Gluconeogenesis Is Essential for Trypanosome Development in The
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

A Simple Colorimetric RNA Polymerase Assay
Virology 274, 429–437 (2000) doi:10.1006/viro.2000.0492, available online at http://www.idealibrary.com on Exploiting Polymerase Promiscuity: A Simple Colorimetric RNA Polymerase Assay William Vassiliou,* Jeffery B. Epp,† Bin-Bin Wang,* Alfred M. Del Vecchio,‡,1 Theodore Widlanski,§ and C. Cheng Kao*,2 *Department of Biology, Indiana University, Bloomington, Indiana 47405; †Dow AgroSciences, Indianapolis, Indiana 46268; ‡SmithKline Beecham Inc., Collegeville, Pennsylvania 19426; and §Department of Chemistry, Indiana University, Bloomington, Indiana 47405 Received May 18, 2000; accepted June 29, 2000 We developed a convenient colorimetric assay for monitoring RNA synthesis from DNA-dependent RNA polymerases (DdRp) and viral RNA-dependent RNA polymerases (RdRp). ATP and GTP with a p-nitrophenyl moiety attached to the ␥-phosphate were synthesized (PNP–NTPs). These PNP–NTPs can be used for RNA synthesis by several RNA polymerases, including the RdRps from brome mosaic virus and bovine viral diarrhea virus and the DdRps from bacteriophage T7 and SP6. When the polymerase reactions were performed in the presence of alkaline phosphatase, which digests the p-nitrophe- nylpyrophosphate side-product of phosphoryl transfer to the chromogenic p-nitrophenylate, an increase in absorbence at 405 nm was observed. These nucleotide analogues were used in continuous colorimetric monitoring of polymerase activity. Furthermore, the PNP–NTPs were found to be stable and utilized by RNA polymerases in the presence of human plasma. This simple colorimetric polymerase assay can be performed in a standard laboratory spectrophotometer and will be useful in screens for inhibitors of viral RNA synthesis. © 2000 Academic Press INTRODUCTION polymerases are primarily tested using radioactive assays that require special handling of the isotope RNA and DNA polymerases carry out the synthesis of and are incompatible with continuous monitoring of oligonucleotides by transfer of a nucleoside monophos- polymerase activity (Ferrari et al., 1999). -

The Conserved Structure of Plant Telomerase RNA Provides the Missing Link for an Evolutionary Pathway from Ciliates to Humans
The conserved structure of plant telomerase RNA provides the missing link for an evolutionary pathway from ciliates to humans Jiarui Songa, Dhenugen Logeswaranb,1, Claudia Castillo-Gonzáleza,1, Yang Lib, Sreyashree Bosea, Behailu Birhanu Aklilua, Zeyang Mac,d, Alexander Polkhovskiya,e, Julian J.-L. Chenb,2, and Dorothy E. Shippena,2 aDepartment of Biochemistry and Biophysics, Texas A&M University, College Station, TX 77843; bSchool of Molecular Sciences, Arizona State University, Tempe, AZ 85287; cNational Maize Improvement Center of China, China Agricultural University, 100193 Beijing, China; dCollege of Agronomy and Biotechnology, China Agricultural University, 100193 Beijing, China; and eCenter of Life Sciences, Skolkovo Institute of Science and Technology, 121205 Moscow, Russian Federation Edited by Thomas R. Cech, University of Colorado Boulder, Boulder, CO, and approved October 24, 2019 (received for review September 4, 2019) Telomerase is essential for maintaining telomere integrity. Although transcribed by RNA polymerase III (Pol III) (6, 7). The La-related telomerase function is widely conserved, the integral telomerase protein P65 in Tetrahymena recognizes the 3′ poly-U tail of TR RNA (TR) that provides a template for telomeric DNA synthesis has and bends the RNA to facilitate telomerase RNP assembly (8, 9). diverged dramatically. Nevertheless, TR molecules retain 2 highly In contrast, fungi maintain much larger TR molecules (900 to conserved structural domains critical for catalysis: a template- 2,400 nt) that are transcribed by RNA polymerase II (Pol II) (3). proximal pseudoknot (PK) structure and a downstream stem-loop The 3′ end maturation of fungal TRs requires components of the structure. Here we introduce the authentic TR from the plant canonical snRNA biogenesis pathway and results in RNP assembly Arabidopsis thaliana, called AtTR, identified through next-generation sequencing of RNAs copurifying with Arabidopsis TERT. -

