Ph-Dependent NMDA Receptor Antagonists Emory Tech ID: 01021
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Stems for Nonproprietary Drug Names
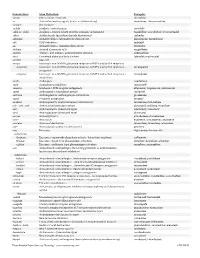
USAN STEM LIST STEM DEFINITION EXAMPLES -abine (see -arabine, -citabine) -ac anti-inflammatory agents (acetic acid derivatives) bromfenac dexpemedolac -acetam (see -racetam) -adol or analgesics (mixed opiate receptor agonists/ tazadolene -adol- antagonists) spiradolene levonantradol -adox antibacterials (quinoline dioxide derivatives) carbadox -afenone antiarrhythmics (propafenone derivatives) alprafenone diprafenonex -afil PDE5 inhibitors tadalafil -aj- antiarrhythmics (ajmaline derivatives) lorajmine -aldrate antacid aluminum salts magaldrate -algron alpha1 - and alpha2 - adrenoreceptor agonists dabuzalgron -alol combined alpha and beta blockers labetalol medroxalol -amidis antimyloidotics tafamidis -amivir (see -vir) -ampa ionotropic non-NMDA glutamate receptors (AMPA and/or KA receptors) subgroup: -ampanel antagonists becampanel -ampator modulators forampator -anib angiogenesis inhibitors pegaptanib cediranib 1 subgroup: -siranib siRNA bevasiranib -andr- androgens nandrolone -anserin serotonin 5-HT2 receptor antagonists altanserin tropanserin adatanserin -antel anthelmintics (undefined group) carbantel subgroup: -quantel 2-deoxoparaherquamide A derivatives derquantel -antrone antineoplastics; anthraquinone derivatives pixantrone -apsel P-selectin antagonists torapsel -arabine antineoplastics (arabinofuranosyl derivatives) fazarabine fludarabine aril-, -aril, -aril- antiviral (arildone derivatives) pleconaril arildone fosarilate -arit antirheumatics (lobenzarit type) lobenzarit clobuzarit -arol anticoagulants (dicumarol type) dicumarol -

United States Patent (10 ) Patent No.: US 10,660,887 B2 Javitt (45 ) Date of Patent: *May 26 , 2020
US010660887B2 United States Patent (10 ) Patent No.: US 10,660,887 B2 Javitt (45 ) Date of Patent : *May 26 , 2020 (54 ) COMPOSITION AND METHOD FOR (56 ) References Cited TREATMENT OF DEPRESSION AND PSYCHOSIS IN HUMANS U.S. PATENT DOCUMENTS 6,228,875 B1 5/2001 Tsai et al. ( 71 ) Applicant : Glytech , LLC , Ft. Lee , NJ (US ) 2004/0157926 A1 * 8/2004 Heresco - Levy A61K 31/198 514/561 (72 ) Inventor: Daniel C. Javitt , Ft. Lee , NJ (US ) 2005/0261340 Al 11/2005 Weiner 2006/0204486 Al 9/2006 Pyke et al . 2008/0194631 Al 8/2008 Trovero et al. ( 73 ) Assignee : GLYTECH , LLC , Ft. Lee , NJ (US ) 2008/0194698 A1 8/2008 Hermanussen et al . 2010/0069399 A1 * 3/2010 Gant CO7D 401/12 ( * ) Notice : Subject to any disclaimer, the term of this 514 / 253.07 patent is extended or adjusted under 35 2010/0216805 Al 8/2010 Barlow 2011/0207776 Al 8/2011 Buntinx U.S.C. 154 ( b ) by 95 days . 2011/0237602 A1 9/2011 Meltzer This patent is subject to a terminal dis 2011/0306586 Al 12/2011 Khan claimer . 2012/0041026 A1 2/2012 Waizumi ( 21 ) Appl. No.: 15 /650,912 FOREIGN PATENT DOCUMENTS CN 101090721 12/2007 ( 22 ) Filed : Jul. 16 , 17 KR 2007 0017136 2/2007 WO 2005/065308 7/2005 (65 ) Prior Publication Data WO 2005/079756 9/2005 WO 2011044089 4/2011 US 2017/0312275 A1 Nov. 2 , 2017 WO 2012/104852 8/2012 WO 2005/000216 9/2013 WO 2013138322 9/2013 Related U.S. Application Data (63 ) Continuation of application No. -

Pharmaceutical Appendix to the Tariff Schedule 2
