Galectin-9 Binds Igm-BCR to Regulate B Cell Signaling
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Human and Mouse CD Marker Handbook Human and Mouse CD Marker Key Markers - Human Key Markers - Mouse
Welcome to More Choice CD Marker Handbook For more information, please visit: Human bdbiosciences.com/eu/go/humancdmarkers Mouse bdbiosciences.com/eu/go/mousecdmarkers Human and Mouse CD Marker Handbook Human and Mouse CD Marker Key Markers - Human Key Markers - Mouse CD3 CD3 CD (cluster of differentiation) molecules are cell surface markers T Cell CD4 CD4 useful for the identification and characterization of leukocytes. The CD CD8 CD8 nomenclature was developed and is maintained through the HLDA (Human Leukocyte Differentiation Antigens) workshop started in 1982. CD45R/B220 CD19 CD19 The goal is to provide standardization of monoclonal antibodies to B Cell CD20 CD22 (B cell activation marker) human antigens across laboratories. To characterize or “workshop” the antibodies, multiple laboratories carry out blind analyses of antibodies. These results independently validate antibody specificity. CD11c CD11c Dendritic Cell CD123 CD123 While the CD nomenclature has been developed for use with human antigens, it is applied to corresponding mouse antigens as well as antigens from other species. However, the mouse and other species NK Cell CD56 CD335 (NKp46) antibodies are not tested by HLDA. Human CD markers were reviewed by the HLDA. New CD markers Stem Cell/ CD34 CD34 were established at the HLDA9 meeting held in Barcelona in 2010. For Precursor hematopoetic stem cell only hematopoetic stem cell only additional information and CD markers please visit www.hcdm.org. Macrophage/ CD14 CD11b/ Mac-1 Monocyte CD33 Ly-71 (F4/80) CD66b Granulocyte CD66b Gr-1/Ly6G Ly6C CD41 CD41 CD61 (Integrin b3) CD61 Platelet CD9 CD62 CD62P (activated platelets) CD235a CD235a Erythrocyte Ter-119 CD146 MECA-32 CD106 CD146 Endothelial Cell CD31 CD62E (activated endothelial cells) Epithelial Cell CD236 CD326 (EPCAM1) For Research Use Only. -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated. -

Manipulating Ig Production in Vivo Through CD180 Stimulation Jay
Manipulating Ig production in vivo through CD180 stimulation Jay Wesley Chaplin A dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy University of Washington 2012 Reading Committee: Edward A. Clark, Chair Keith B. Elkon Thomas R. Hawn Program authorized to offer degree: Molecular and Cellular Biology University of Washington Abstract Manipulating Ig production in vivo through CD180 stimulation Jay Wesley Chaplin Chair of the Supervisory Committee: Professor Edward A. Clark Department of Immunology CD180 is homologous to TLR4 and regulates TLR4 signaling, yet its function is unclear. This thesis reports that injection of anti-CD180 mAb into mice induced rapid polyclonal IgG, with up to 50-fold increases even in immunodeficient mice. Anti-CD180 rapidly increased transitional B cell number in contrast to anti-CD40 which induced primarily FO B cell and myeloid expansion. Combinations of anti-CD180 with MyD88- dependent TLR ligands biased B cell fate toward synergistic proliferation. Thus, CD180 stimulation induces B cell proliferation and differentiation, causing rapid increases in IgG, and integrates MyD88-dependent TLR signals to regulate proliferation and differentiation. This thesis also reports that targeting Ag to CD180 rapidly induces Ag-specific IgG. IgG responses were robust, diverse, and partially T cell-independent, as both CD40- and T cell deficient mice responded after CD180 targeting. IgG production occurred with either hapten- or OVA-conjugated anti-CD180, and was specific for Ag coupled to anti- CD180. Simultaneous BCR and CD180 stimulation enhanced activation compared to either stimulus alone. Adoptive transfer experiments demonstrated that CD180 expression was required on B cells but not on DCs for Ab induction. -

