Na+/Mg2+ Exchanger
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Nuclear and Mitochondrial Genome Defects in Autisms
UC Irvine UC Irvine Previously Published Works Title Nuclear and mitochondrial genome defects in autisms. Permalink https://escholarship.org/uc/item/8vq3278q Journal Annals of the New York Academy of Sciences, 1151(1) ISSN 0077-8923 Authors Smith, Moyra Spence, M Anne Flodman, Pamela Publication Date 2009 DOI 10.1111/j.1749-6632.2008.03571.x License https://creativecommons.org/licenses/by/4.0/ 4.0 Peer reviewed eScholarship.org Powered by the California Digital Library University of California THE YEAR IN HUMAN AND MEDICAL GENETICS 2009 Nuclear and Mitochondrial Genome Defects in Autisms Moyra Smith, M. Anne Spence, and Pamela Flodman Department of Pediatrics, University of California, Irvine, California In this review we will evaluate evidence that altered gene dosage and structure im- pacts neurodevelopment and neural connectivity through deleterious effects on synap- tic structure and function, and evidence that the latter are key contributors to the risk for autism. We will review information on alterations of structure of mitochondrial DNA and abnormal mitochondrial function in autism and indications that interactions of the nuclear and mitochondrial genomes may play a role in autism pathogenesis. In a final section we will present data derived using Affymetrixtm SNP 6.0 microar- ray analysis of DNA of a number of subjects and parents recruited to our autism spectrum disorders project. We include data on two sets of monozygotic twins. Col- lectively these data provide additional evidence of nuclear and mitochondrial genome imbalance in autism and evidence of specific candidate genes in autism. We present data on dosage changes in genes that map on the X chromosomes and the Y chro- mosome. -

309 in PTPRCAP Is Associated with Susceptibility to Diffuse-Type Gastric
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Elsevier - Publisher Connector Volume 11 Number 12 December 2009 pp. 1340–1347 1340 www.neoplasia.com Hyoungseok Ju*, Byungho Lim*, Minjin Kim*, A Regulatory Polymorphism at † ‡ § Yong Sung Kim , Woo Ho Kim , Chunhwa Ihm , − PTPRCAP Seung-Moo Noh¶,DongSooHan#, Hang-Jong Yu**, Position 309 in Is †† Associated with Susceptibility Bo Youl Choi and Changwon Kang* to Diffuse-type Gastric Cancer *Department of Biological Sciences, Korea Advanced 1 Institute of Science and Technology, Daejeon, South Korea; and Gene Expression †Medical Genomics Research Center, Korea Research Institute of Bioscience and Biotechnology, Daejeon, South Korea; ‡Department of Pathology, Seoul National University College of Medicine, Seoul, South Korea; §Department of Laboratory Medicine, Eulji University College of Medicine, Daejeon, South Korea; ¶Department of General Surgery, Chungnam National University College of Medicine, Daejeon, South Korea; #Department of Medicine, Hanyang University Guri Hospital, Guri, South Korea; **Korea Gastric Cancer Center, Seoul Paik Hospital, Seoul, South Korea; ††Department of Preventive Medicine, Hanyang University College of Medicine, Seoul, South Korea Abstract PTPRCAP (CD45-AP) is a positive regulator of protein tyrosine phosphatase PTPRC (CD45), which activates Src family kinases implicated in tumorigenesis. Single-nucleotide polymorphism (SNP) rs869736 located at position −309 of the PTPRCAP promoter was associated with susceptibility to diffuse-type gastric cancer in the current case-control study. The minor-allele homozygote was significantly associated with a 2.5-fold increased susceptibility to diffuse- type gastric cancer (P = .0021, n = 252), but not to intestinal-type (P =.30,n =178),versus the major-allele homo- zygote, when comparing unrelated Korean patients with healthy controls (n = 406). -

