Wittig and Wittig-Horner Reactions Under Phase Transfer Catalysis
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Supporting Information a Convenient Chromatography-Free Method For
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry This journal is © The Royal Society of Chemistry 2012 Supporting Information A Convenient Chromatography-Free Method for the Purification of Alkenes Produced in the Wittig Reaction Peter A. Byrne, Kamalraj V. Rajendran, Jimmy Muldoon and Declan G. Gilheany Centre for Synthesis and Chemical Biology, School of Chemistry and Chemical Biology, University College Dublin, Belfield, Dublin 4, Ireland 1. General Experimental 2 2. Synthesis of phosphonium salts 4 3. Procedures for Wittig reactions & phosphine oxide removal 11 4. Characterisation of purified alkenes 15 5. Reduction of phosphine chalcogenides 34 6. Synthesis & isolation of neomenthyl chloride and reduction of phosphine oxide-by-product 37 7. NMR spectra of phosphonium salts 42 8. NMR spectra of purified alkenes and regenerated phosphines from Wittig reactions 56 9. NMR spectra of phosphines produced by reduction of phosphine chalcogenides 85 10. NMR spectra of purified products from Appel-type reactions 90 11. References 91 1 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry This journal is © The Royal Society of Chemistry 2012 1. General Experimental All chemicals were supplied by Aldrich, with the exception of Zeoprep silica, 2- methylbenzaldehyde ( o-tolualdehyde, Fluka), ( tert -butoxycarbonylmethyl)- -1 triphenylphosphonium bromide (Fluka), 1 mol L LiAlH 4 in THF (Acros Organics) and Merck standardised alumina 90. All chemicals were used without further purification except diethyl ether, toluene, and THF, which were processed through an Innovative Technology Inc. Pure Solv-400-3-MD solvent purification (Grubbs still) system and stored in Strauss flasks under a nitrogen atmosphere, and ethyl acetate and dichloromethane, which were degassed by passing a stream of dry nitrogen gas (oxygen- free) through the solvent for one hour for the purposes of work-ups in phosphine syntheses. -

Synthetic Routes to Bromo-Terminated Phosphonate Films and Alkynyl Pyridine Compounds for Click Coupling
University of Mary Washington Eagle Scholar Student Research Submissions Spring 5-7-2018 Synthetic Routes to Bromo-Terminated Phosphonate Films and Alkynyl Pyridine Compounds for Click Coupling Poornima Sunder Follow this and additional works at: https://scholar.umw.edu/student_research Part of the Biochemistry Commons Recommended Citation Sunder, Poornima, "Synthetic Routes to Bromo-Terminated Phosphonate Films and Alkynyl Pyridine Compounds for Click Coupling" (2018). Student Research Submissions. 226. https://scholar.umw.edu/student_research/226 This Honors Project is brought to you for free and open access by Eagle Scholar. It has been accepted for inclusion in Student Research Submissions by an authorized administrator of Eagle Scholar. For more information, please contact [email protected]. Synthetic Routes to Bromo-Terminated Phosphonate Films and Alkynyl Pyridine Compounds for Click Coupling Poornima Rachel Sunder Thesis submitted to the faculty of University of Mary Washington in partial fulfillment of the requirements for graduation with Honors in Chemistry (2018) ABSTRACT Click reactions are a highly versatile class of reactions that produce a diverse range of products. Copper-catalyzed azide-alkyne cycloaddition (CuAAC) click reactions require an azide and a terminal alkyne and produce a coupled product that is “clicked” through a triazole ring that can have a variety of substituents. In this work, bromo-terminated phosphonate films on copper oxide surfaces were explored as the platform for click coupling, as the terminal azide needed for the reaction can be generated through an in situ SN2 reaction with a terminal bromo group. The reactions were characterized using model reactions in solution before being conducted on modified copper oxide surfaces. -

