Wo 2009/045539 A2
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Supplement Ii to the Japanese Pharmacopoeia Fifteenth Edition
SUPPLEMENT II TO THE JAPANESE PHARMACOPOEIA FIFTEENTH EDITION Official From October 1, 2009 English Version THE MINISTRY OF HEALTH, LABOUR AND WELFARE Notice: This English Version of the Japanese Pharmacopoeia is published for the conven- ience of users unfamiliar with the Japanese language. When and if any discrepancy arises between the Japanese original and its English translation, the former is authentic. The Ministry of Health, Labour and Welfare Ministerial Notification No. 425 Pursuant to Paragraph 1, Article 41 of the Pharmaceutical Affairs Law (Law No. 145, 1960), we hereby revise a part of the Japanese Pharmacopoeia (Ministerial Notification No. 285, 2006) as follows*, and the revised Japanese Pharmacopoeia shall come into ef- fect on October 1, 2009. However, in the case of drugs which are listed in the Japanese Pharmacopoeia (hereinafter referred to as “previous Pharmacopoeia”) [limited to those listed in the Japanese Pharmacopoeia whose standards are changed in accordance with this notification (hereinafter referred to as “new Pharmacopoeia”)] and drugs which have been approved as of October 1, 2009 as prescribed under Paragraph 1, Article 14 of the same law [including drugs the Minister of Health, Labour and Welfare specifies (the Ministry of Health and Welfare Ministerial Notification No. 104, 1994) as those ex- empted from marketing approval pursuant to Paragraph 1, Article 14 of the Pharmaceu- tical Affairs Law (hereinafter referred to as “drugs exempted from approval”)], the Name and Standards established in the previous Pharmacopoeia (limited to part of the Name and Standards for the drugs concerned) may be accepted to conform to the Name and Standards established in the new Pharmacopoeia before and on March 31, 2011. -

(12) Patent Application Publication (10) Pub. No.: US 2006/0110428A1 De Juan Et Al
US 200601 10428A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2006/0110428A1 de Juan et al. (43) Pub. Date: May 25, 2006 (54) METHODS AND DEVICES FOR THE Publication Classification TREATMENT OF OCULAR CONDITIONS (51) Int. Cl. (76) Inventors: Eugene de Juan, LaCanada, CA (US); A6F 2/00 (2006.01) Signe E. Varner, Los Angeles, CA (52) U.S. Cl. .............................................................. 424/427 (US); Laurie R. Lawin, New Brighton, MN (US) (57) ABSTRACT Correspondence Address: Featured is a method for instilling one or more bioactive SCOTT PRIBNOW agents into ocular tissue within an eye of a patient for the Kagan Binder, PLLC treatment of an ocular condition, the method comprising Suite 200 concurrently using at least two of the following bioactive 221 Main Street North agent delivery methods (A)-(C): Stillwater, MN 55082 (US) (A) implanting a Sustained release delivery device com (21) Appl. No.: 11/175,850 prising one or more bioactive agents in a posterior region of the eye so that it delivers the one or more (22) Filed: Jul. 5, 2005 bioactive agents into the vitreous humor of the eye; (B) instilling (e.g., injecting or implanting) one or more Related U.S. Application Data bioactive agents Subretinally; and (60) Provisional application No. 60/585,236, filed on Jul. (C) instilling (e.g., injecting or delivering by ocular ion 2, 2004. Provisional application No. 60/669,701, filed tophoresis) one or more bioactive agents into the Vit on Apr. 8, 2005. reous humor of the eye. Patent Application Publication May 25, 2006 Sheet 1 of 22 US 2006/0110428A1 R 2 2 C.6 Fig. -

