Heterozygous Colon Cancer-Associated Mutations of SAMHD1 Have Functional Significance
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Population Size and the Rate of Evolution
Review Population size and the rate of evolution 1,2 1 3 Robert Lanfear , Hanna Kokko , and Adam Eyre-Walker 1 Ecology Evolution and Genetics, Research School of Biology, Australian National University, Canberra, ACT, Australia 2 National Evolutionary Synthesis Center, Durham, NC, USA 3 School of Life Sciences, University of Sussex, Brighton, UK Does evolution proceed faster in larger or smaller popu- mutations occur and the chance that each mutation lations? The relationship between effective population spreads to fixation. size (Ne) and the rate of evolution has consequences for The purpose of this review is to synthesize theoretical our ability to understand and interpret genomic varia- and empirical knowledge of the relationship between tion, and is central to many aspects of evolution and effective population size (Ne, Box 1) and the substitution ecology. Many factors affect the relationship between Ne rate, which we term the Ne–rate relationship (NeRR). A and the rate of evolution, and recent theoretical and positive NeRR implies faster evolution in larger popula- empirical studies have shown some surprising and tions relative to smaller ones, and a negative NeRR implies sometimes counterintuitive results. Some mechanisms the opposite (Figure 1A,B). Although Ne has long been tend to make the relationship positive, others negative, known to be one of the most important factors determining and they can act simultaneously. The relationship also the substitution rate [5–8], several novel predictions and depends on whether one is interested in the rate of observations have emerged in recent years, causing some neutral, adaptive, or deleterious evolution. Here, we reassessment of earlier theory and highlighting some gaps synthesize theoretical and empirical approaches to un- in our understanding. -

Transformations of Lamarckism Vienna Series in Theoretical Biology Gerd B
Transformations of Lamarckism Vienna Series in Theoretical Biology Gerd B. M ü ller, G ü nter P. Wagner, and Werner Callebaut, editors The Evolution of Cognition , edited by Cecilia Heyes and Ludwig Huber, 2000 Origination of Organismal Form: Beyond the Gene in Development and Evolutionary Biology , edited by Gerd B. M ü ller and Stuart A. Newman, 2003 Environment, Development, and Evolution: Toward a Synthesis , edited by Brian K. Hall, Roy D. Pearson, and Gerd B. M ü ller, 2004 Evolution of Communication Systems: A Comparative Approach , edited by D. Kimbrough Oller and Ulrike Griebel, 2004 Modularity: Understanding the Development and Evolution of Natural Complex Systems , edited by Werner Callebaut and Diego Rasskin-Gutman, 2005 Compositional Evolution: The Impact of Sex, Symbiosis, and Modularity on the Gradualist Framework of Evolution , by Richard A. Watson, 2006 Biological Emergences: Evolution by Natural Experiment , by Robert G. B. Reid, 2007 Modeling Biology: Structure, Behaviors, Evolution , edited by Manfred D. Laubichler and Gerd B. M ü ller, 2007 Evolution of Communicative Flexibility: Complexity, Creativity, and Adaptability in Human and Animal Communication , edited by Kimbrough D. Oller and Ulrike Griebel, 2008 Functions in Biological and Artifi cial Worlds: Comparative Philosophical Perspectives , edited by Ulrich Krohs and Peter Kroes, 2009 Cognitive Biology: Evolutionary and Developmental Perspectives on Mind, Brain, and Behavior , edited by Luca Tommasi, Mary A. Peterson, and Lynn Nadel, 2009 Innovation in Cultural Systems: Contributions from Evolutionary Anthropology , edited by Michael J. O ’ Brien and Stephen J. Shennan, 2010 The Major Transitions in Evolution Revisited , edited by Brett Calcott and Kim Sterelny, 2011 Transformations of Lamarckism: From Subtle Fluids to Molecular Biology , edited by Snait B. -