Harmonized Tariff Schedule of the United States (2007) (Rev. 2) Annotated for Statistical Reporting Purposes PHARMACEUTICAL APPENDIX TO THE HARMONIZED TARIFF SCHEDULE Harmonized Tariff Schedule of the United States (2007) (Rev. 2) Annotated for Statistical Reporting Purposes PHARMACEUTICAL APPENDIX TO THE TARIFF SCHEDULE 2 Table 1. This table enumerates products described by International Non-proprietary Names (INN) which shall be entered free of duty under general note 13 to the tariff schedule. The Chemical Abstracts Service (CAS) registry numbers also set forth in this table are included to assist in the identification of the products concerned. For purposes of the tariff schedule, any references to a product enumerated in this table includes such product by whatever name known. ABACAVIR 136470-78-5 ACIDUM LIDADRONICUM 63132-38-7 ABAFUNGIN 129639-79-8 ACIDUM SALCAPROZICUM 183990-46-7 ABAMECTIN 65195-55-3 ACIDUM SALCLOBUZICUM 387825-03-8 ABANOQUIL 90402-40-7 ACIFRAN 72420-38-3 ABAPERIDONUM 183849-43-6 ACIPIMOX 51037-30-0 ABARELIX 183552-38-7 ACITAZANOLAST 114607-46-4 ABATACEPTUM 332348-12-6 ACITEMATE 101197-99-3 ABCIXIMAB 143653-53-6 ACITRETIN 55079-83-9 ABECARNIL 111841-85-1 ACIVICIN 42228-92-2 ABETIMUSUM 167362-48-3 ACLANTATE 39633-62-0 ABIRATERONE 154229-19-3 ACLARUBICIN 57576-44-0 ABITESARTAN 137882-98-5 ACLATONIUM NAPADISILATE 55077-30-0 ABLUKAST 96566-25-5 ACODAZOLE 79152-85-5 ABRINEURINUM 178535-93-8 ACOLBIFENUM 182167-02-8 ABUNIDAZOLE 91017-58-2 ACONIAZIDE 13410-86-1 ACADESINE 2627-69-2 ACOTIAMIDUM 185106-16-5 ACAMPROSATE 77337-76-9 -
Downloaded from Survive Nursing | Survivenursing.Com V20110426
Generic Stem Stem Definition Examples -abine (see -arabine, -citabine) decitabine -ac Anti-inflammatory agents (acetic acid derivatives) bromfenac; dexpemedolac -acetam See -racetam -actide Synthetic corticotropins seractide -adol or -aldol- Analgesics (mixed opiate receptor agonists/ antagonists) tazadolene; spiradolene; levonantradol -adox Antibacterials (quinoline dioxide derivatives) carbadox -afenone Antiarrhythmics (propafenone derivatives) alprafenone; diprafenone -afil PDE5 inhibitors tadalafil -aj- Antiarrhythmics (ajmaline derivatives) lorajmine -aldrate Antacid aluminum salts magaldrate -algron Alpha1 - and alpha2 - adrenoreceptor agonists dabuzalgron -alol Combined alpha and beta blockers labetalol; medroxalol -amivir (see -vir) -ampa Ionotropic non-NMDA glutamate receptors (AMPA and/or KA receptors) -ampanel Ionotropic non-NMDA glutamate receptors (AMPA and/or KA receptors) ; becampanel antagonists -ampator Ionotropic non-NMDA glutamate receptors (AMPA and/or KA receptors) ; forampator modulators -andr- Androgens nandrolone -anib Angiogenesis inhibitors semaxanib -anserin Serotonin 5-HT2 receptor antagonists altanserin; tropanserin; adatanserin -antel Anthelmintics (undefined group) carbantel -antrone Antineoplastics; anthraquinone derivatives pixantrone -apsel P-selectin antagonists torapsel -arabine Antineoplastics (arabinofuranosyl derivatives) fazarabine; fludarabine aril-, -aril, -aril- Antiviral (arildone derivatives) pleconaril; arildone; fosarilate -arit Antirheumatics (lobenzarit type) lobenzarit; clobuzarit -arol -

Understanding History, and Not Repeating It. Neuroprotection For