19.13 the Wittig Alkene Synthesis 933
19_BRCLoudon_pgs5-0.qxd 12/9/08 11:41 AM Page 933 19.13 THE WITTIG ALKENE SYNTHESIS 933 PROBLEMS 19.32 Draw the structures of all aldehydes or ketones that could in principle give the following product after application of either the Wolff–Kishner or Clemmensen reduction. H3C CH2CH(CH3)2 L L 19.33 Outline a synthesis of 1,4-dimethoxy-2-propylbenzene from hydroquinone (p-hydroxyphenol) and any other reagents. 19.13 THE WITTIG ALKENE SYNTHESIS Our tour through aldehyde and ketone chemistry started with simple additions; then addition followed by substitution (acetal formation); then additions followed by elimination (imine and enamine formation). Another addition–elimination reaction, called the Wittig alkene synthe- sis, is an important method for preparing alkenes from aldehydes and ketones. An example of the Wittig alkene synthesis is the preparation of methylenecyclohexane from cyclohexanone. 1 1 A | A | O CH_ 2 PPh3 CH2 Ph3P O _ (19.70) 1 + 3 anL ylid + L 1 3 triphenylphosphine cyclohexanone methylenecyclohexane oxide The Wittig synthesis is especially important because it gives alkenes in which the position of the double bond is unambiguous; in other words, the Wittig synthesis is completely regios- elective.Itcanbeusedforthepreparationofalkenesthatwouldbedifficulttoprepareby other reactions. For example, methylenecyclohexane, which is readily prepared by the Wittig synthesis (Eq. 19.70), cannot be prepared by dehydration of 1-methylcyclohexanol; 1- methylcyclohexene is obtained instead, because alcohol dehydration gives the alkene iso- mer(s) in which the double bond has the greatest number of alkyl substituents (Sec. 10.1). OH " H2SO4 " CH3 H2O CH3 L + 1-methylcyclohexene H2SO4 (19.71) A CH2 (little or none formed) methylenecyclohexane The nucleophile in the Wittig alkene synthesis is a type of ylid. -
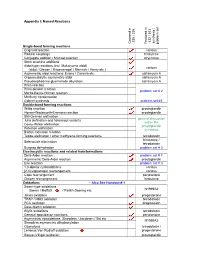
Appendix I: Named Reactions Single-Bond Forming Reactions Co
Appendix I: Named Reactions 235 / 335 432 / 533 synthesis / / synthesis Covered in Covered Featured in problem set problem Single-bond forming reactions Grignard reaction various Radical couplings hirstutene Conjugate addition / Michael reaction strychnine Stork enamine additions Aldol-type reactions (incl. Mukaiyama aldol) various (aldol / Claisen / Knoevenagel / Mannich / Henry etc.) Asymmetric aldol reactions: Evans / Carreira etc. saframycin A Organocatalytic asymmetric aldol saframycin A Pseudoephedrine glycinamide alkylation saframycin A Prins reaction Prins-pinacol reaction problem set # 2 Morita-Baylis-Hillman reaction McMurry condensation Gabriel synthesis problem set #3 Double-bond forming reactions Wittig reaction prostaglandin Horner-Wadsworth-Emmons reaction prostaglandin Still-Gennari olefination general discussion Julia olefination and heteroaryl variants within the Corey-Winter olefination prostaglandin Peterson olefination synthesis Barton extrusion reaction Tebbe olefination / other methylene-forming reactions tetrodotoxin hirstutene / Selenoxide elimination tetrodotoxin Burgess dehydration problem set # 3 Electrocyclic reactions and related transformations Diels-Alder reaction problem set # 1 Asymmetric Diels-Alder reaction prostaglandin Ene reaction problem set # 3 1,3-dipolar cycloadditions various [2,3] sigmatropic rearrangement various Cope rearrangement periplanone Claisen rearrangement hirstutene Oxidations – Also See Handout # 1 Swern-type oxidations (Swern / Moffatt / Parikh-Doering etc. N1999A2 Jones oxidation -

Studies in Directed Catalytic Asymmetric Hydroboration of 1,2-Disubstituted Unsaturated Amide