Supplementary Materials
Supplementary Materials Figure S1. Differentially abundant spots between the mid-log phase cells grown on xylan or xylose. Red and blue circles denote spots with increased and decreased abundance respectively in the xylan growth condition. The identities of the circled spots are summarized in Table 3. Figure S2. Differentially abundant spots between the stationary phase cells grown on xylan or xylose. Red and blue circles denote spots with increased and decreased abundance respectively in the xylan growth condition. The identities of the circled spots are summarized in Table 4. S2 Table S1. Summary of the non-polysaccharide degrading proteins identified in the B. proteoclasticus cytosol by 2DE/MALDI-TOF. Protein Locus Location Score pI kDa Pep. Cov. Amino Acid Biosynthesis Acetylornithine aminotransferase, ArgD Bpr_I1809 C 1.7 × 10−4 5.1 43.9 11 34% Aspartate/tyrosine/aromatic aminotransferase Bpr_I2631 C 3.0 × 10−14 4.7 43.8 15 46% Aspartate-semialdehyde dehydrogenase, Asd Bpr_I1664 C 7.6 × 10−18 5.5 40.1 17 50% Branched-chain amino acid aminotransferase, IlvE Bpr_I1650 C 2.4 × 10−12 5.2 39.2 13 32% Cysteine synthase, CysK Bpr_I1089 C 1.9 × 10−13 5.0 32.3 18 72% Diaminopimelate dehydrogenase Bpr_I0298 C 9.6 × 10−16 5.6 35.8 16 49% Dihydrodipicolinate reductase, DapB Bpr_I2453 C 2.7 × 10−6 4.9 27.0 9 46% Glu/Leu/Phe/Val dehydrogenase Bpr_I2129 C 1.2 × 10−30 5.4 48.6 31 64% Imidazole glycerol phosphate synthase Bpr_I1240 C 8.0 × 10−3 4.7 22.5 8 44% glutamine amidotransferase subunit Ketol-acid reductoisomerase, IlvC Bpr_I1657 C 3.8 × 10−16 -

Yeast Genome Gazetteer P35-65
gazetteer Metabolism 35 tRNA modification mitochondrial transport amino-acid metabolism other tRNA-transcription activities vesicular transport (Golgi network, etc.) nitrogen and sulphur metabolism mRNA synthesis peroxisomal transport nucleotide metabolism mRNA processing (splicing) vacuolar transport phosphate metabolism mRNA processing (5’-end, 3’-end processing extracellular transport carbohydrate metabolism and mRNA degradation) cellular import lipid, fatty-acid and sterol metabolism other mRNA-transcription activities other intracellular-transport activities biosynthesis of vitamins, cofactors and RNA transport prosthetic groups other transcription activities Cellular organization and biogenesis 54 ionic homeostasis organization and biogenesis of cell wall and Protein synthesis 48 plasma membrane Energy 40 ribosomal proteins organization and biogenesis of glycolysis translation (initiation,elongation and cytoskeleton gluconeogenesis termination) organization and biogenesis of endoplasmic pentose-phosphate pathway translational control reticulum and Golgi tricarboxylic-acid pathway tRNA synthetases organization and biogenesis of chromosome respiration other protein-synthesis activities structure fermentation mitochondrial organization and biogenesis metabolism of energy reserves (glycogen Protein destination 49 peroxisomal organization and biogenesis and trehalose) protein folding and stabilization endosomal organization and biogenesis other energy-generation activities protein targeting, sorting and translocation vacuolar and lysosomal -