An Unusually Low Microsatellite Mutation Rate in Dictyostelium Discoideum,An Organism with Unusually Abundant Microsatellites
Copyright Ó 2007 by the Genetics Society of America DOI: 10.1534/genetics.107.076067 An Unusually Low Microsatellite Mutation Rate in Dictyostelium discoideum,an Organism With Unusually Abundant Microsatellites Ryan McConnell, Sara Middlemist, Clea Scala, Joan E. Strassmann and David C. Queller1 Department of Ecology and Evolutionary Biology, Rice University, Houston, Texas 77005 Manuscript received May 18, 2007 Accepted for publication September 4, 2007 ABSTRACT The genome of the social amoeba Dictyostelium discoideum is known to have a very high density of microsatellite repeats, including thousands of triplet microsatellite repeats in coding regions that apparently code for long runs of single amino acids. We used a mutation accumulation study to see if unusually high microsatellite mutation rates contribute to this pattern. There was a modest bias toward mutations that increase repeat number, but because upward mutations were smaller than downward ones, this did not lead to a net average increase in size. Longer microsatellites had higher mutation rates than shorter ones, but did not show greater directional bias. The most striking finding is that the overall mutation rate is the lowest reported for microsatellites: 1 3 10À6 for 10 dinucleotide loci and 6 3 10À6 for 52 trinucleotide loci (which were longer). High microsatellite mutation rates therefore do not explain the high incidence of microsatellites. The causal relation may in fact be reversed, with low mutation rates evolving to protect against deleterious fitness effects of mutation at the numerous microsatellites. ICROSATELLITES, also known as simple se- In humans, certain triplet repeats that occur in or M quence repeats, are long stretches of a short near coding regions are subject to expansions that (1–6 bp), tandemly repeated DNA unit, such as the directly cause genetic diseases (Ashley and Warren motif CAA repeated 20 times. -

Adaptive Tuning of Mutation Rates Allows Fast Response to Lethal Stress In
Manuscript 1 Adaptive tuning of mutation rates allows fast response to lethal stress in 2 Escherichia coli 3 4 a a a a a,b 5 Toon Swings , Bram Van den Bergh , Sander Wuyts , Eline Oeyen , Karin Voordeckers , Kevin J. a,b a,c a a,* 6 Verstrepen , Maarten Fauvart , Natalie Verstraeten , Jan Michiels 7 8 a 9 Centre of Microbial and Plant Genetics, KU Leuven - University of Leuven, Kasteelpark Arenberg 20, 10 3001 Leuven, Belgium b 11 VIB Laboratory for Genetics and Genomics, Vlaams Instituut voor Biotechnologie (VIB) Bioincubator 12 Leuven, Gaston Geenslaan 1, 3001 Leuven, Belgium c 13 Smart Systems and Emerging Technologies Unit, imec, Kapeldreef 75, 3001 Leuven, Belgium * 14 To whom correspondence should be addressed: Jan Michiels, Department of Microbial and 2 15 Molecular Systems (M S), Centre of Microbial and Plant Genetics, Kasteelpark Arenberg 20, box 16 2460, 3001 Leuven, Belgium, [email protected], Tel: +32 16 32 96 84 1 Manuscript 17 Abstract 18 19 While specific mutations allow organisms to adapt to stressful environments, most changes in an 20 organism's DNA negatively impact fitness. The mutation rate is therefore strictly regulated and often 21 considered a slowly-evolving parameter. In contrast, we demonstrate an unexpected flexibility in 22 cellular mutation rates as a response to changes in selective pressure. We show that hypermutation 23 independently evolves when different Escherichia coli cultures adapt to high ethanol stress. 24 Furthermore, hypermutator states are transitory and repeatedly alternate with decreases in mutation 25 rate. Specifically, population mutation rates rise when cells experience higher stress and decline again 26 once cells are adapted. -