University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Student Research Projects, Dissertations, and Chemistry, Department of Theses - Chemistry Department Summer 7-2017 STUDIES IN DIRECTED CATALYTIC ASYMMETRIC HYDROBORATION OF 1,2-DISUBSTITUTED UNSATURATED AMIDE Shuyang Zhang University of Nebraska - Lincoln, [email protected] Follow this and additional works at: http://digitalcommons.unl.edu/chemistrydiss Part of the Organic Chemistry Commons Zhang, Shuyang, "STUDIES IN DIRECTED CATALYTIC ASYMMETRIC HYDROBORATION OF 1,2-DISUBSTITUTED UNSATURATED AMIDE" (2017). Student Research Projects, Dissertations, and Theses - Chemistry Department. 85. http://digitalcommons.unl.edu/chemistrydiss/85 This Article is brought to you for free and open access by the Chemistry, Department of at DigitalCommons@University of Nebraska - Lincoln. It has been accepted for inclusion in Student Research Projects, Dissertations, and Theses - Chemistry Department by an authorized administrator of DigitalCommons@University of Nebraska - Lincoln. STUDIES IN DIRECTED CATALYTIC ASYMMETRIC HYDROBORATION OF 1,2- DISUBSTITUTED UNSATURATED AMIDE by Shuyang Zhang A THESIS Presented to the Faculty of The Graduate College at the University of Nebraska In Partial Fulfillment of Requirements For the Degree of Master of Science Major: Chemistry Under the Supervision of Professor James M. Takacs Lincoln, Nebraska July 2017 DIRECTED CATALYTIC ASYMMETRIC HYDROBORATION OF 1,2- DISUBSTITUTED ALKENES Shuyang Zhang, M.S. University of Nebraska 2017 Advisor: Professor James M. Takacs The Rh-catalyzed, substrate directed catalytic asymmetric hydroboration of γ,δ-unsaturated amides provides a direct route to enantioenriched acyclic secondary γ- borylated carbonyl derivatives with high regio- and enantioselectivity. The catalytic condition optimization and substrate scope study is discussed, including the effects in catalytical asymmetric hydroboration on pre-installed chiral γ,δ-unsaturated amides. -

H-Phosphonates: Versatile Synthetic Precursors to Biologically Active Phosphorus Compounds*
Pure Appl. Chem., Vol. 79, No. 12, pp. 2217–2227, 2007. doi:10.1351/pac200779122217 © 2007 IUPAC H-Phosphonates: Versatile synthetic precursors to biologically active phosphorus compounds* Adam Kraszewski1,‡ and Jacek Stawinski1,2,‡ 1Institute of Bioorganic Chemistry, Polish Academy of Sciences, Noskowskiego 12/14, 61-704 Poznan, Poland; 2Department of Organic Chemistry, Arrhenius Laboratory, Stockholm University, S-10691 Stockholm, Sweden Abstract: In this review, a short account of H-phosphonate chemistry and its application to the synthesis of biologically important phosphates and their analogs is given. Keywords: H-phosphonates; phosphate analogs; biological activity; nucleic acids; drugs. INTRODUCTION Located at the crossroad of various bioinformation exchange pathways, phosphorus-containing com- pounds play a key role in living organisms as carriers of genetic information and important signalling, regulatory, energy transfer, and structural compounds [1]. Due to this pivotal role, biologically impor- tant phosphorus compounds have become therapeutic targets in various modern medicinal techniques, such as antisense [2] and antigene [3] approaches to modulation gene expression, or a gene silencing technique using short interfering RNA (siRNA) [4]. To maximize the desired biological effect and to minimize cytotoxicity, a biologically active com- pound has to have very precisely adjusted chemical and pharmacokinetic properties. This can be achieved by, e.g., changing pKa values of the ionizeable functions, changing electronegativity of the substituents, their size, hydrophobicity, etc. These features can also influence transport of a drug across cell membranes, its preferential degradation in normal or activation in the neoplastic or virus-infected cells, and can contribute to the overall efficiency of the drug. A prominent example here are antiviral pronucleotides in which the phosphate moiety is masked with alkyl or aryl groups that are removed chemically and enzymatically in cells to produce an active antiviral nucleotide [5]. -