Table S1. List of Oligonucleotide Primers Used
Table S1. List of oligonucleotide primers used. Cla4 LF-5' GTAGGATCCGCTCTGTCAAGCCTCCGACC M629Arev CCTCCCTCCATGTACTCcgcGATGACCCAgAGCTCGTTG M629Afwd CAACGAGCTcTGGGTCATCgcgGAGTACATGGAGGGAGG LF-3' GTAGGCCATCTAGGCCGCAATCTCGTCAAGTAAAGTCG RF-5' GTAGGCCTGAGTGGCCCGAGATTGCAACGTGTAACC RF-3' GTAGGATCCCGTACGCTGCGATCGCTTGC Ukc1 LF-5' GCAATATTATGTCTACTTTGAGCG M398Arev CCGCCGGGCAAgAAtTCcgcGAGAAGGTACAGATACGc M398Afwd gCGTATCTGTACCTTCTCgcgGAaTTcTTGCCCGGCGG LF-3' GAGGCCATCTAGGCCATTTACGATGGCAGACAAAGG RF-5' GTGGCCTGAGTGGCCATTGGTTTGGGCGAATGGC RF-3' GCAATATTCGTACGTCAACAGCGCG Nrc2 LF-5' GCAATATTTCGAAAAGGGTCGTTCC M454Grev GCCACCCATGCAGTAcTCgccGCAGAGGTAGAGGTAATC M454Gfwd GATTACCTCTACCTCTGCggcGAgTACTGCATGGGTGGC LF-3' GAGGCCATCTAGGCCGACGAGTGAAGCTTTCGAGCG RF-5' GAGGCCTGAGTGGCCTAAGCATCTTGGCTTCTGC RF-3' GCAATATTCGGTCAACGCTTTTCAGATACC Ipl1 LF-5' GTCAATATTCTACTTTGTGAAGACGCTGC M629Arev GCTCCCCACGACCAGCgAATTCGATagcGAGGAAGACTCGGCCCTCATC M629Afwd GATGAGGGCCGAGTCTTCCTCgctATCGAATTcGCTGGTCGTGGGGAGC LF-3' TGAGGCCATCTAGGCCGGTGCCTTAGATTCCGTATAGC RF-5' CATGGCCTGAGTGGCCGATTCTTCTTCTGTCATCGAC RF-3' GACAATATTGCTGACCTTGTCTACTTGG Ire1 LF-5' GCAATATTAAAGCACAACTCAACGC D1014Arev CCGTAGCCAAGCACCTCGgCCGAtATcGTGAGCGAAG D1014Afwd CTTCGCTCACgATaTCGGcCGAGGTGCTTGGCTACGG LF-3' GAGGCCATCTAGGCCAACTGGGCAAAGGAGATGGA RF-5' GAGGCCTGAGTGGCCGTGCGCCTGTGTATCTCTTTG RF-3' GCAATATTGGCCATCTGAGGGCTGAC Kin28 LF-5' GACAATATTCATCTTTCACCCTTCCAAAG L94Arev TGATGAGTGCTTCTAGATTGGTGTCggcGAAcTCgAGCACCAGGTTG L94Afwd CAACCTGGTGCTcGAgTTCgccGACACCAATCTAGAAGCACTCATCA LF-3' TGAGGCCATCTAGGCCCACAGAGATCCGCTTTAATGC RF-5' CATGGCCTGAGTGGCCAGGGCTAGTACGACCTCG -

Yeast As a Tool to Understand the Significance of Human Disease
G C A T T A C G G C A T genes Review Yeast as a Tool to Understand the Significance of Human Disease-Associated Gene Variants Tiziana Cervelli and Alvaro Galli * Yeast Genetics and Genomics Group, Laboratory of Functional Genetics and Genomics, Institute of Clinical Physiology CNR, Via Moruzzi 1, 56125 Pisa, Italy; [email protected] * Correspondence: [email protected] Abstract: At present, the great challenge in human genetics is to provide significance to the growing amount of human disease-associated gene variants identified by next generation DNA sequencing technologies. Increasing evidences suggest that model organisms are of pivotal importance to addressing this issue. Due to its genetic tractability, the yeast Saccharomyces cerevisiae represents a valuable model organism for understanding human genetic variability. In the present review, we show how S. cerevisiae has been used to study variants of genes involved in different diseases and in different pathways, highlighting the versatility of this model organism. Keywords: yeast; Saccharomyces cerevisiae; functional assays; Mendelian disease; cancer; human gene variants 1. Introduction The advent of next generation DNA sequences technologies allows us to identify Citation: Cervelli, T.; Galli, A. Yeast individual genetic variants, and this revolutionized the strategy for discovering the cause as a Tool to Understand the of several genetic and multifactorial diseases. The association between genetic variants Significance of Human and disease risk can potentially overturn our understanding of common disease. The Disease-Associated Gene Variants. discovery of pathways and processes that are causally involved in a disease, so far not Genes 2021, 12, 1303. https:// linked, may open the way towards the development of targeted therapies and prevention doi.org/10.3390/genes12091303 strategies. -