(12) United States Patent (10) Patent No.: US 6,264,917 B1 Klaveness Et Al
USOO6264,917B1 (12) United States Patent (10) Patent No.: US 6,264,917 B1 Klaveness et al. (45) Date of Patent: Jul. 24, 2001 (54) TARGETED ULTRASOUND CONTRAST 5,733,572 3/1998 Unger et al.. AGENTS 5,780,010 7/1998 Lanza et al. 5,846,517 12/1998 Unger .................................. 424/9.52 (75) Inventors: Jo Klaveness; Pál Rongved; Dagfinn 5,849,727 12/1998 Porter et al. ......................... 514/156 Lovhaug, all of Oslo (NO) 5,910,300 6/1999 Tournier et al. .................... 424/9.34 FOREIGN PATENT DOCUMENTS (73) Assignee: Nycomed Imaging AS, Oslo (NO) 2 145 SOS 4/1994 (CA). (*) Notice: Subject to any disclaimer, the term of this 19 626 530 1/1998 (DE). patent is extended or adjusted under 35 O 727 225 8/1996 (EP). U.S.C. 154(b) by 0 days. WO91/15244 10/1991 (WO). WO 93/20802 10/1993 (WO). WO 94/07539 4/1994 (WO). (21) Appl. No.: 08/958,993 WO 94/28873 12/1994 (WO). WO 94/28874 12/1994 (WO). (22) Filed: Oct. 28, 1997 WO95/03356 2/1995 (WO). WO95/03357 2/1995 (WO). Related U.S. Application Data WO95/07072 3/1995 (WO). (60) Provisional application No. 60/049.264, filed on Jun. 7, WO95/15118 6/1995 (WO). 1997, provisional application No. 60/049,265, filed on Jun. WO 96/39149 12/1996 (WO). 7, 1997, and provisional application No. 60/049.268, filed WO 96/40277 12/1996 (WO). on Jun. 7, 1997. WO 96/40285 12/1996 (WO). (30) Foreign Application Priority Data WO 96/41647 12/1996 (WO). -

Effect of Early Vasopressin Vs Norepinephrine On
Protocol Number: CRO1888 VANISH Vasopressin vs Noradrenaline as Initial therapy in Septic Shock PROTOCOL NUMBER: CRO1888 EudraCT NUMBER: 2011-005363-24 SPONSOR: Imperial College London FUNDER: National Institute for Health Research DEVELOPMENT PHASE: PHASE IV STUDY COORDINATION CENTRE: Imperial Clinical Trials Unit PROTOCOL Version & Date: Version 2.1, 02.08.2013 Property of: Dr Anthony Gordon May not be used, divulged or published without the consent of: Dr Anthony Gordon Confidential Final Version2.1, 02.08.2013 Page 1 of 31 Downloaded From: https://jamanetwork.com/ on 09/29/2021 Protocol Number: CRO1888 Study Management Group Chief Investigator: Dr Anthony Gordon Co-investigators: Dr Stephen Brett Prof Gavin Perkins Prof Deborah Ashby Statistician: Dr Alexina Mason Trial Management: Ms Neeraja Thirunavukkarasu Study Coordination Centre For general queries, supply of trial documentation, and collection of data, please contact: Study Coordinator: Ms Neeraja Thirunavukkarasu Address: ICU – 11N, Charing Cross Hospital, Fulham Palace Road London W6 8RF E-mail: [email protected] Clinical Queries Clinical queries should be directed to Ms Neeraja Thirunavukkarasu who will direct the query to the appropriate person Sponsor Imperial College London is the main research Sponsor for this study. For further information regarding the sponsorship conditions, please contact the Head of Regulatory Compliance at: Joint Research Compliance Office 510, Lab block, 5th Floor Charing Cross Hospital Fulham Palace Road London W6 8RF Tel: 0203 311 0213 Fax: 0203 311 0203 Funder National Institute for Health Research – Research for Patient Benefit and Clinician Scientist award schemes This protocol describes the VANISH study and provides information about procedures for entering participants. -

Vaginal Administration of Contraceptives
Scientia Pharmaceutica Review Vaginal Administration of Contraceptives Esmat Jalalvandi 1,*, Hafez Jafari 2 , Christiani A. Amorim 3 , Denise Freitas Siqueira Petri 4 , Lei Nie 5,* and Amin Shavandi 2,* 1 School of Engineering and Physical Sciences, Heriot-Watt University, Edinburgh EH14 4AS, UK 2 BioMatter Unit, École Polytechnique de Bruxelles, Université Libre de Bruxelles, Avenue F.D. Roosevelt, 50-CP 165/61, 1050 Brussels, Belgium; [email protected] 3 Pôle de Recherche en Gynécologie, Institut de Recherche Expérimentale et Clinique, Université Catholique de Louvain, 1200 Brussels, Belgium; [email protected] 4 Fundamental Chemistry Department, Institute of Chemistry, University of São Paulo, Av. Prof. Lineu Prestes 748, São Paulo 05508-000, Brazil; [email protected] 5 College of Life Sciences, Xinyang Normal University, Xinyang 464000, China * Correspondence: [email protected] (E.J.); [email protected] (L.N.); [email protected] (A.S.); Tel.: +32-2-650-3681 (A.S.) Abstract: While contraceptive drugs have enabled many people to decide when they want to have a baby, more than 100 million unintended pregnancies each year in the world may indicate the contraceptive requirement of many people has not been well addressed yet. The vagina is a well- established and practical route for the delivery of various pharmacological molecules, including contraceptives. This review aims to present an overview of different contraceptive methods focusing on the vaginal route of delivery for contraceptives, including current developments, discussing the potentials and limitations of the modern methods, designs, and how well each method performs for delivering the contraceptives and preventing pregnancy. -