The Hydrolysis of Phosphinates and Phosphonates: a Review
molecules Review The Hydrolysis of Phosphinates and Phosphonates: A Review Nikoletta Harsági and György Keglevich * Department of Organic Chemistry and Technology, Budapest University of Technology and Economics, 1521 Budapest, Hungary; [email protected] * Correspondence: [email protected]; Tel.: +36-1-463-1111 (ext. 5883) Abstract: Phosphinic and phosphonic acids are useful intermediates and biologically active com- pounds which may be prepared from their esters, phosphinates and phosphonates, respectively, by hydrolysis or dealkylation. The hydrolysis may take place both under acidic and basic conditions, but the C-O bond may also be cleaved by trimethylsilyl halides. The hydrolysis of P-esters is a challenging task because, in most cases, the optimized reaction conditions have not yet been explored. Despite the importance of the hydrolysis of P-esters, this field has not yet been fully surveyed. In order to fill this gap, examples of acidic and alkaline hydrolysis, as well as the dealkylation of phosphinates and phosphonates, are summarized in this review. Keywords: hydrolysis; dealkylation; phosphinates; phosphonates; P-acids 1. Introduction Phosphinic and phosphonic acids are of great importance due to their biological activity (Figure1)[ 1]. Most of them are known as antibacterial agents [2,3]. Multidrug- resistant (MDR) and extensively drug-resistant (XDR) pathogens may cause major problems Citation: Harsági, N.; Keglevich, G. in the treatment of bacterial infections. However, Fosfomycin has remained active against The Hydrolysis of Phosphinates and both Gram-positive and Gram-negative MDR and XDR bacteria [2]. Acyclic nucleoside Phosphonates: A Review. Molecules phosphonic derivatives like Cidofovir, Adefovir and Tenofovir play an important role 2021, 26, 2840. -

The Wittig Reaction
UC Berkeley College of Chemistry Chemistry 112B Organic Chemistry The Wittig Reaction Author: Jonathan Melville Graduate Student Instructor: Rebecca Triano March 13, 2014 1 Introduction The Wittig reaction, discovered in 1954 by Georg Wittig, is one of the most common tech- niques used for the stereoselective preparation of alkenes. Broadly speaking, the reaction allows for the formation of an alkene product and a triphenylphosphine oxide side product from the reaction of an aldehyde or ketone and a \Wittig reagent" (a triphenylphospho- nium ylide). In our experiment, we formed methyl (2E)-3-(2-nitrophenyl)acrylate and triphenylphosphine oxide from the reaction of 2-nitrobenzaldehyde and methyl (triph- enylphosphoranylidene) acetate. The key step of the mechanism of the ylide reaction is the nucleophilic addition of the ylide to the electrophilic carbonyl group, forming a 4-membered ring that dissociates into the product molecules. The stereoselectivity of the reaction is predicated on the stability of triphenylphosphonium ylide, which determines which of two ring intermediates form: the sterically-unfavored cis intermediate that forms via a fast yet reversible process, or the slow, irreversible trans intermediate. Because ylides contain (by definition) adjacent positive and negative charges (a positive on the phosphonium, and a negative on the carbon adjacent to the residue), R groups that can better stabilize the adjacent negative charge produce more stable ylides. We can thus classify three distinct levels of stabiliza- tion of the ylide by its residue: neutral substituents that do not stabilize negative charge (like alkyl groups or hydrogens) produce an unstabilized ylide that preferentially forms cis product, whereas electron-withdrawing residues (like nitro or ester groups) stabilize the charge and preferentially form trans product, while \semi-stabilized" substituents (generally aromatic rings) produce a mixture of the two. -