RNA-Dependent RNA Polymerase Speed and Fidelity Are Not the Only Determinants of the Mechanism Or Efficiency of Recombination
G C A T T A C G G C A T genes Article RNA-Dependent RNA Polymerase Speed and Fidelity are not the Only Determinants of the Mechanism or Efficiency of Recombination Hyejeong Kim y, Victor D. Ellis III, Andrew Woodman, Yan Zhao, Jamie J. Arnold y and , Craig E. Cameron y * Department of Biochemistry and Molecular Biology, The Pennsylvania State University, 201 Althouse Laboratory, University Park, PA 16802, USA; [email protected] (H.K.); [email protected] (V.D.E.); [email protected] (A.W.); [email protected] (Y.Z.); [email protected] (J.J.A.) * Correspondence: [email protected]; Tel.: +1-919-966-9699 Present address: Department of Microbiology and Immunology, School of Medicine, University of North y Carolina at Chapel Hill, 125 Mason Farm Rd., Chapel Hill, NC 27599-7290, USA. Received: 15 October 2019; Accepted: 21 November 2019; Published: 25 November 2019 Abstract: Using the RNA-dependent RNA polymerase (RdRp) from poliovirus (PV) as our model system, we have shown that Lys-359 in motif-D functions as a general acid in the mechanism of nucleotidyl transfer. A K359H (KH) RdRp derivative is slow and faithful relative to wild-type enzyme. In the context of the KH virus, RdRp-coding sequence evolves, selecting for the following substitutions: I331F (IF, motif-C) and P356S (PS, motif-D). We have evaluated IF-KH, PS-KH, and IF-PS-KH viruses and enzymes. The speed and fidelity of each double mutant are equivalent. Each exhibits a unique recombination phenotype, with IF-KH being competent for copy-choice recombination and PS-KH being competent for forced-copy-choice recombination. -