Preferred Drug List
Kansas State Employee ANALGESICS Second Generation cefprozil Health Plan NSAIDs cefuroxime axetil diclofenac sodium delayed-rel Preferred Drug List diflunisal Third Generation etodolac cefdinir 2021 ibuprofen cefixime (SUPRAX) meloxicam nabumetone Erythromycins/Macrolides naproxen sodium tabs azithromycin naproxen tabs clarithromycin oxaprozin clarithromycin ext-rel sulindac erythromycin delayed-rel erythromycin ethylsuccinate NSAIDs, COMBINATIONS erythromycin stearate diclofenac sodium delayed-rel/misoprostol fidaxomicin (DIFICID) Effective 04/01/2021 NSAIDs, TOPICAL Fluoroquinolones diclofenac sodium gel 1% ciprofloxacin For questions or additional information, diclofenac sodium soln levofloxacin access the State of Kansas website at moxifloxacin http://www.kdheks.gov/hcf/sehp or call COX-2 INHIBITORS Penicillins the Kansas State Employees Prescription celecoxib amoxicillin Drug Program at 1-800-294-6324. amoxicillin/clavulanate The Preferred Drug List is subject to change. GOUT amoxicillin/clavulanate ext-rel To locate covered prescriptions online, allopurinol ampicillin access the State of Kansas website at colchicine tabs dicloxacillin http://www.kdheks.gov/hcf/sehp for the probenecid penicillin VK most current drug list. colchicine (MITIGARE) Tetracyclines What is a Preferred Drug List? OPIOID ANALGESICS doxycycline hyclate A Preferred Drug List is a list of safe and buprenorphine transdermal minocycline cost-effective drugs, chosen by a committee codeine/acetaminophen tetracycline of physicians and pharmacists. Drug lists fentanyl -

The Effect of Triamcinolone Acetonide on Collagen Synthesis
THE EFFECT OF TRIAMCINOLONE ACETONIDE ON COLLAGEN SYNTHESIS BY HUMAN AND MOUSE DERMAL FIBROBLASTS IN CELL CULTURE by ELAINE MEI LI|TAN B. Sc., The University of British Columbia, 1976 A THESIS SUBMITTED IN PARTIAL FULFILMENT OF THE REQUIREMENTS FOR THE DEGREE OF MASTER OF SCIENCE in THE FACULTY OF GRADUATE STUDIES (Division of Pharmaceutics and Biopharmaceutics) We accept this thesis as conforming to the required standard THE UNIVERSITY OF BRITISH COLUMBIA JANUARY 1980 (?) Elaine Mei Li Jan, 1980 ii In presenting this thesis in partial fulfilment of the requirements fo an advanced degree at the University of British Columbia, I agree that the Library shall make it freely available for reference and study. I further agree that permission for extensive copying of this thesis for scholarly purposes may be granted by the Head of my Department or by his representatives. It is understood that copying or publication of this thesis for financial gain shall not be allowed without my written permission. Department of The University of British Columbia 2075 Wesbrook Place Vancouver, Canada V6T 1WS Date SO, tffd - iii - ABSTRACT Glucocorticoids are known to affect metabolic activities of cells. The mechanism of glucocorticoid actions in adult human dermal and mouse L-929 fibroblasts have yet to be fully ascertained. This study endeavors to examine the effects of one glucocorticoid, triamcinolone acetonide, on cellular proliferation and collagen synthesis and to compare such effects in the human and mouse cell lines. Cellular proliferation and collagen synthesis are analyzed and quantitated by cell counts and selective digestion of the protein by bacterial collagenase, respectively. -