Estimating Effective Population Size Or Mutation Rate Using the Frequencies of Mutations of Various Classes in a Sample of DNA Sequences
Copyright 0 1994 by the Genetics Society of America Estimating Effective Population Size or Mutation Rate Using the Frequencies of Mutations of Various Classes in a Sample of DNA Sequences Yun-xin FU Center for Demographic and Population Genetics, University of Texas, Houston, Texas 77225 Manuscript received February 18, 1994 Accepted for publication August 27, 1994 ABSTRACT Mutations resulting in segregating sites of a sample of DNA sequences can be classified by size and type and the frequencies of mutations of different sizes and types can be inferred from the sample. A framework for estimating the essential parameter 8 = 4Nu utilizing the frequencies of mutations of various sizes and types is developed in this paper, where N is the effective size of a population and p is mutation rate per sequence per generation.The framework is a combination of coalescent theory, general linear model and Monte-Carlo integration, which leads to two new estimators 6, and 6, as well as a general Watterson’s estimator 6, and a general Tajima’s estimator 6,. The greatest strength of the framework is that it can be used under a variety of population models. The properties of the framework and the four estimators bK, e,,, 6, and 6, are investigated under three important population models: the neutral Wright-Fisher model, the neutral model with recombination and the neutral Wright’s finite-islands model. Under all these models, it is shown that 6, is the best estimator among the four even when recombination rate or migration rate has to be estimated. Under the neutral Wright-Fisher model, it is shown that the new estimator 6,has avariance close to alower bound ofvariances of allunbiased estimators of Owhichsuggests that 6, is a:ery efficient estimator. -

Leeds Thesis Template
Determining the Link Between Genome Integrity and Seed Quality Robbie Michael Gillett Submitted in accordance with the requirements for the degree of Doctor of Philosophy The University of Leeds Faculty of Biological Sciences School of Biology January, 2017 - ii - The candidate confirms that the work submitted is his own and that appropriate credit has been given where reference has been made to the work of others. This copy has been supplied on the understanding that it is copyright material and that no quotation from the thesis may be published without proper acknowledgement. The right of Robbie M. Gillett to be identified as Author of this work has been asserted by him in accordance with the Copyright, Designs and Patents Act 1988. © 2016 The University of Leeds and Robbie Michael Gillett - iii - Acknowledgements I would like to thank my supervisor Dr Christopher West and co-supervisor Dr. Wanda Waterworth for all of their support, expertise and incredible patience and for always being available to help. I would like to thank Aaron Barrett, James Cooper, Valérie Tennant, and all my other friends at university for their help throughout my PhD. Thanks to Vince Agboh and Grace Hoysteed for their combined disjointedness and to Ashley Hines, Daniel Johnston and Darryl Ransom for being a source of entertainment for many years. I would like to thank my international Sona friends, Lindsay Hoffman and Jan Maarten ten Katen. Finally unreserved thanks to my loving parents who supported me throughout the tough times and to my Grandma and Granddad, Jean and Dennis McCarthy, who were always very vocal with their love, support and pride. -

