Pinilla1, Angus G. Dalgleish2, Satvinder Mudan3, Izhar Bagwan4, Anthony J. Walker1 and Helmout Modjtahedi1 1School of Life Sciences, Pharmacy and Chemistry, Kingston University London, Kingston-upon-Thames, Surrey, UK 2Department of Cellular and Molecular Medicine, St George’s University of London, London, UK 3Department of Surgery of Hammersmith Campus, Imperial College, London, UK 4Department of Histopathology, Royal Surrey County Hospital, Guildford, UK Correspondence to: Helmout Modjtahedi, email: [email protected] Keywords: pancreatic cancer; monoclonal antibodies; CD109 antigen; tissue arrays; immunohistochemistry Received: December 10, 2017 Accepted: March 15, 2018 Published: April 13, 2018 Copyright: Arias-Pinilla et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License 3.0 (CC BY 3.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. ABSTRACT Pancreatic cancer is one of the most aggressive and lethal types of cancer, and more effective therapeutic agents are urgently needed. Overexpressed cell surface antigens are ideal targets for therapy with monoclonal antibody (mAb)-based drugs, but none have been approved for the treatment of pancreatic cancer. Here, we report development of two novel mouse mAbs, KU42.33C and KU43.13A, against the human pancreatic cancer cell line BxPC-3. Using ELISA, flow cytometry, competitive assay and immunoprecipitation followed by mass spectrometry, we discovered that these two mAbs target two distinct epitopes on the external domain of CD109 that are overexpressed by varying amounts in human pancreatic cancer cell lines. -

Multiomics of Azacitidine-Treated AML Cells Reveals Variable And
Multiomics of azacitidine-treated AML cells reveals variable and convergent targets that remodel the cell-surface proteome Kevin K. Leunga, Aaron Nguyenb, Tao Shic, Lin Tangc, Xiaochun Nid, Laure Escoubetc, Kyle J. MacBethb, Jorge DiMartinob, and James A. Wellsa,1 aDepartment of Pharmaceutical Chemistry, University of California, San Francisco, CA 94143; bEpigenetics Thematic Center of Excellence, Celgene Corporation, San Francisco, CA 94158; cDepartment of Informatics and Predictive Sciences, Celgene Corporation, San Diego, CA 92121; and dDepartment of Informatics and Predictive Sciences, Celgene Corporation, Cambridge, MA 02140 Contributed by James A. Wells, November 19, 2018 (sent for review August 23, 2018; reviewed by Rebekah Gundry, Neil L. Kelleher, and Bernd Wollscheid) Myelodysplastic syndromes (MDS) and acute myeloid leukemia of DNA methyltransferases, leading to loss of methylation in (AML) are diseases of abnormal hematopoietic differentiation newly synthesized DNA (10, 11). It was recently shown that AZA with aberrant epigenetic alterations. Azacitidine (AZA) is a DNA treatment of cervical (12, 13) and colorectal (14) cancer cells methyltransferase inhibitor widely used to treat MDS and AML, can induce interferon responses through reactivation of endoge- yet the impact of AZA on the cell-surface proteome has not been nous retroviruses. This phenomenon, termed viral mimicry, is defined. To identify potential therapeutic targets for use in com- thought to induce antitumor effects by activating and engaging bination with AZA in AML patients, we investigated the effects the immune system. of AZA treatment on four AML cell lines representing different Although AZA treatment has demonstrated clinical benefit in stages of differentiation. The effect of AZA treatment on these AML patients, additional therapeutic options are needed (8, 9). -

The VE-Cadherin/Amotl2 Mechanosensory Pathway Suppresses Aortic In�Ammation and the Formation of Abdominal Aortic Aneurysms
The VE-cadherin/AmotL2 mechanosensory pathway suppresses aortic inammation and the formation of abdominal aortic aneurysms Yuanyuan Zhang Karolinska Institute Evelyn Hutterer Karolinska Institute Sara Hultin Karolinska Institute Otto Bergman Karolinska Institute Maria Forteza Karolinska Institute Zorana Andonovic Karolinska Institute Daniel Ketelhuth Karolinska University Hospital, Stockholm, Sweden Joy Roy Karolinska Institute Per Eriksson Karolinska Institute Lars Holmgren ( [email protected] ) Karolinska Institute Article Keywords: arterial endothelial cells (ECs), vascular disease, abdominal aortic aneurysms Posted Date: June 15th, 2021 DOI: https://doi.org/10.21203/rs.3.rs-600069/v1 License: This work is licensed under a Creative Commons Attribution 4.0 International License. Read Full License The VE-cadherin/AmotL2 mechanosensory pathway suppresses aortic inflammation and the formation of abdominal aortic aneurysms Yuanyuan Zhang1, Evelyn Hutterer1, Sara Hultin1, Otto Bergman2, Maria J. Forteza2, Zorana Andonovic1, Daniel F.J. Ketelhuth2,3, Joy Roy4, Per Eriksson2 and Lars Holmgren1*. 1Department of Oncology-Pathology, BioClinicum, Karolinska Institutet, Stockholm, Sweden. 2Department of Medicine Solna, BioClinicum, Karolinska Institutet, Karolinska University Hospital, Stockholm, Sweden. 3Department of Cardiovascular and Renal Research, Institutet of Molecular Medicine, Univ. of Southern Denmark, Odense, Denmark 4Department of Molecular Medicine and Surgery, Karolinska Institutet, Karolinska University Hospital, Stockholm, -