Exploring Functional Pairing Between Surface Glycoconjugates And
Exploring functional pairing between surface PNAS PLUS glycoconjugates and human galectins using programmable glycodendrimersomes Qi Xiaoa, Anna-Kristin Ludwigb, Cecilia Romanòc, Irene Buzzaccheraa,d,e,f, Samuel E. Shermana, Maria Vetroc, Sabine Vértesyb, Herbert Kaltnerb, Ellen H. Reedg, Martin Möllerd,e, Christopher J. Wilsonf, Daniel A. Hammerg,h, Stefan Oscarsonc, Michael L. Kleini,1, Hans-Joachim Gabiusb, and Virgil Perceca,1 aRoy & Diana Vagelos Laboratories, Department of Chemistry, University of Pennsylvania, Philadelphia, PA 19104-6323; bInstitute of Physiological Chemistry, Faculty of Veterinary Medicine, Ludwig-Maximilians-University, 80539 Munich, Germany; cCentre for Synthesis and Chemical Biology, University College Dublin, Dublin 4, Ireland; dDWI − Leibniz Institute for Interactive Materials, RWTH Aachen University, 52074 Aachen, Germany; eInstitute of Technical and Macromolecular Chemistry, RWTH Aachen University, 52074 Aachen, Germany; fNovioSense B.V., 6534 AT Nijmegen, The Netherlands; gDepartment of Bioengineering, University of Pennsylvania, Philadelphia, PA 19104-6321; hDepartment of Chemical and Biomolecular Engineering, University of Pennsylvania, Philadelphia, PA 19104-6391; and iInstitute of Computational Molecular Science, Temple University, Philadelphia, PA 19122 Contributed by Michael L. Klein, January 4, 2018 (sent for review November 16, 2017; reviewed by Timothy J. Deming and Yoshiko Miura) Precise translation of glycan-encoded information into cellular lection process are not yet defined quantitatively. Confronted with activity depends critically on highly specific functional pairing the combination of natural glycan complexity and the result of between glycans and their human lectin counter receptors. Sulfo- evolutionary structure diversification within a lectin family, the glycolipids, such as sulfatides, are important glycolipid components ultimate experimental strategy would seem to require full glycome of the biological membranes found in the nervous and immune and lectin network analysis. -

(RP105) Activates B Cells to Rapidly Produce Polyclonal Ig Via a T Cell and Myd88-Independent Pathway
Anti-CD180 (RP105) Activates B Cells To Rapidly Produce Polyclonal Ig via a T Cell and MyD88-Independent Pathway This information is current as Jay W. Chaplin, Shinji Kasahara, Edward A. Clark and of October 6, 2021. Jeffrey A. Ledbetter J Immunol 2011; 187:4199-4209; Prepublished online 14 September 2011; doi: 10.4049/jimmunol.1100198 http://www.jimmunol.org/content/187/8/4199 Downloaded from Supplementary http://www.jimmunol.org/content/suppl/2011/09/14/jimmunol.110019 Material 8.DC1 http://www.jimmunol.org/ References This article cites 33 articles, 16 of which you can access for free at: http://www.jimmunol.org/content/187/8/4199.full#ref-list-1 Why The JI? Submit online. • Rapid Reviews! 30 days* from submission to initial decision • No Triage! Every submission reviewed by practicing scientists by guest on October 6, 2021 • Fast Publication! 4 weeks from acceptance to publication *average Subscription Information about subscribing to The Journal of Immunology is online at: http://jimmunol.org/subscription Permissions Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Email Alerts Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts The Journal of Immunology is published twice each month by The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2011 by The American Association of Immunologists, Inc. All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. The Journal of Immunology Anti-CD180 (RP105) Activates B Cells To Rapidly Produce Polyclonal Ig via a T Cell and MyD88-Independent Pathway Jay W. -