The Microbiota-Produced N-Formyl Peptide Fmlf Promotes Obesity-Induced Glucose
Page 1 of 230 Diabetes Title: The microbiota-produced N-formyl peptide fMLF promotes obesity-induced glucose intolerance Joshua Wollam1, Matthew Riopel1, Yong-Jiang Xu1,2, Andrew M. F. Johnson1, Jachelle M. Ofrecio1, Wei Ying1, Dalila El Ouarrat1, Luisa S. Chan3, Andrew W. Han3, Nadir A. Mahmood3, Caitlin N. Ryan3, Yun Sok Lee1, Jeramie D. Watrous1,2, Mahendra D. Chordia4, Dongfeng Pan4, Mohit Jain1,2, Jerrold M. Olefsky1 * Affiliations: 1 Division of Endocrinology & Metabolism, Department of Medicine, University of California, San Diego, La Jolla, California, USA. 2 Department of Pharmacology, University of California, San Diego, La Jolla, California, USA. 3 Second Genome, Inc., South San Francisco, California, USA. 4 Department of Radiology and Medical Imaging, University of Virginia, Charlottesville, VA, USA. * Correspondence to: 858-534-2230, [email protected] Word Count: 4749 Figures: 6 Supplemental Figures: 11 Supplemental Tables: 5 1 Diabetes Publish Ahead of Print, published online April 22, 2019 Diabetes Page 2 of 230 ABSTRACT The composition of the gastrointestinal (GI) microbiota and associated metabolites changes dramatically with diet and the development of obesity. Although many correlations have been described, specific mechanistic links between these changes and glucose homeostasis remain to be defined. Here we show that blood and intestinal levels of the microbiota-produced N-formyl peptide, formyl-methionyl-leucyl-phenylalanine (fMLF), are elevated in high fat diet (HFD)- induced obese mice. Genetic or pharmacological inhibition of the N-formyl peptide receptor Fpr1 leads to increased insulin levels and improved glucose tolerance, dependent upon glucagon- like peptide-1 (GLP-1). Obese Fpr1-knockout (Fpr1-KO) mice also display an altered microbiome, exemplifying the dynamic relationship between host metabolism and microbiota. -

Style Specifications Thesis
Structural Studies of Salvage Enzymes in Nucleotide Biosynthesis Martin Welin Faculty of Natural Resources and Agricultural Sciences Department of Molecular Biology Uppsala Doctoral thesis Swedish University of Agricultural Sciences Uppsala 2007 Acta Universitatis Agriculturae Sueciae 2007:21 ISSN 1652-6880 ISBN 978-91-576-7320-6 © 2007 Martin Welin, Uppsala Tryck: SLU Service/Repro, Uppsala 2007 Abstract Welin, M., 2007. Structural Studies of Salvage Enzymes in Nucleotide Biosynthesis. Doctoral dissertation. ISSN 1652-6880, ISBN 978-91-576-7320-6 There are two routes to produce deoxyribonucleoside triphosphates (dNTPs) precursors for DNA synthesis, the de novo and the salvage pathways. Deoxyribonucleoside kinases (dNKs) perform the initial phosphorylation of deoxyribonucleosides (dNs). Furthermore, they can act as activators for several medically important nucleoside analogs (NAs) for treatment against cancer or viral infections. Several disorders are characterized by mutations in enzymes involved in the nucleotide biosynthesis, such as Lesch-Nyhan disease that is linked to hypoxanthine guanine phosphoribosyltransferase (HPRT). In this thesis, the structures of human thymidine kinase 1 (TK1), a mycoplasmic deoxyadenosine kinase (Mm-dAK), and phosphoribosyltransferase domain containing 1 (PRTFDC1) are presented. Furthermore, a structural investigation of Drosophila melanogaster dNK (Dm-dNK) N64D mutant was carried out. The obtained structural information reveals the basis for substrate specificity for TKs and the bacterial dAKs. The TK1 revealed a structure different from other known dNK structures, containing an α/β domain similar to the RecA-F1ATPase family, and a lasso-like domain stabilized by a structural zinc. The Mm-dAK structure was similar to its human counterparts, but with some alterations in the proximity of the active site. -