Human Induced Pluripotent Stem Cell–Derived Podocytes Mature Into Vascularized Glomeruli Upon Experimental Transplantation
BASIC RESEARCH www.jasn.org Human Induced Pluripotent Stem Cell–Derived Podocytes Mature into Vascularized Glomeruli upon Experimental Transplantation † Sazia Sharmin,* Atsuhiro Taguchi,* Yusuke Kaku,* Yasuhiro Yoshimura,* Tomoko Ohmori,* ‡ † ‡ Tetsushi Sakuma, Masashi Mukoyama, Takashi Yamamoto, Hidetake Kurihara,§ and | Ryuichi Nishinakamura* *Department of Kidney Development, Institute of Molecular Embryology and Genetics, and †Department of Nephrology, Faculty of Life Sciences, Kumamoto University, Kumamoto, Japan; ‡Department of Mathematical and Life Sciences, Graduate School of Science, Hiroshima University, Hiroshima, Japan; §Division of Anatomy, Juntendo University School of Medicine, Tokyo, Japan; and |Japan Science and Technology Agency, CREST, Kumamoto, Japan ABSTRACT Glomerular podocytes express proteins, such as nephrin, that constitute the slit diaphragm, thereby contributing to the filtration process in the kidney. Glomerular development has been analyzed mainly in mice, whereas analysis of human kidney development has been minimal because of limited access to embryonic kidneys. We previously reported the induction of three-dimensional primordial glomeruli from human induced pluripotent stem (iPS) cells. Here, using transcription activator–like effector nuclease-mediated homologous recombination, we generated human iPS cell lines that express green fluorescent protein (GFP) in the NPHS1 locus, which encodes nephrin, and we show that GFP expression facilitated accurate visualization of nephrin-positive podocyte formation in -

Research Article Sex Difference of Ribosome in Stroke-Induced Peripheral Immunosuppression by Integrated Bioinformatics Analysis
Hindawi BioMed Research International Volume 2020, Article ID 3650935, 15 pages https://doi.org/10.1155/2020/3650935 Research Article Sex Difference of Ribosome in Stroke-Induced Peripheral Immunosuppression by Integrated Bioinformatics Analysis Jian-Qin Xie ,1,2,3 Ya-Peng Lu ,1,3 Hong-Li Sun ,1,3 Li-Na Gao ,2,3 Pei-Pei Song ,2,3 Zhi-Jun Feng ,3 and Chong-Ge You 2,3 1Department of Anesthesiology, Lanzhou University Second Hospital, Lanzhou, Gansu 730030, China 2Laboratory Medicine Center, Lanzhou University Second Hospital, Lanzhou, Gansu 730030, China 3The Second Clinical Medical College of Lanzhou University, Lanzhou, Gansu 730030, China Correspondence should be addressed to Chong-Ge You; [email protected] Received 13 April 2020; Revised 8 October 2020; Accepted 18 November 2020; Published 3 December 2020 Academic Editor: Rudolf K. Braun Copyright © 2020 Jian-Qin Xie et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Ischemic stroke (IS) greatly threatens human health resulting in high mortality and substantial loss of function. Recent studies have shown that the outcome of IS has sex specific, but its mechanism is still unclear. This study is aimed at identifying the sexually dimorphic to peripheral immune response in IS progression, predicting potential prognostic biomarkers that can lead to sex- specific outcome, and revealing potential treatment targets. Gene expression dataset GSE37587, including 68 peripheral whole blood samples which were collected within 24 hours from known onset of symptom and again at 24-48 hours after onset (20 women and 14 men), was downloaded from the Gene Expression Omnibus (GEO) datasets. -