Clinical Neurology and Neurosurgery 129 (2015) 1–9 Contents lists available at ScienceDirect Clinical Neurology and Neurosurgery jo urnal homepage: www.elsevier.com/locate/clineuro Review Understanding history, and not repeating it. Neuroprotection for acute ischemic stroke: From review to preview a a b b,c a,b,c,d,∗ Stephen Grupke , Jason Hall , Michael Dobbs , Gregory J. Bix , Justin F. Fraser a Department of Neurosurgery, University of Kentucky, Lexington, USA b Department of Neurology, University of Kentucky, Lexington, USA c Department of Anatomy and Neurobiology, University of Kentucky, Lexington, USA d Department of Radiology, University of Kentucky, Lexington, USA a r t i c l e i n f o a b s t r a c t Article history: Background: Neuroprotection for ischemic stroke is a growing field, built upon the elucidation of the Received 17 April 2014 biochemical pathways of ischemia first studied in the 1970s. Beginning in the early 1990s, means by Received in revised form 7 November 2014 which to pharmacologically intervene and counteract these pathways have been sought, though with Accepted 13 November 2014 little clinical success. Through a comprehensive review of translations from laboratory to clinic, we aim Available online 3 December 2014 to evaluate individual mechanisms of action, while highlighting potential barriers to success that will guide future research. Keywords: Methods: The MEDLINE database and The Internet Stroke Center clinical trials registry were queried Acute stroke Angiography for trials involving the use of neuroprotective agents in acute ischemic stroke in human subjects. For the purpose of the review, neuroprotective agents refer to medications used to preserve or protect the Brain ischemia Drug trials potentially ischemic tissue after an acute stroke, excluding treatments designed to re-establish perfusion. -

Ep 2932971 A1
(19) TZZ ¥ __T (11) EP 2 932 971 A1 (12) EUROPEAN PATENT APPLICATION (43) Date of publication: (51) Int Cl.: 21.10.2015 Bulletin 2015/43 A61K 31/54 (2006.01) A61K 31/445 (2006.01) A61K 9/08 (2006.01) A61K 9/51 (2006.01) (2006.01) (21) Application number: 15000954.6 A61L 31/00 (22) Date of filing: 06.03.2006 (84) Designated Contracting States: • MCCORMACK, Stephen, Joseph AT BE BG CH CY CZ DE DK EE ES FI FR GB GR Claremont, CA 91711 (US) HU IE IS IT LI LT LU LV MC NL PL PT RO SE SI • SCHLOSS, John, Vinton SK TR Valencia, CA 91350 (US) • NAGY, Anna Imola (30) Priority: 04.03.2005 US 658207 P Saugus, CA 91350 (US) • PANANEN, Jacob, E. (62) Document number(s) of the earlier application(s) in 306 Los Angeles, CA 90042 (US) accordance with Art. 76 EPC: 06736872.0 / 1 861 104 (74) Representative: Ali, Suleman et al Avidity IP Limited (71) Applicant: Otonomy, Inc. Broers Building, Hauser Forum San Diego, CA 92121 (US) 21 JJ Thomson Avenue Cambridge CB3 0FA (GB) (72) Inventors: • LOBL, Thomas, Jay Remarks: Valencia, This application was filed on 09-04-2015 as a CA 91355-1995 (US) divisional application to the application mentioned under INID code 62. (54) KETAMINE FORMULATIONS (57) Formulations of ketamine for administration to the inner or middle ear. EP 2 932 971 A1 Printed by Jouve, 75001 PARIS (FR) EP 2 932 971 A1 Description [0001] This application claims the benefit of Serial No. 60/658,207 filed March 4, 2005. -

WO 2011/089216 Al