Tandem-, Domino- and One-Pot Reactions Involving Wittig- And
DOI: 10.5772/intechopen.70364 Provisional chapter Chapter 1 Tandem-, Domino- and One-Pot Reactions Involving Wittig- and Horner-Wadsworth-Emmons Olefination Tandem-, Domino- and One-Pot Reactions Involving Wittig- and Horner-Wadsworth-Emmons Olefination Fatima Merza, Ahmed Taha and Thies Thiemann Fatima Merza, Ahmed Taha and Thies Thiemann Additional information is available at the end of the chapter Additional information is available at the end of the chapter http://dx.doi.org/10.5772/intechopen.70364 Abstract The Wittig olefination utilizing phosphoranes and the related Horner-Wadsworth- Emmons (HWE) reaction using phosphonates transform aldehydes and ketones into substituted alkenes. Because of the versatility of the reactions and the compatibility of many functional groups towards the transformations, both Wittig olefination and HWE reactions are a mainstay in the arsenal of organic synthesis. Here, an overview is given on Wittig- and Horner-Wadsworth-Emmons (HWE) reactions run in combination with other transformations in one-pot procedures. The focus lies on one-pot oxidation Wittig/HWE protocols, Wittig/HWE olefinations run in concert with metal catalyzed cross-coupling reactions, Domino Wittig/HWE—cycloaddition and Wittig-Michael transformations. Keywords: Wittig olefination, one-pot reactions, Domino reactions, tandem reactions, Horner-Wadsworth-Emmons olefination 1. Introduction The Wittig olefination utilizing phosphoranes and the related Horner-Wadsworth-Emmons (HWE) reaction using phosphonates transform aldehydes and ketones into substituted alkenes. Because of the versatility of the reactions and the compatibility of many functional groups in the transformations, both Wittig olefination and HWE reactions are a mainstay in the arsenal of organic synthesis. The mechanism of the Wittig olefination has been the subject of intense debate [1]. -

Investigation of Factors Influencing the Stereochemistry of the Wittig Reaction
THE INVESTIGATION OF FACTORS INFLUENCING THE STEREOCHEMISTRY OF THE WITTIG REACTION By JEROME THOMAS KRESSE A DISSERTATION PRESENTED TO THE GRADUATE COUNCIL OF THE UNIVERSITY OF FLORIDA IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF DOCTOR OF PHILOSOPHY UNIVERSITY OF FLORIDA December, 1965 ACKNOWLEDGMENTS The author wishes to express his appreciation to his research director, Dr. George B. Butler, for his guidance and encouragement during the execution of this work. The author also expresses his gratitude to his fellow graduate students and associates for their helpful suggestions and criticisms. Particular thanks are due Mrs. Frances Kost and Mrs. Thyra Johnston for their conscientious typing of this dissertation. The author also thanks his wife for her patience, encouragement and under- standing. Without her cooperation this work would not have been possible. The financial support of the Petroleum Research Fund is also gratefully acknowledged. TABLE OF CONTENTS PAGE ACKNOWLEDGMENTS ii LIST OF TABLES v LIST OF FIGURES vi CHAPTER I INTRODUCTION 1 Historical Background 1 Stereochemistry 9 Statement of the Problem 19 Method of Attack 20 H DISCUSSION AND RESULTS 22 Solvents 22 Temperature Effects 27 Reaction Times 33 Concentration Effects 34 Substituent Effects 37 Anion Effects 49 1, 4 -Addition and Isomerization 51 TABLE OF CONTEXTS (Continued) CHAPTER PAGE EI EXPERIMENTAL 54 Equipment and Data 54 Source and Purification of Materials 54 Solvents 55 Aldehydes " 57 Miscellaneous Chemicals 57 Preparation and Purification of Phosphonium -

Bridging Mcmurry and Wittig in One-Pot