Microcephaly with Simplified Gyration, Epilepsy, and Infantile
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by El Servicio de Difusión de la Creación Intelectual ARTICLE Microcephaly with Simplified Gyration, Epilepsy, and Infantile Diabetes Linked to Inappropriate Apoptosis of Neural Progenitors Cathryn J. Poulton,1 Rachel Schot,1 Sima Kheradmand Kia,1 Marta Jones,3 Frans W. Verheijen,1 Hanka Venselaar,4 Marie-Claire Y. de Wit,2 Esther de Graaff,5 Aida M. Bertoli-Avella,1 and Grazia M.S. Mancini1,* We describe a syndrome of primary microcephaly with simplified gyral pattern in combination with severe infantile epileptic enceph- alopathy and early-onset permanent diabetes in two unrelated consanguineous families with at least three affected children. Linkage analysis revealed a region on chromosome 18 with a significant LOD score of 4.3. In this area, two homozygous nonconserved missense mutations in immediate early response 3 interacting protein 1 (IER3IP1) were found in patients from both families. IER3IP1 is highly expressed in the fetal brain cortex and fetal pancreas and is thought to be involved in endoplasmic reticulum stress response. We reported one of these families previously in a paper on Wolcott-Rallison syndrome (WRS). WRS is characterized by increased apoptotic cell death as part of an uncontrolled unfolded protein response. Increased apoptosis has been shown to be a cause of microcephaly in animal models. An autopsy specimen from one patient showed increased apoptosis in the cerebral cortex and pancreas beta cells, implicating premature cell death as the pathogenetic mechanism. Both patient fibroblasts and control fibroblasts treated with siRNA specific for IER3IP1 showed an increased susceptibility to apoptotic cell death under stress conditions in comparison to controls. -

MIKULASOVA Et Al. MMEJ DRIVES 8Q24 REARRANGEMENTS in MYELOMA
MIKULASOVA et al. MMEJ DRIVES 8q24 REARRANGEMENTS IN MYELOMA Microhomology-mediated end joining drives complex rearrangements and over-expression of MYC and PVT1 in multiple myeloma Authors Aneta Mikulasova1,2, Cody Ashby1, Ruslana G. Tytarenko1, Pingping Qu3, Adam Rosenthal3, Judith A. Dent1, Katie R. Ryan1, Michael A. Bauer1, Christopher P. Wardell1, Antje Hoering3, Konstantinos Mavrommatis4, Matthew Trotter5, Shayu Deshpande1, Erming Tian1, Jonathan Keats6, Daniel Auclair7, Graham H. Jackson8, Faith E. Davies1, Anjan Thakurta4, Gareth J. Morgan1, and Brian A. Walker1 Affiliations 1Myeloma Center, University of Arkansas for Medical Sciences, Little Rock, AR, USA 2Institute of Cellular Medicine, Newcastle University, Newcastle upon Tyne, United Kingdom 3Cancer Research and Biostatistics, Seattle, WA, USA 4Celgene Corporation, Summit, NJ, USA 5Celgene Institute for Translational Research Europe, Seville, Spain 6Translational Genomics Research Institute, Phoenix, AZ, USA. 7Multiple Myeloma Research Foundation, Norwalk, CT, USA. 8Northern Institute for Cancer Research, Newcastle University, Newcastle upon Tyne, United Kingdom MIKULASOVA et al. MMEJ DRIVES 8q24 REARRANGEMENTS IN MYELOMA Contents Supplementary Methods ............................................................................................................... 2 Data Analysis .............................................................................................................................. 2 External Datasets ...................................................................................................................... -