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date t 28 July 2011 (28.07.2011) WO 2011/089216 Al (51) International Patent Classification: (81) Designated States (unless otherwise indicated, for every A61K 47/48 (2006.01) C07K 1/13 (2006.01) kind of national protection available): AE, AG, AL, AM, C07K 1/1 07 (2006.01) AO, AT, AU, AZ, BA, BB, BG, BH, BR, BW, BY, BZ, CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, DO, (21) Number: International Application DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, PCT/EP201 1/050821 HN, HR, HU, ID, J , IN, IS, JP, KE, KG, KM, KN, KP, (22) International Filing Date: KR, KZ, LA, LC, LK, LR, LS, LT, LU, LY, MA, MD, 2 1 January 201 1 (21 .01 .201 1) ME, MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, NO, NZ, OM, PE, PG, PH, PL, PT, RO, RS, RU, SC, SD, (25) Filing Language: English SE, SG, SK, SL, SM, ST, SV, SY, TH, TJ, TM, TN, TR, (26) Publication Language: English TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, ZW. (30) Priority Data: (84) Designated States (unless otherwise indicated, for every 1015 1465. 1 22 January 2010 (22.01 .2010) EP kind of regional protection available): ARIPO (BW, GH, GM, KE, LR, LS, MW, MZ, NA, SD, SL, SZ, TZ, UG, (71) Applicant (for all designated States except US): AS- ZM, ZW), Eurasian (AM, AZ, BY, KG, KZ, MD, RU, TJ, CENDIS PHARMA AS [DK/DK]; Tuborg Boulevard TM), European (AL, AT, BE, BG, CH, CY, CZ, DE, DK, 12, DK-2900 Hellerup (DK). -

International Nonproprietary Names for Pharmaceutical Substances
WHO DRUG INFORMATION VOLUME 12 • NUMBER 1B • 1998 RECOMMENDED INN LIST 39 INTERNATIONAL NONPROPRIETARY NAMES FOR PHARMACEUTICAL SUBSTANCES WORLD HEALTH ORGANIZATION • GENEVA Volume 12, Number 1, 1998 World Health Organization, Geneva WHO Drug Information Contents Scopolamine patch to prevent peri-operative nausea 15 Pemoline withdrawn 15 General Policy Issues Melatonin on prescription only 16 Dangers of purchasing medical products Chlormezanone withdrawn 16 through the Internet 1 Alcohol warning on over-the-counter pain medications 16 Nefazodone: adverse reaction reports 16 Personal Perspectives Daclizumab: first monoclonal antibody Fifty years of drug regulation: solid against transplant rejection 16 accomplishments and an important future 3 Reboxetine not for use in elderly 17 Counterfeit antimalarials detected 17 Reports on Individual Drugs Hormone replacement therapy and breast cancer 6 ATC/DDD Classification Mefloquine effectiveness impaired by high (final) 18 withdrawal rates 7 Zopiclone and zolpidem hypnotics and drug ATC/DDD Classification dependence 8 (temporary) 20 General Information Essential Drugs Cardiovascular disease and hormonal contraceptives: latest results 9 WHO Model List (revised in December 1997) 22 Current availability of vaccines for diarrhoeal diseases and typhoid 10 Recent Publications and Regulatory Matters Documents Extended release oral dosage forms: Troglitazone and liver injury 13 guidance for applicants 36 Phenolphthalein products withdrawn 13 United Nations Consolidated List 36 Serotonin re-uptake inhibitors and neonatal Manual on quality assurance 36 withdrawal symptoms 14 Medicinal claims and food products 37 Raloxifene for the prevention of osteoporosis 14 Paediatric prescribing information 37 Mibefradil: low heart rate and severe interactions reported 14 New anti-obesity drug approved 15 Recommended International Nonproprietary Names: List 39 39 i WHO Drug Information Vol. -

(12) Patent Application Publication (10) Pub. No.: US 2010/0184806 A1 Barlow Et Al