Supplemental Figures 032819.Pptx
Summary of Supplemental Figures and Tables 1) Supplemental Figure 1. Frequency of phenotypically defined endothelial cells is unchanged in aged mice. 2) Supplemental Figure 2. Aged LSKs have increased myeloid/megakaryocytic bias 3) Supplemental Figure 3. RBCs are decreased and Platelets are increased in peripheral blood of aged mice 4) Supplemental Figure 4. Aged BMME cultures have increased MSC populations. 5) Supplemental Figure 5. Aged BMME cells increase young LSK cell engraftment following competitive transplantation 6) Supplemental Figure 6. Flow cytometry gating strategy for sorting of young and aged murine Mφs 7) Supplemental Figure 7. Flow cytometry gating strategy for sorting of human Mφs 8) Supplemental Figure 8. Axl-/- LT-HSCs have increased cell engraftment following competitive transplantation 9) Supplemental Figure 9. Schematic representation of mechanisms by which age-dependent defects in marrow Mφs induce megakaryocytic bias in HSC. 10) Table S1. List of antibodies used in the flow cytometric and cell sorting analyses described 11) Table S2. List of cell populations analyzed and their respective immunophenotypes 12) Table S3. List of top GO-BP categories enriched for significantly upregulated genes from murine aged vs young marrow macrophages 13) Table S4. List of significant KEGG Pathways enriched for significantly upregulated genes from murine aged vs young marrow macrophages 1 A Sinusoidal Arteriolar BV786 – CD31 Sca1 – PE-Cy7 B C Arteriolar EC Sinusoidal ECs (Lin-, CD45-, CD31+ Sca1+) (CD45-Lin-CD31+Sca1-) 0.08 0.15 0.06 0.10 0.04 0.05 0.02 PercentLiveof Cells PercentLiveof Cells 0.00 0.00 Young Aged Young Aged Supplemental Figure 1. -

The Effect of Bovine Galectin-1, a Conceptus Secretory Protein, on the Endometrial Transcriptome
Graduate Theses, Dissertations, and Problem Reports 2019 The Effect of Bovine Galectin-1, a Conceptus Secretory Protein, on the Endometrial Transcriptome Lindsay Faye Grose West Virginia University, [email protected] Follow this and additional works at: https://researchrepository.wvu.edu/etd Part of the Beef Science Commons, Dairy Science Commons, Genetics Commons, Large or Food Animal and Equine Medicine Commons, and the Other Physiology Commons Recommended Citation Grose, Lindsay Faye, "The Effect of Bovine Galectin-1, a Conceptus Secretory Protein, on the Endometrial Transcriptome" (2019). Graduate Theses, Dissertations, and Problem Reports. 4088. https://researchrepository.wvu.edu/etd/4088 This Thesis is protected by copyright and/or related rights. It has been brought to you by the The Research Repository @ WVU with permission from the rights-holder(s). You are free to use this Thesis in any way that is permitted by the copyright and related rights legislation that applies to your use. For other uses you must obtain permission from the rights-holder(s) directly, unless additional rights are indicated by a Creative Commons license in the record and/ or on the work itself. This Thesis has been accepted for inclusion in WVU Graduate Theses, Dissertations, and Problem Reports collection by an authorized administrator of The Research Repository @ WVU. For more information, please contact [email protected]. The Effect of Bovine Galectin-1, a Conceptus Secretory Protein, on the Endometrial Transcriptome Lindsay Faye Grose Thesis submitted to the Davis College of Agriculture, Natural Resources and Design at West Virginia University in partial fulfillment of the requirements for the degree of Master of Science in Reproductive Physiology Daniel J. -

Recognition of Microbial Glycans by Soluble Human Lectins
Available online at www.sciencedirect.com ScienceDirect Recognition of microbial glycans by soluble human lectins 3 1 1,2 Darryl A Wesener , Amanda Dugan and Laura L Kiessling Human innate immune lectins that recognize microbial glycans implicated in the regulation of microbial colonization and can conduct microbial surveillance and thereby help prevent in protection against infection. Seminal research on the infection. Structural analysis of soluble lectins has provided acute response to bacterial infection led to the identifica- invaluable insight into how these proteins recognize their tion of secreted factors that include C-reactive protein cognate carbohydrate ligands and how this recognition gives (CRP) and mannose-binding lectin (MBL) [1,3]. Both rise to biological function. In this opinion, we cover the CRP and MBL can recognize carbohydrate antigens on structural features of lectins that allow them to mediate the surface of pathogens, including Streptococcus pneumo- microbial recognition, highlighting examples from the collectin, niae and Staphylococcus aureus and then promote comple- Reg protein, galectin, pentraxin, ficolin and intelectin families. ment-mediated opsonization and cell killing [4]. Since These analyses reveal how some lectins (e.g., human intelectin- these initial observations, other lectins have been impli- 1) can recognize glycan epitopes that are remarkably diverse, cated in microbial recognition. Like MBL some of these yet still differentiate between mammalian and microbial proteins are C-type lectins, while others are members of glycans. We additionally discuss strategies to identify lectins the ficolin, pentraxin, galectin, or intelectin families. that recognize microbial glycans and highlight tools that Many of the lectins that function in microbial surveillance facilitate these discovery efforts. -