T7 RNA Polymerase from Escherichia Coli BL 21/Par 1219 Nucleoside-Triphosphate: RNA Nucleotidyltransferase (DNA-Directed), EC 2.7.7.6 Cat
For life science research only. Not for use in diagnostic procedures. T7 RNA Polymerase From Escherichia coli BL 21/pAR 1219 Nucleoside-triphosphate: RNA nucleotidyltransferase (DNA-directed), EC 2.7.7.6 Cat. No. 10 881 767 001 y Version 22 1,000 U Content version: March 2016 Cat. No. 10 881 775 001 5,000 U Store at Ϫ15 to Ϫ25° C Product Overview Standard Transcription Assay Pack Content Additional Reagents Required • lin. template DNA including T7 RNA promotor Vial Content • Ribonucleoside triphosphates T7 RNA • 1,000 U • labeled nucleotide (according to application) Polymerase • 5,000 U •RNAse inhibitor Enzyme storage buffer: 10 mM Potassium phosphate, • Water, PCR Grade 200 mM KCl, 0.1 mM EDTA, 30 mM 2-mercaptoethanol, 50% glycerol (v/v), 0.1% Tween 20, pH 7.9 (+4°C). Radioactive Assay Supplied Transcrip- Buffer composition (10x conc.): Pipet the following components into a microfuge tube, mix and make up to a tion buffer 0.4 M Tris-HCl, pH 8.0 (+20°C), 60 mM MgCl2 , 100 mM final volume of 20 µl: dithiothreitol, 20 mM spermidine. Storage and Stability Reagent Volume/Concentration Stable at -15 to -25°C until the expiration date printed on the label. Template DN A 0.5 µg Product Description Nucleotides ATP, GTP, CTP, UTP each 0.5 mM final T7 RNA polymerase is commonly used to transcribe DNA which has been α 32 cloned into vectors which have two phage promoters in opposite orientation. Labeled nucleotide [ - P] CTP [400 Ci/ 0.1 µl aqueous solution RNA can be selectively synthesized from either strand of the insert DNA with mmol; 15 TBq/mmol] different polymerases. -
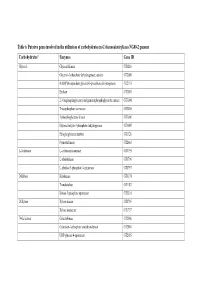
Table 6. Putative Genes Involved in the Utilization of Carbohydrates in G
Table 6. Putative genes involved in the utilization of carbohydrates in G. thermodenitrificans NG80-2 genome Carbohydrates* Enzymes Gene ID Glycerol Glycerol Kinase GT1216 Glycerol-3-phosphate dehydrogenase, aerobic GT2089 NAD(P)H-dependent glycerol-3-phosphate dehydrogenase GT2153 Enolase GT3003 2,3-bisphosphoglycerate-independentphosphoglycerate mutase GT3004 Triosephosphate isomerase GT3005 3-phosphoglycerate kinase GT3006 Glyceraldehyde-3-phosphate dehydrogenase GT3007 Phosphoglycerate mutase GT1326 Pyruvate kinase GT2663 L-Arabinose L-arabinose isomerase GT1795 L-ribulokinase GT1796 L-ribulose 5-phosphate 4-epimerase GT1797 D-Ribose Ribokinase GT3174 Transketolase GT1187 Ribose 5-phosphate epimerase GT3316 D-Xylose Xylose kinase GT1756 Xylose isomerase GT1757 D-Galactose Galactokinase GT2086 Galactose-1-phosphate uridyltransferase GT2084 UDP-glucose 4-epimerase GT2085 Carbohydrates* Enzymes Gene ID D-Fructose 1-phosphofructokinase GT1727 Fructose-1,6-bisphosphate aldolase GT1805 Fructose-1,6-bisphosphate aldolase type II GT3331 Triosephosphate isomerase GT3005 D-Mannose Mannnose-6 phospate isomelase GT3398 6-phospho-1-fructokinase GT2664 D-Mannitol Mannitol-1-phosphate dehydrogenase GT1844 N-Acetylglucosamine N-acetylglucosamine-6-phosphate deacetylase GT2205 N-acetylglucosamine-6-phosphate isomerase GT2204 D-Maltose Alpha-1,4-glucosidase GT0528, GT1643 Sucrose Sucrose phosphorylase GT3215 D-Trehalose Alpha-glucosidase GT1643 Glucose kinase GT2381 Inositol Myo-inositol catabolism protein iolC;5-dehydro-2- GT1807 deoxygluconokinase -

Datasheet for T7 RNA Polymerase
Applications: 1X RNAPol Reaction Buffer: Incubate at 37°C for 1 hour. For shorter (< 300 nt) • Radiolabeled RNA probes 40 mM Tris-HCl transcripts incubate at 37°C for 2–16 hours. T7 RNA Polymerase 6 mM MgCl • Non-isotopic RNA labeling 2 2 mM spermidine Notes on use: • Preparation of RNA vaccines 1 mM dithiothreitol For radio labeled high specific activity RNA 1-800-632-7799 • Guide RNA for gene targeting pH 7.9 @ 25°C probes, the concentration of the radioactive [email protected] nucleotide should be limited to 6 μM. www.neb.com • mRNA for in vitro translation and micro injection Unit Definition: One unit is defined as the amount of M0251S 013160118011 • RNA structure, processing and catalysis studies enzyme required to incorporate 1 nmol ATP into an To protect RNA against ribonuclease, RNase inhibitor (NEB #M0314 or #M0307) should be • RNA amplification acid-insoluble material in 1 hour at 37°C. added to a final concentration of 1 U/μl. M0251S • Anti-sense RNA for gene expression experiment Unit Assay Conditions: 1X RNAPol Reaction Buffer, T7 RNA Polymerase is extremely sensitive to salt supplemented with 0.5 mM each ATP, UTP, GTP, CTP 5,000 units 50,000 U/ml Lot: 0131601 Supplied in: 100 mM NaCl, 50 mM Tris-HCl (pH 7.9), inhibition. The overall salt concentration should and 1 µg T7 DNA in 50 µl. RECOMBINANT Store at –20°C Exp: 1/18 1 mM EDTA, 20 mM 2-mercaptoethanol, 0.1% Triton not exceed 50 mM. X-100 and 50% glycerol. Description: Bacteriophage T7 RNA Polymerase is Protocol for Standard RNA Synthesis: Assemble the Quality Control Assays a DNA-dependent RNA polymerase that is highly reaction at room temperature in the following order. -