Pharmaceutical Compoundingand Dispensing, Second
Pharmaceutical Compounding and Dispensing Pharmaceutical Compounding and Dispensing SECOND EDITION John F Marriott BSc, PhD, MRPharmS, FHEA Professor of Clinical Pharmacy Aston University School of Pharmacy, UK Keith A Wilson BSc, PhD, FRPharmS Head of School Aston University School of Pharmacy, UK Christopher A Langley BSc, PhD, MRPharmS, MRSC, FHEA Senior Lecturer in Pharmacy Practice Aston University School of Pharmacy, UK Dawn Belcher BPharm, MRPharmS, FHEA Teaching Fellow, Pharmacy Practice Aston University School of Pharmacy, UK Published by the Pharmaceutical Press 1 Lambeth High Street, London SE1 7JN, UK 1559 St Paul Avenue, Gurnee, IL 60031, USA Ó Pharmaceutical Press 2010 is a trade mark of Pharmaceutical Press Pharmaceutical Press is the publishing division of the Royal Pharmaceutical Society of Great Britain First edition published 2006 Second edition published 2010 Typeset by Thomson Digital, Noida, India Printed in Great Britain by TJ International, Padstow, Cornwall ISBN 978 0 85369 912 5 All rights reserved. No part of this publication may be reproduced, stored in a retrieval system, or transmitted in any form or by any means, without the prior written permission of the copyright holder. The publisher makes no representation, express or implied, with regard to the accuracy of the information contained in this book and cannot accept any legal responsibility or liability for any errors or omissions that may be made. The right of John F Marriott, Keith A Wilson, Christopher A Langley and Dawn Belcher to be identified as the author of this work has been asserted by them in accordance with the Copyright, Designs and Patents Act, 1988. -

Federal Register / Vol. 60, No. 80 / Wednesday, April 26, 1995 / Notices DIX to the HTSUS—Continued
20558 Federal Register / Vol. 60, No. 80 / Wednesday, April 26, 1995 / Notices DEPARMENT OF THE TREASURY Services, U.S. Customs Service, 1301 TABLE 1.ÐPHARMACEUTICAL APPEN- Constitution Avenue NW, Washington, DIX TO THE HTSUSÐContinued Customs Service D.C. 20229 at (202) 927±1060. CAS No. Pharmaceutical [T.D. 95±33] Dated: April 14, 1995. 52±78±8 ..................... NORETHANDROLONE. A. W. Tennant, 52±86±8 ..................... HALOPERIDOL. Pharmaceutical Tables 1 and 3 of the Director, Office of Laboratories and Scientific 52±88±0 ..................... ATROPINE METHONITRATE. HTSUS 52±90±4 ..................... CYSTEINE. Services. 53±03±2 ..................... PREDNISONE. 53±06±5 ..................... CORTISONE. AGENCY: Customs Service, Department TABLE 1.ÐPHARMACEUTICAL 53±10±1 ..................... HYDROXYDIONE SODIUM SUCCI- of the Treasury. NATE. APPENDIX TO THE HTSUS 53±16±7 ..................... ESTRONE. ACTION: Listing of the products found in 53±18±9 ..................... BIETASERPINE. Table 1 and Table 3 of the CAS No. Pharmaceutical 53±19±0 ..................... MITOTANE. 53±31±6 ..................... MEDIBAZINE. Pharmaceutical Appendix to the N/A ............................. ACTAGARDIN. 53±33±8 ..................... PARAMETHASONE. Harmonized Tariff Schedule of the N/A ............................. ARDACIN. 53±34±9 ..................... FLUPREDNISOLONE. N/A ............................. BICIROMAB. 53±39±4 ..................... OXANDROLONE. United States of America in Chemical N/A ............................. CELUCLORAL. 53±43±0 -
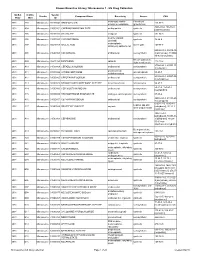
Known Bioactive Library: Microsource 1 - US Drug Collection
Known Bioactive Library: Microsource 1 - US Drug Collection ICCB-L ICCB-L Vendor Vendor Compound Name Bioactivity Source CAS Plate Well ID antifungal, inhibits Penicillium 2091 A03 Microsource 00200046 GRISEOFULVIN 126-07-8 mitosis in metaphase griseofulvum 3505-38-2, 486-16-8 2091 A04 Microsource 01500161 CARBINOXAMINE MALEATE antihistaminic synthetic [carbinoxamine] 2091 A05 Microsource 00200331 SALSALATE analgesic synthetic 552-94-3 muscle relaxant 2091 A06 Microsource 01500162 CARISOPRODOL synthetic 78-44-4 (skeletal) antineoplastic, 2091 A07 Microsource 00210369 GALLIC ACID insect galls 149-91-7 astringent, antibacterial 66592-87-8, 50370-12- 2091 A08 Microsource 01500163 CEFADROXIL antibacterial semisynthetic 2 [anhydrous], 119922- 89-9 [hemihydrate] Rheum palmatum, 2091 A09 Microsource 00211468 DANTHRON cathartic 117-10-2 Xyris semifuscata 27164-46-1, 25953-19- 2091 A10 Microsource 01500164 CEFAZOLIN SODIUM antibacterial semisynthetic 9 [cefazolin] glucocorticoid, 2091 A11 Microsource 00300024 HYDROCORTISONE adrenal glands 50-23-7 antiinflammatory 64485-93-4, 63527-52- 2091 A12 Microsource 01500165 CEFOTAXIME SODIUM antibacterial semisynthetic 6 [cefotaxime] 2091 A13 Microsource 00300029 DESOXYCORTICOSTERONE ACETATE mineralocorticoid adrenocortex 56-47-3 58-71-9, 153-61-7 2091 A14 Microsource 01500166 CEPHALOTHIN SODIUM antibacterial semisynthetic [cephalothin] 2091 A15 Microsource 00300034 TESTOSTERONE PROPIONATE androgen, antineoplastic semisynthetic 57-85-2 24356-60-3, 21593-23- 2091 A16 Microsource 01500167 CEPHAPIRIN SODIUM -