Biochemical Basis for Differential Deoxyadenosine Toxicity To
Proc. Natl. Acad. Sci. USA Vol. 76, No. 5, pp. 2434-2437, May 1979 Medical Sciences Biochemical basis for differential deoxyadenosine toxicity to T and B lymphoblasts: Role for 5'-nucleotidase (deoxyadenosine kinase/deoxyadenylate kinase/immunodeficiency) ROBERT L. WORTMANN, BEVERLY S. MITCHELL, N. LAWRENCE EDWARDS, AND IRVING H. Fox Human Purine Research Center, Departments of Internal Medicine and Biological Chemistry, Clinical Research Center, University of Michigan Medical Center, Ann Arbor, Michigan 48109 Communicated by James B. Wyngaarden, March 7, 1979 ABSTRACT Deoxyadenosine metabolism was investigated tained from Calbiochem. Erythro-9-[3-(2-hydroxynonyl)]- in cultured human cells to elucidate the biochemical basis for adenine (EHNA) was a gift from G. B. Elion of Burroughs the sensitivity of T lymphoblasts and the resistance of B lym- Wellcome (Research Triangle Park, NC). Horse serum was phoblasts to deoxyadenosine toxicity. T lymphoblasts have a 20- to 45-fold greater capacity to synthesize deoxyadenosine nu- obtained from Flow Laboratories (Rockville, MD), and Eagle's cleotides than B lymphoblasts at deoxyadenosine concentrations minimal essential medium was purchased from GIBCO. From of 50-300 ,uM. During the synthesis of dATP, T lymphoblasts Amersham/Searle, [U-14C]deoxyadenosine (505 mCi/mmol), accumulate large quantities of dADP, whereas B lymphoblasts [8-14C]hypoxanthine (52.5 mCi/mmol), and [U-14C]deoxy- do not accumu ate dADP. Enzymes affecting deoxyadenosine adenosine monophosphate (574 mCi/mmol) were purchased; nucleotide synthesis were assayed in these cells. No substantial and, from ICN, [8-14C]adenosine monophosphate (34.4 mCi/ differences were evident in activities of deoxyadenosine kinase (ATP: deoxyadenosine 5'-phosphotransferase, EC 2.7.1.76) or mmol) was purchased (1 Ci = 3.7 X 1010 becquerels). -
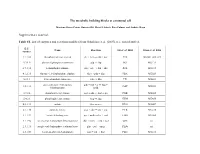
The Metabolic Building Blocks of a Minimal Cell Supplementary
The metabolic building blocks of a minimal cell Mariana Reyes-Prieto, Rosario Gil, Mercè Llabrés, Pere Palmer and Andrés Moya Supplementary material. Table S1. List of enzymes and reactions modified from Gabaldon et. al. (2007). n.i.: non identified. E.C. Name Reaction Gil et. al. 2004 Glass et. al. 2006 number 2.7.1.69 phosphotransferase system glc + pep → g6p + pyr PTS MG041, 069, 429 5.3.1.9 glucose-6-phosphate isomerase g6p ↔ f6p PGI MG111 2.7.1.11 6-phosphofructokinase f6p + atp → fbp + adp PFK MG215 4.1.2.13 fructose-1,6-bisphosphate aldolase fbp ↔ gdp + dhp FBA MG023 5.3.1.1 triose-phosphate isomerase gdp ↔ dhp TPI MG431 glyceraldehyde-3-phosphate gdp + nad + p ↔ bpg + 1.2.1.12 GAP MG301 dehydrogenase nadh 2.7.2.3 phosphoglycerate kinase bpg + adp ↔ 3pg + atp PGK MG300 5.4.2.1 phosphoglycerate mutase 3pg ↔ 2pg GPM MG430 4.2.1.11 enolase 2pg ↔ pep ENO MG407 2.7.1.40 pyruvate kinase pep + adp → pyr + atp PYK MG216 1.1.1.27 lactate dehydrogenase pyr + nadh ↔ lac + nad LDH MG460 1.1.1.94 sn-glycerol-3-phosphate dehydrogenase dhp + nadh → g3p + nad GPS n.i. 2.3.1.15 sn-glycerol-3-phosphate acyltransferase g3p + pal → mag PLSb n.i. 2.3.1.51 1-acyl-sn-glycerol-3-phosphate mag + pal → dag PLSc MG212 acyltransferase 2.7.7.41 phosphatidate cytidyltransferase dag + ctp → cdp-dag + pp CDS MG437 cdp-dag + ser → pser + 2.7.8.8 phosphatidylserine synthase PSS n.i. cmp 4.1.1.65 phosphatidylserine decarboxylase pser → peta PSD n.i. -

The Mutation Rate and Cancer (Tumorigenesis͞evolution͞selection͞mutator Phenotype͞genomic Instability)