Digital Comprehensive Summaries of Uppsala Dissertations from the Faculty of Science and Technology 1755 Bridging McMurry and Wittig in One-Pot Olefins from Stereoselective, Reductive Couplings of Two Aldehydes via Phosphaalkenes JURI MAI ACTA UNIVERSITATIS UPSALIENSIS ISSN 1651-6214 ISBN 978-91-513-0536-3 UPPSALA urn:nbn:se:uu:diva-368873 2019 Dissertation presented at Uppsala University to be publicly examined in Häggsalen, Ångströmlaboratoriet, Lägerhyddsvägen 1, Uppsala, Friday, 15 February 2019 at 10:15 for the degree of Doctor of Philosophy. The examination will be conducted in English. Faculty examiner: Professor Dr. Christian Müller (Freie Universität Berlin, Institute of Chemistry and Biochemistry). Abstract Mai, J. 2019. Bridging McMurry and Wittig in One-Pot. Olefins from Stereoselective, Reductive Couplings of Two Aldehydes via Phosphaalkenes. Digital Comprehensive Summaries of Uppsala Dissertations from the Faculty of Science and Technology 1755. 112 pp. Uppsala: Acta Universitatis Upsaliensis. ISBN 978-91-513-0536-3. The formation of C=C bonds is of great importance for fundamental and industrial synthetic organic chemistry. There are many different methodologies for the construction of C=C bonds in the literature, but currently only the McMurry reaction allows the reductive coupling of two carbonyl compounds to form alkenes. This thesis contributes to the field of carbonyl olefinations and presents the development of a new synthetic protocol for a one-pot reductive coupling of two aldehydes to alkenes based on organophosphorus chemistry. The coupling reagent, a phosphanylphosphonate, reacts with an aldehyde to yield a phosphaalkene intermediate which upon activation with a base undergoes an olefination with a second aldehyde. A general overview of synthetic methods for carbonyl olefinations and the chemistry of phosphaalkenes is given in the background chapter. -

Olefination Reactions Lecture Notes OTBS OPMB O
Olefination Reactions Lecture Notes OTBS OPMB O Key Reviews: O Me O Me XX Wittig Reaction K. C. Nicolaou and co-workers, Ann. 1997, 1283. Horner-Wadsworth-Emmons and Tebbe Olefinations S. E. Kelly, Comprehensive Org. Synth. 1991, Vol. 1, 729. Peterson Olefination D. J. Ager, Synthesis 1984, 384. Julia (Julia-Lythgoe) Olefination B. M. Trost, Bull. Chem. Soc. Jpn. 1988, 61, 107. Wittig Olefination: Background and Principles R1 n-BuLi, Ph P 3 O X LDA, R R1 LiHMDS PPh + 1 3 X Ph3P + R1 Ph3P pKa = 18-20 ylide when R = alkyl, H Ph Ph Ph Ph Ph R1 P Ph P -[Ph3P=O] O R1 O R strong bond 1 formation drives reaction oxaphosphatane betaine G. Wittig and G. Schollkopf, Chem. Ber. 1954, 87, 1318. Wittig Olefination: Background and Principles Stereoselectivity with non-stabilized ylides Me OMe Me Ph3P Ph3P Ph3P Me Ph3P Ph3P Not stable; must be made in situ and used immediately Wittig Olefination: Background and Principles Stereoselectivity with non-stabilized ylides Me OMe Me Ph3P Ph3P Ph3P Me Ph3P Ph3P Not stable; must be made in situ and used immediately Addition to carbonyl is an irreversible and concerted [2+2] cycloaddition such that the R groups on the aldehyde and the ylide are as far apart as possible PPh3 Ph3P O H H H O + Me Me Ph3P R O R H -[Ph3P=O] As the size of the R groups increases, selectivity for Z-alkene increases Me Nonpolar solvents favor initial addition Polar solvents favor the elimination Z-alkene Wittig Olefination: Background and Principles Stereoselectivity with stabilized ylides Me O O Ph P Ph3P 3 Ph3P Me OEt semistabilized Incredibly stable; not moisture sensitive, can be chromatographed Price for stability is lower reactivity: reacts well with aldehydes, slowly with ketones Wittig Olefination: Background and Principles Stereoselectivity with stabilized ylides Me O O Ph P Ph3P 3 Ph3P Me OEt semistabilized Incredibly stable; not moisture sensitive, can be chromatographed Price for stability is lower reactivity: reacts well with aldehydes, slowly with ketones Initial addition to carbonyl is reversible so the thermodynamic elimination product results.