Single-Cell Transcriptomes Reveal a Complex Cellular Landscape in the Middle Ear and Differential Capacities for Acute Response to Infection
fgene-11-00358 April 9, 2020 Time: 15:55 # 1 ORIGINAL RESEARCH published: 15 April 2020 doi: 10.3389/fgene.2020.00358 Single-Cell Transcriptomes Reveal a Complex Cellular Landscape in the Middle Ear and Differential Capacities for Acute Response to Infection Allen F. Ryan1*, Chanond A. Nasamran2, Kwang Pak1, Clara Draf1, Kathleen M. Fisch2, Nicholas Webster3 and Arwa Kurabi1 1 Departments of Surgery/Otolaryngology, UC San Diego School of Medicine, VA Medical Center, La Jolla, CA, United States, 2 Medicine/Center for Computational Biology & Bioinformatics, UC San Diego School of Medicine, VA Medical Center, La Jolla, CA, United States, 3 Medicine/Endocrinology, UC San Diego School of Medicine, VA Medical Center, La Jolla, CA, United States Single-cell transcriptomics was used to profile cells of the normal murine middle ear. Clustering analysis of 6770 transcriptomes identified 17 cell clusters corresponding to distinct cell types: five epithelial, three stromal, three lymphocyte, two monocyte, Edited by: two endothelial, one pericyte and one melanocyte cluster. Within some clusters, Amélie Bonnefond, Institut National de la Santé et de la cell subtypes were identified. While many corresponded to those cell types known Recherche Médicale (INSERM), from prior studies, several novel types or subtypes were noted. The results indicate France unexpected cellular diversity within the resting middle ear mucosa. The resolution of Reviewed by: Fabien Delahaye, uncomplicated, acute, otitis media is too rapid for cognate immunity to play a major Institut Pasteur de Lille, France role. Thus innate immunity is likely responsible for normal recovery from middle ear Nelson L. S. Tang, infection. The need for rapid response to pathogens suggests that innate immune The Chinese University of Hong Kong, China genes may be constitutively expressed by middle ear cells. -

Downloaded from Ftp://Ftp.Uniprot.Org/ on July 3, 2019) Using Maxquant (V1.6.10.43) Search Algorithm
bioRxiv preprint doi: https://doi.org/10.1101/2020.11.17.385096; this version posted November 17, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-ND 4.0 International license. The proteomic landscape of resting and activated CD4+ T cells reveal insights into cell differentiation and function Yashwanth Subbannayya1, Markus Haug1, Sneha M. Pinto1, Varshasnata Mohanty2, Hany Zakaria Meås1, Trude Helen Flo1, T.S. Keshava Prasad2 and Richard K. Kandasamy1,* 1Centre of Molecular Inflammation Research (CEMIR), and Department of Clinical and Molecular Medicine (IKOM), Norwegian University of Science and Technology, N-7491 Trondheim, Norway 2Center for Systems Biology and Molecular Medicine, Yenepoya (Deemed to be University), Mangalore, India *Correspondence to: Professor Richard Kumaran Kandasamy Norwegian University of Science and Technology (NTNU) Centre of Molecular Inflammation Research (CEMIR) PO Box 8905 MTFS Trondheim 7491 Norway E-mail: [email protected] (Kandasamy R K) Tel.: +47-7282-4511 1 bioRxiv preprint doi: https://doi.org/10.1101/2020.11.17.385096; this version posted November 17, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-ND 4.0 International license. Abstract CD4+ T cells (T helper cells) are cytokine-producing adaptive immune cells that activate or regulate the responses of various immune cells. -

Quantitative Interactomics in Primary T Cells Unveils TCR Signal
Quantitative interactomics in primary T cells unveils TCR signal diversification extent and dynamics Guillaume Voisinne, Kristof Kersse, Karima Chaoui, Liaoxun Lu, Julie Chaix, Lichen Zhang, Marisa Goncalves Menoita, Laura Girard, Youcef Ounoughene, Hui Wang, et al. To cite this version: Guillaume Voisinne, Kristof Kersse, Karima Chaoui, Liaoxun Lu, Julie Chaix, et al.. Quantitative interactomics in primary T cells unveils TCR signal diversification extent and dynamics. Nature Immunology, Nature Publishing Group, 2019, 20 (11), pp.1530 - 1541. 10.1038/s41590-019-0489-8. hal-03013469 HAL Id: hal-03013469 https://hal.archives-ouvertes.fr/hal-03013469 Submitted on 23 Nov 2020 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. RESOURCE https://doi.org/10.1038/s41590-019-0489-8 Quantitative interactomics in primary T cells unveils TCR signal diversification extent and dynamics Guillaume Voisinne 1, Kristof Kersse1, Karima Chaoui2, Liaoxun Lu3,4, Julie Chaix1, Lichen Zhang3, Marisa Goncalves Menoita1, Laura Girard1,5, Youcef Ounoughene1, Hui Wang3, Odile Burlet-Schiltz2, Hervé Luche5,6, Frédéric Fiore5, Marie Malissen1,5,6, Anne Gonzalez de Peredo 2, Yinming Liang 3,6*, Romain Roncagalli 1* and Bernard Malissen 1,5,6* The activation of T cells by the T cell antigen receptor (TCR) results in the formation of signaling protein complexes (signalo somes), the composition of which has not been analyzed at a systems level. -