Hydrocortisone Sodium Phosphate OH the Individual Container May Be Too Small to Bear All the Appro- Priate Labelling Information
Formocortal/Hydrocortisone 1535 Halcinonide (BAN, USAN, rINN) ⊗ Preparations Hydrocortisone Butyrate (BANM, USAN, rINNM) ⊗ Alcinonide; Halcinonid; Halcinónida; Halcinonidum; Halsinonid; Proprietary Preparations (details are given in Part 3) Butirato de hidrocortisona; Cortisol Butyrate; Hidrokortizon Halsinonidi; SQ-18566. 21-Chloro-9α-fluoro-11β-hydroxy- Austria: Sicorten; Belg.: Sicorten†; Ger.: Sicorten†; Hong Kong: Si- Bütirat; Hydrocortisone, Butyrate d’; Hydrocortisoni Butiras. Hy- corten; Neth.: Sicorten†; Port.: Sicorten†; Spain: Sicorten; Switz.: Si- 16α,17α-isopropylidenedioxypregn-4-ene-3,20-dione. corten; Turk.: Sicorten. drocortisone 17α-butyrate. Гальцинонид Multi-ingredient: Ger.: Sicorten Plus; Hong Kong: Sicorten Plus†; Isra- Гидрокортизона Бутират el: Sicorten Plus†; Port.: Sicorten Plus†; Spain: Sicorten Plus; Switz.: Si- C24H32ClFO5 = 455.0. corten Plus; Venez.: Sicorten Plus†. C25H36O6 = 432.5. CAS — 3093-35-4. CAS — 13609-67-1. ATC — D07AD02. ATC — D07AB02. ATC Vet — QD07AD02. ATC Vet — QD07AB02. Hydrocortisone (BAN, rINN) ⊗ Pharmacopoeias. In Chin., Jpn, and US. Anti-inflammatory Hormone; Compound F; Cortisol; Hidrocor- O CH3 USP 31 (Hydrocortisone Butyrate). A white to practically Cl tisona; Hidrokortizon; Hidrokortizonas; Hydrocortisonum; Hy- white, practically odourless crystalline powder. Practically insol- CH H3C O 3 drokortison; Hydrokortisoni; Hydrokortyzon; 17-Hydroxycorti- uble in water; soluble in alcohol, in acetone, and in methyl alco- HO O costerone; NSC-10483. 11β,17α,21-Trihydroxypregn-4-ene- hol; freely soluble in chloroform; slightly soluble in ether. 3,20-dione. H3CH Гидрокортизон Hydrocortisone Cipionate (BANM, rINNM) ⊗ C H O = 362.5. FH 21 30 5 Cipionato de hidrocortisona; Cortisol Cypionate; Hydrocorti- CAS — 50-23-7. O sone, Cipionate d’; Hydrocortisone Cyclopentylpropionate; Hy- ATC — A01AC03; A07EA02; C05AA01; D07AA02; drocortisone Cypionate; Hydrocortisoni Cipionas. Hydrocorti- H02AB09; S01BA02; S02BA01. -

Blue Choice Essential Value Formulary July 2021
ISi Arkansas • '9. BlueCross BlueShield Arkansas Blue Cross and Blue Shield Blue Choice Formulary Effective 10/01/2021 INTRODUCTION ........................................................................................................................................................................................................................ 4 PREFACE ................................................................................................................................................................................................................................... 4 PHARMACY AND THERAPEUTICS (P&T) COMMITTEE ........................................................................................................................................................ 4 DRUG LIST PRODUCT DESCRIPTIONS.................................................................................................................................................................................. 4 GENERIC SUBSTITUTION ........................................................................................................................................................................................................ 5 SPECIALTY MEDICATIONS ..................................................................................................................................................................................................... 5 PLAN DESIGN ..........................................................................................................................................................................................................................