US 20100184806A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2010/0184806 A1 Barlow et al. (43) Pub. Date: Jul. 22, 2010 (54) MODULATION OF NEUROGENESIS BY PPAR (60) Provisional application No. 60/826,206, filed on Sep. AGENTS 19, 2006. (75) Inventors: Carrolee Barlow, Del Mar, CA (US); Todd Carter, San Diego, CA Publication Classification (US); Andrew Morse, San Diego, (51) Int. Cl. CA (US); Kai Treuner, San Diego, A6II 3/4433 (2006.01) CA (US); Kym Lorrain, San A6II 3/4439 (2006.01) Diego, CA (US) A6IP 25/00 (2006.01) A6IP 25/28 (2006.01) Correspondence Address: A6IP 25/18 (2006.01) SUGHRUE MION, PLLC A6IP 25/22 (2006.01) 2100 PENNSYLVANIA AVENUE, N.W., SUITE 8OO (52) U.S. Cl. ......................................... 514/337; 514/342 WASHINGTON, DC 20037 (US) (57) ABSTRACT (73) Assignee: BrainCells, Inc., San Diego, CA (US) The instant disclosure describes methods for treating diseases and conditions of the central and peripheral nervous system (21) Appl. No.: 12/690,915 including by stimulating or increasing neurogenesis, neuro proliferation, and/or neurodifferentiation. The disclosure (22) Filed: Jan. 20, 2010 includes compositions and methods based on use of a peroxi some proliferator-activated receptor (PPAR) agent, option Related U.S. Application Data ally in combination with one or more neurogenic agents, to (63) Continuation-in-part of application No. 1 1/857,221, stimulate or increase a neurogenic response and/or to treat a filed on Sep. 18, 2007. nervous system disease or disorder. Patent Application Publication Jul. 22, 2010 Sheet 1 of 9 US 2010/O184806 A1 Figure 1: Human Neurogenesis Assay Ciprofibrate Neuronal Differentiation (TUJ1) 100 8090 Ciprofibrates 10-8.5 10-8.0 10-7.5 10-7.0 10-6.5 10-6.0 10-5.5 10-5.0 10-4.5 Conc(M) Patent Application Publication Jul. -

Harmonized Tariff Schedule of the United States (2004) -- Supplement 1 Annotated for Statistical Reporting Purposes
Harmonized Tariff Schedule of the United States (2004) -- Supplement 1 Annotated for Statistical Reporting Purposes PHARMACEUTICAL APPENDIX TO THE HARMONIZED TARIFF SCHEDULE Harmonized Tariff Schedule of the United States (2004) -- Supplement 1 Annotated for Statistical Reporting Purposes PHARMACEUTICAL APPENDIX TO THE TARIFF SCHEDULE 2 Table 1. This table enumerates products described by International Non-proprietary Names (INN) which shall be entered free of duty under general note 13 to the tariff schedule. The Chemical Abstracts Service (CAS) registry numbers also set forth in this table are included to assist in the identification of the products concerned. For purposes of the tariff schedule, any references to a product enumerated in this table includes such product by whatever name known. Product CAS No. Product CAS No. ABACAVIR 136470-78-5 ACEXAMIC ACID 57-08-9 ABAFUNGIN 129639-79-8 ACICLOVIR 59277-89-3 ABAMECTIN 65195-55-3 ACIFRAN 72420-38-3 ABANOQUIL 90402-40-7 ACIPIMOX 51037-30-0 ABARELIX 183552-38-7 ACITAZANOLAST 114607-46-4 ABCIXIMAB 143653-53-6 ACITEMATE 101197-99-3 ABECARNIL 111841-85-1 ACITRETIN 55079-83-9 ABIRATERONE 154229-19-3 ACIVICIN 42228-92-2 ABITESARTAN 137882-98-5 ACLANTATE 39633-62-0 ABLUKAST 96566-25-5 ACLARUBICIN 57576-44-0 ABUNIDAZOLE 91017-58-2 ACLATONIUM NAPADISILATE 55077-30-0 ACADESINE 2627-69-2 ACODAZOLE 79152-85-5 ACAMPROSATE 77337-76-9 ACONIAZIDE 13410-86-1 ACAPRAZINE 55485-20-6 ACOXATRINE 748-44-7 ACARBOSE 56180-94-0 ACREOZAST 123548-56-1 ACEBROCHOL 514-50-1 ACRIDOREX 47487-22-9 ACEBURIC ACID 26976-72-7 -