Proc. Natl. Acad. Sci. USA Vol. 93, pp. 14800–14803, December 1996 Medical Sciences The mutation rate and cancer (tumorigenesisyevolutionyselectionymutator phenotypeygenomic instability) I. P. M. TOMLINSON,M.R.NOVELLI, AND W. F. BODMER* Cancer Genetics Laboratory, Imperial Cancer Research Fund, 44, Lincoln’s Inn Fields, London WC2A 3PX, United Kingdom Contributed by W. F. Bodmer, September 9, 1996 ABSTRACT The selection of advantageous mutations un- mutations at loci such as those involved in DNA replication or derlies tumorigenesis. The growth of a tumor is therefore a repair. Thus, p53 mutations might lead to a mutator pheno- form of evolution at the somatic level, in which the population type, because the detection of and response to DNA damage is comprised of individual cells within the tumor. Models of are impaired (11). Many cancers have a mutator phenotype tumorigenesis have considered the relative importance of that results from mutations at loci involved in DNA mismatch mutation and selection. We show that selection is more repair (MMR) (12). In the hereditary nonpolyposis colorectal important than an increased mutation rate in the growth of a cancer (HNPCC) syndrome, one MMR allele is mutant in the tumor. Some cancers may acquire a ‘‘mutator phenotype,’’ germ line and the other allele mutates somatically. In sporadic probably leading to faster growth, but mutator phenotypes are cancers—for example, in up to 15% of all colon cancers— not necessary for carcinogenesis. somatic mutations (including allele loss) inactivate both mis- match repair alleles (13). It is widely accepted that tumorigenesis is a form of somatic Despite this evidence, the role of the mutator phenotype (or evolution, an idea which dates back to the writings of Boveri, ‘‘genomic instability’’) in sporadic cancers remains controver- James Murray, Little, and Tyzzer in the early years of this sial. -

The Wayward Dog: Is the Australian Native Dog Or Dingo a Distinct Species?
Zootaxa 4317 (2): 201–224 ISSN 1175-5326 (print edition) http://www.mapress.com/j/zt/ Article ZOOTAXA Copyright © 2017 Magnolia Press ISSN 1175-5334 (online edition) https://doi.org/10.11646/zootaxa.4317.2.1 http://zoobank.org/urn:lsid:zoobank.org:pub:3CD420BC-2AED-4166-85F9-CCA0E4403271 The Wayward Dog: Is the Australian native dog or Dingo a distinct species? STEPHEN M. JACKSON1,2,3,9, COLIN P. GROVES4, PETER J.S. FLEMING5,6, KEN P. APLIN3, MARK D.B. ELDRIDGE7, ANTONIO GONZALEZ4 & KRISTOFER M. HELGEN8 1Animal Biosecurity & Food Safety, NSW Department of Primary Industries, Orange, New South Wales 2800, Australia. 2School of Biological, Earth and Environmental Sciences, University of New South Wales, Sydney, NSW 2052. 3Division of Mammals, National Museum of Natural History, Smithsonian Institution, Washington, DC 20013-7012, USA. E-mail: [email protected] 4School of Archaeology & Anthropology, Australian National University, Canberra, ACT 0200, Australia. E: [email protected]; [email protected] 5Vertebrate Pest Research Unit, Biosecurity NSW, NSW Department of Primary Industries, Orange, New South Wales 2800, Australia. E-mail: [email protected] 6 School of Environmental & Rural Science, University of New England, Armidale, NSW 2351, Australia. 7Australian Museum Research Institute, Australian Museum, 1 William St. Sydney, NSW 2010, Australia. E-mail: [email protected] 8School of Biological Sciences, Environment Institute, and ARC (Australian Research Council) Centre for Australian Biodiversity and Heritage, University of Adelaide, Adelaide, SA 5005, Australia. E-mail: [email protected] 9Corresponding author. E-mail: [email protected] Abstract The taxonomic identity and status of the Australian Dingo has been unsettled and controversial since its initial description in 1792.