Functional Activity in Vitro / in Vivo Product Guide How to Automate a TOC
Functional Activity In Vitro / In Vivo Product Guide how to automate a TOC http://help.adobe.com/en_US/indesign/cs/using/WS49FB9AF6-38AB-42fb-B056-8DACE18DDF63a.html Table of Contents 1. Functional Activity 1 eBioscience, an Affymetrix company, is committed to Functional Grade Antibodies . 2 developing and manufacturing high-quality, innovative reagents using an ISO certified process. As a provider of Recombinant Proteins . 3 more than 11,000 products, we empower our customers worldwide to obtain exceptional results by using reagents 2. T Cell and B Cell Activation 4 that offer a new standard of excellence in the areas of T Cell Activation . 4 innovation, quality and value. B Cell Activation . 5 Co-stimulation . 7 3. Cell Differentiation 9 T helper (Th) Cell Differentiation . 9 Monocyte, Macrophage and Dendritic Cell Differentiation . 13 Natural Killer Cell Differentiation . .14 4. Product Guide 15 Functional Grade Antibodies by Cell Type B Cells . 15 General T Cells . .16 Th1 Cells . .17 Th2 Cells . .17 Th9 Cells . .17 Th17 Cells . 18 Th22 Cells . 18 T Follicular Helper Cells (Tfh) . 18 Treg Cells. .18 CD8 T Cells. .19 Unless indicated otherwise, all products are For Research Use Only. Not for use in diagnostic or therapeutic procedures. Natural Killer (NK) Cells. .19 All designated trademarks used in this publication are the property of their respective owners. Monocyte, Macrophage and Dendritic Cells . 20 ©Affymetrix, Inc. All rights reserved. BestProtocols®, eBioscience®, eFluor®, Full Spectrum Megakaryocyte and Erythrocyte Cells. .21 Cell Analysis®, InstantOne ELISA™, OneComp eBeads™, ProcartaPlex™, Ready-SET-Go!®, SAFE™ Super AquaBlue®, The New Standard of Excellence® and UltraComp eBeads™ are trademarks or registered trademarks of eBioscience, Inc. -

CD24, CD27, CD36 and CD302 Gene Expression for Outcome Prediction in Patients with Multiple Myeloma
www.impactjournals.com/oncotarget/ Oncotarget, 2017, Vol. 8, (No. 58), pp: 98931-98944 Research Paper CD24, CD27, CD36 and CD302 gene expression for outcome prediction in patients with multiple myeloma Elina Alaterre6,2, Sebastien Raimbault6, Hartmut Goldschmidt4,5, Salahedine Bouhya7, Guilhem Requirand1,2, Nicolas Robert1,2, Stéphanie Boireau1,2, Anja Seckinger4,5, Dirk Hose4,5, Bernard Klein1,2,3 and Jérôme Moreaux1,2,3 1Department of Biological Haematology, CHU Montpellier, Montpellier, France 2Institute of Human Genetics, CNRS-UM UMR9002, Montpellier, France 3University of Montpellier, UFR Medecine, Montpellier, France 4Medizinische Klinik und Poliklinik V, Universitätsklinikum Heidelberg, Heidelberg, Germany 5Nationales Centrum für Tumorerkrankungen, Heidelberg, Germany 6HORIBA Medical, Parc Euromédecine, Montpellier, France 7CHU Montpellier, Department of Clinical Hematology, Montpellier, France Correspondence to: Jérôme Moreaux, email: [email protected] Keywords: multiple myeloma; prognostic factor; gene expression profiling; cluster differentiation Received: February 10, 2017 Accepted: August 27, 2017 Published: October 30, 2017 Copyright: Alaterre et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License 3.0 (CC BY 3.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. ABSTRACT Multiple myeloma (MM) is a B cell neoplasia characterized by clonal plasma cell (PC) proliferation. Minimal residual disease monitoring by multi-parameter flow cytometry is a powerful tool for predicting treatment efficacy and MM outcome. In this study, we compared CD antigens expression between normal and malignant plasma cells to identify new potential markers to discriminate normal from malignant plasma cells, new potential therapeutic targets for monoclonal-based treatments and new prognostic factors. -

Independence Blue Cross
Independence Blue Cross The following codes are under management for members who have health benefits covered by Independence Blue Cross, administered by eviCore healthcare. Commercial Effective 7/1/2016 Medicare Effective 1/1/2021 Procedure How Code is Managed Commercial & Effective Date Termination Date Code Full Description Medicare Legend: Requires Prior Authorization- Requests containing these codes should be submitted directly to eviCore Claim Policies Apply-eviCore manages this code with claim edits. This code by itself does not require prior authorization. However, all procedure codes (81105-81599) included in a multiple procedure code panel are subject to medical necessity review if any code requires prior authorization. This ensures a holistic approach to a panel test. Human Platelet Antigen 1 genotyping (HPA-1), ITGB3 (integrin, beta 3 [platelet glycoprotein 81105 IIIa], antigen CD61 [GPIIIa]) (eg, neonatal alloimmune thrombocytopenia [NAIT], post- Claim Policies Apply 01/01/18 None transfusion purpura), gene analysis, common variant, HPA-1a/b (L33P) Human Platelet Antigen 2 genotyping (HPA-2), GP1BA (glycoprotein Ib [platelet], alpha 81106 polypeptide [GPIba]) (eg, neonatal alloimmune thrombocytopenia [NAIT], post-transfusion Claim Policies Apply 01/01/18 None purpura), gene analysis, common variant, HPA-2a/b (T145M) Human Platelet Antigen 3 genotyping (HPA-3), ITGA2B (integrin, alpha 2b [platelet 81107 glycoprotein IIb of IIb/IIIa complex], antigen CD41 [GPIIb]) (eg, neonatal alloimmune Claim Policies Apply 01/01/18 None thrombocytopenia