Insulin Controls Triacylglycerol Synthesis Through Control of Glycerol Metabolism and Despite Increased Lipogenesis
nutrients Article Insulin Controls Triacylglycerol Synthesis through Control of Glycerol Metabolism and Despite Increased Lipogenesis Ana Cecilia Ho-Palma 1,2 , Pau Toro 1, Floriana Rotondo 1, María del Mar Romero 1,3,4, Marià Alemany 1,3,4, Xavier Remesar 1,3,4 and José Antonio Fernández-López 1,3,4,* 1 Department of Biochemistry and Molecular Biomedicine, Faculty of Biology, University of Barcelona, 08028 Barcelona, Spain; [email protected] (A.C.H.-P.); [email protected] (P.T.); fl[email protected] (F.R.); [email protected] (M.d.M.R.); [email protected] (M.A.); [email protected] (X.R.) 2 Faculty of Medicine, Universidad Nacional del Centro del Perú, 12006 Huancayo, Perú 3 Institute of Biomedicine, University of Barcelona, 08028 Barcelona, Spain 4 Centro de Investigación Biomédica en Red Fisiopatología de la Obesidad y Nutrición (CIBER-OBN), 08028 Barcelona, Spain * Correspondence: [email protected]; Tel: +34-93-4021546 Received: 7 February 2019; Accepted: 22 February 2019; Published: 28 February 2019 Abstract: Under normoxic conditions, adipocytes in primary culture convert huge amounts of glucose to lactate and glycerol. This “wasting” of glucose may help to diminish hyperglycemia. Given the importance of insulin in the metabolism, we have studied how it affects adipocyte response to varying glucose levels, and whether the high basal conversion of glucose to 3-carbon fragments is affected by insulin. Rat fat cells were incubated for 24 h in the presence or absence of 175 nM insulin and 3.5, 7, or 14 mM glucose; half of the wells contained 14C-glucose. We analyzed glucose label fate, medium metabolites, and the expression of key genes controlling glucose and lipid metabolism. -

2 Points Chem 465 Biochemistry II Test 1 Spring 2017 Multiple Choice
Name: 2 points Chem 465 Biochemistry II Test 1 Spring 2017 Multiple choice (4 points apiece): 1. Which of the following statements about the oxidative decarboxylation of pyruvate in aerobic conditions in animal cells is correct? A) One of the products of the reactions of the pyruvate dehydrogenase complex is a thioester of acetate. B) The methyl (-CH3) group is eliminated as CO2. C) The process occurs in the cytosolic compartment of the cell. D) The pyruvate dehydrogenase complex uses all of the following as cofactors: NAD+, lipoic acid, pyridoxal phosphate (PLP), and FAD. E) The reaction is so important to energy production that pyruvate dehydrogenase operates at full speed under all conditions. 2. In comparison with the resting state, actively contracting human muscle tissue has a: A) higher concentration of ATP. B) higher rate of lactate formation. C) lower consumption of glucose. D) lower rate of consumption of oxygen E) lower ratio of NADH to NAD+. 3.The metabolic function of the pentose phosphate pathway is: A) act as a source of ADP biosynthesis. B) generate NADPH and pentoses for the biosynthesis of fatty acids and nucleic acids. C) participate in oxidation-reduction reactions during the formation of H2O. D) provide intermediates for the citric acid cycle. E) synthesize phosphorus pentoxide. 4. Gluconeogenesis must use "bypass reactions" to circumvent three reactions in the glycolytic pathway that are highly exergonic and essentially irreversible. Reactions carried out by which three of the enzymes listed must be bypassed in the gluconeogenic pathway? 1) Hexokinase 2) Phosphoglycerate kinase 3) Phosphofructokinase-1 4) Pyruvate kinase 5) Triosephosphate isomerase A) 1, 2, 3 B) 1, 2, 4 C) 1, 4, 5 D) 1, 3, 4 E) 2, 3, 4 5.