Human Lectins, Their Carbohydrate Affinities and Where to Find Them
biomolecules Review Human Lectins, Their Carbohydrate Affinities and Where to Review HumanFind Them Lectins, Their Carbohydrate Affinities and Where to FindCláudia ThemD. Raposo 1,*, André B. Canelas 2 and M. Teresa Barros 1 1, 2 1 Cláudia D. Raposo * , Andr1 é LAQVB. Canelas‐Requimte,and Department M. Teresa of Chemistry, Barros NOVA School of Science and Technology, Universidade NOVA de Lisboa, 2829‐516 Caparica, Portugal; [email protected] 12 GlanbiaLAQV-Requimte,‐AgriChemWhey, Department Lisheen of Chemistry, Mine, Killoran, NOVA Moyne, School E41 of ScienceR622 Co. and Tipperary, Technology, Ireland; canelas‐ [email protected] NOVA de Lisboa, 2829-516 Caparica, Portugal; [email protected] 2* Correspondence:Glanbia-AgriChemWhey, [email protected]; Lisheen Mine, Tel.: Killoran, +351‐212948550 Moyne, E41 R622 Tipperary, Ireland; [email protected] * Correspondence: [email protected]; Tel.: +351-212948550 Abstract: Lectins are a class of proteins responsible for several biological roles such as cell‐cell in‐ Abstract:teractions,Lectins signaling are pathways, a class of and proteins several responsible innate immune for several responses biological against roles pathogens. such as Since cell-cell lec‐ interactions,tins are able signalingto bind to pathways, carbohydrates, and several they can innate be a immuneviable target responses for targeted against drug pathogens. delivery Since sys‐ lectinstems. In are fact, able several to bind lectins to carbohydrates, were approved they by canFood be and a viable Drug targetAdministration for targeted for drugthat purpose. delivery systems.Information In fact, about several specific lectins carbohydrate were approved recognition by Food by andlectin Drug receptors Administration was gathered for that herein, purpose. plus Informationthe specific organs about specific where those carbohydrate lectins can recognition be found by within lectin the receptors human was body. -

SUPPLEMENTARY METHODS Cell Culture.-Human Peripheral Blood
SUPPLEMENTARY METHODS Cell culture.-Human peripheral blood mononuclear cells (PBMC) were isolated from buffy coats from normal donors over a Lymphoprep (Nycomed Pharma) gradient. Monocytes were purified from PBMC by magnetic cell sorting using CD14 microbeads (Miltenyi Biotech). Monocytes were cultured at 0.5 x 106 cells/ml for 7 days in RPMI 1640 (standard RPMI, which contains 1 mg/L folic acid) supplemented with 10% fetal calf serum, at 37ºC in a humidified atmosphere with 5% CO2, and containing GM-CSF (1000U/ml) or M-CSF (10 ng/ml, ImmunoTools) to generate GM-CSF- polarized macrophages (GM-MØ) or M-CSF-polarized macrophages (M-MØ). MTX pharmacokinetic studies in RA patients administered 25 mg MTX showed peak plasma levels of 1-2 µM MTX two hours after drug administration, but plasma levels decline to 10-50 nM MTX within 24-48 hours [1, 2]. MTX (50 nM), pemetrexed (PMX, 50 nM), folic acid (FA, 50 nM) [3], thymidine (dT, 10 µM), pifithrin-α (PFT, 25-50 µM), nutlin-3 (10 µM, Sigma-Aldrich) was added once on monocytes together with the indicated cytokine, or on monocytes and 7-day differentiated macrophages for 48h. Gene expression profiling.-For long-term MTX treatment, RNA was isolated from three independent preparations of monocytes either unexposed or exposed to MTX (50 nM) and differentiated to GM-MØ or M-MØ for 7-days. For short-term schedule, RNA was isolated from three independent samples of fully differentiated GM-MØ either unexposed or exposed to MTX (50 nM) for 48h, by using RNeasy Mini kit (QIAGEN). -