The Effects of Aniracetam Treatment on Cognitive Performance and AMPA Receptor Glur2 Subunit Expression After Moderate Fluid Percussion Injury in Rats
Virginia Commonwealth University VCU Scholars Compass Theses and Dissertations Graduate School 2004 The Effects of Aniracetam Treatment on Cognitive Performance and AMPA Receptor GluR2 Subunit Expression After Moderate Fluid Percussion Injury in Rats Anna Igorevna Baranova Virginia Commonwealth University Follow this and additional works at: https://scholarscompass.vcu.edu/etd Part of the Psychology Commons © The Author Downloaded from https://scholarscompass.vcu.edu/etd/799 This Dissertation is brought to you for free and open access by the Graduate School at VCU Scholars Compass. It has been accepted for inclusion in Theses and Dissertations by an authorized administrator of VCU Scholars Compass. For more information, please contact [email protected]. i THE EFFECTS OF ANIRACETAM TREATMENT ON COGNITIVE PERFORMANCE AND AMPA RECEPTOR GLUR2 SUBUNIT EXPRESSION AFTER MODERATE FLUID PERCUSSION INJURY IN RATS A dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy at Virginia Commonwealth University By Anna I. Baranova Virginia Commonwealth University Director: Robert J. Hamm Professor Department of Psychology Virginia Commonwealth University Richmond, VA September, 2004 ii Acknowledgements To Denis Kashin 3/14/1975 – 7/21/1995 iii Table of Contents LIST OF FIGURES ........................................................................................................... vi LIST OF ABBREVIATIONS........................................................................................... vii -

(12) Patent Application Publication (10) Pub. No.: US 2008/0008748A1 Beyreuther Et Al
US 200800O8748A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2008/0008748A1 Beyreuther et al. (43) Pub. Date: Jan. 10, 2008 (54) METHOD FOR TREATING PAIN USING A (30) Foreign Application Priority Data SUBSTITUTED 2-AMNOTETRALIN COMPOUND Jun. 22, 2006 (EP).............................. EP O6 O12 815.4 (76) Inventors: Bettina Beyreuther, Dusseldorf (DE); Publication Classification Dieter Scheller, Neuss (DE); Joachim (51) Int. Cl. Freitag, Nurtingen (DE); Josephe A6II 3L/38 (2006.01) Bianchine, Potomac, MD (US) A6II 3L/439 (2006.01) A 6LX 9/70 (2006.01) A6IP 25/04 (2006.01) Correspondence Address: (52) U.S. Cl. ........................... 424/449; 514/289: 514/438 HARNESS, DICKEY, & PIERCE, P.L.C 7700 BONHOMME, STE 400 (57) ABSTRACT ST. LOUIS MO 63105 US 9 (US) A method for treating pain, particularly non-inflammatory musculoskeletal pain Such as fibromyalgia, myofascial pain (21) Appl. No.: 11/764,907 or back pain, in a subject comprises administering to the Subject a substituted 2-aminotetralin compound as defined (22) Filed: Jun. 19, 2007 herein, illustratively rotigotine. vehicle 0.3 mg/kg 1 mg/kg 3 mg/kg rotigotine 100 80 2O - 10-15 time after formalin injection (min) Patent Application Publication Jan. 10, 2008 Sheet 1 of 2 US 2008/0008748A1 vehicle 0.3 mg/kg E 1 mg/kg 3 mg/kg rotigotine 100 6 O 4. O 2 O 10-15 15-20 20-25 25-30 time after formalin injection (min) Fig. 1 vehicle 0.3 mg/kg 1 mg/kg 3 mg/kg rotigotine 300 250 200 15 O 1 O O 5 O O-5 10-30 time after formalin injection (min) Fig.