Tesis Andrea Flores UAM.Pdf
¿Qué somos y cómo realizaremos eso que somos? Octavio Paz, Nobel Prize of Literature 1980 “Te enterramos ayer. Ayer te enterramos. Te echamos tierra ayer. Quedaste en la tierra ayer. Estás rodeada de tierra desde ayer. Arriba y abajo y a los lados, por tus pies y por tu cabeza, está la tierra desde ayer. Te metimos en la tierra, te tapamos con tierra ayer. Perteneces a la tierra desde ayer. Ayer te enterramos en la tierra, ayer". Jaime Sabines. DEDICO ESTA TESIS A MI ABUE, POR SER MI PIEDRA DE TOQUE Y LA MUJER QUE MÁS HE ADMIRADO Cecilia Caballero Jiménez (1924 -2014) ACKNOWLEDGEMENTS To all my lab -mates in the Department of Physical and Chemical Biology (CIB) , for your company through the years. To Antonio, for your guidance. To the people at the Institute of Chemistry (UNAM), for receiving me as part of your group. To all the people in the Glycopharm Marie Curi e ITN, for your inspiration in my scientific research, I wish to have an everlasting relation with you. To my students, for your admiration and your will to never stop learning. To my best friends , scattered around the globe, I love you . To Sandy and Silvi a in particular. To my siblings, and my nephew and niece, Alan and Talia. For your presence in my life. I love you deeply. And to all that were a part of my path in the obtention of this PhD, para ustedes . OUTLINE OUTLINE List of abbreviations ..……………………………………………………... v Glosary ………..…….………………… …………………………………... vi ABSTRACT ………………………………………………………………. ix RESUMEN ………………………………………………………………... xi INTRODUCTION ……………………………… ………………………... 1 1. CELL GLYCOSYLATION …….………….. -
Type of the Paper (Article
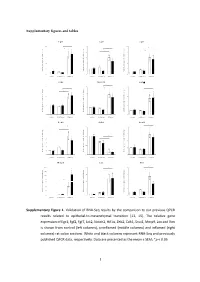
Supplementary figures and tables E g r 1 F g f2 F g f7 1 0 * 5 1 0 * * e e e * g g g * n n n * a a a 8 4 * 8 h h h * c c c d d d * l l l o o o * f f f * n n n o o o 6 3 6 i i i s s s s s s e e e r r r p p p x x x e e e 4 2 4 e e e n n n e e e g g g e e e v v v i i i t t t 2 1 2 a a a l l l e e e R R R 0 0 0 c o n tro l u n in fla m e d in fla m e d c o n tro l u n in fla m e d in fla m e d c o n tro l u n in fla m e d in fla m e d J a k 2 N o tc h 2 H if1 * 3 4 6 * * * e e e g g g n n n a a * * a * h h * h c c c 3 * d d * d l l l * o o o f f 2 f 4 n n n o o o i i i s s s s s s e e e r r 2 r p p p x x x e e e e e e n n n e e 1 e 2 g g g e e 1 e v v v i i i t t t a a a l l l e e e R R R 0 0 0 c o n tro l u n in fla m e d in fla m e d c o n tro l u n in fla m e d in fla m e d c o n tro l u n in fla m e d in fla m e d Z e b 2 C d h 1 S n a i1 * * 7 1 .5 4 * * e e e g g g 6 n n n * a a a * h h h c c c 3 * d d d l l l 5 o o o f f f 1 .0 * n n n * o o o i i i 4 * s s s s s s e e e r r r 2 p p p x x x 3 e e e e e e n n n e e e 0 .5 g g g 2 e e e 1 v v v i i i t t t a a a * l l l e e e 1 * R R R 0 0 .0 0 c o n tro l u n in fla m e d in fla m e d c o n tro l u n in fla m e d in fla m e d c o n tro l u n in fla m e d in fla m e d M m p 9 L o x V im 2 0 0 2 0 8 * * * e e e * g g g 1 5 0 * n n n * a a a * h h h * c c c 1 5 * 6 d d d l l l 1 0 0 o o o f f f n n n o o o i i i 5 0 s s s s s s * e e e r r r 1 0 4 3 0 p p p * x x x e e e * e e e n n n e e e 2 0 g g g e e e 5 2 v v v i i i t t t a a a l l l 1 0 e e e R R R 0 0 0 c o n tro l u n in fla m e d in fla m e d c o n tro l u n in fla m e d in fla m e d c o n tro l u n in fla m e d in fla m e d Supplementary Figure 1.