Epilepsy and Chromosome 18 Abnormalities: a Review
Seizure 32 (2015) 78–83 Contents lists available at ScienceDirect Seizure jou rnal homepage: www.elsevier.com/locate/yseiz Review Epilepsy and chromosome 18 abnormalities: A review a, a a b Alberto Verrotti *, Alessia Carelli , Lorenza di Genova , Pasquale Striano a Department of Pediatrics, Perugia University, Perugia, Italy b Pediatric Neurology and Muscolar Diseases Unit, Department of Neurosciences, Rehabilitation, Ophthalmology, Genetics, Maternal and Child Health, University of Genoa, G. Gaslini Institute, Genova, Italy A R T I C L E I N F O A B S T R A C T Article history: Purpose: To analyze the various types of epilepsy in subjects with chromosome 18 aberrations in order to Received 9 April 2015 define epilepsy and its main clinical, electroclinical and prognostic aspects in chromosome 18 anomalies. Received in revised form 8 June 2015 Methods: A careful overview of recent works concerning chromosome 18 aberrations and epilepsy has Accepted 19 September 2015 been carried out considering the major groups of chromosomal 18 aberrations, identified using MEDLINE and EMBASE database from 1980 to 2015. Keywords: Results: Epilepsy seems to be particularly frequent in patients with trisomy or duplication of Chromosome 18 chromosome 18 with a prevalence of up to 65%. Approximately, over half of the patients develop epilepsy Chromosomal aberrations during the first year of life. Epilepsy can be focal or generalized; infantile spasms have also been reported. Epilepsy Seizures Brain imagines showed anatomical abnormalities in 38% of patients. Some antiepileptic drugs as valproic acid and carbamazepine were useful for treating seizures although a large majority of patients need polytherapy. -
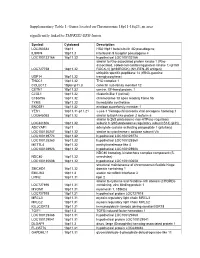
Supplementary Table 1: Genes Located on Chromosome 18P11-18Q23, an Area Significantly Linked to TMPRSS2-ERG Fusion
Supplementary Table 1: Genes located on Chromosome 18p11-18q23, an area significantly linked to TMPRSS2-ERG fusion Symbol Cytoband Description LOC260334 18p11 HSA18p11 beta-tubulin 4Q pseudogene IL9RP4 18p11.3 interleukin 9 receptor pseudogene 4 LOC100132166 18p11.32 hypothetical LOC100132166 similar to Rho-associated protein kinase 1 (Rho- associated, coiled-coil-containing protein kinase 1) (p160 LOC727758 18p11.32 ROCK-1) (p160ROCK) (NY-REN-35 antigen) ubiquitin specific peptidase 14 (tRNA-guanine USP14 18p11.32 transglycosylase) THOC1 18p11.32 THO complex 1 COLEC12 18pter-p11.3 collectin sub-family member 12 CETN1 18p11.32 centrin, EF-hand protein, 1 CLUL1 18p11.32 clusterin-like 1 (retinal) C18orf56 18p11.32 chromosome 18 open reading frame 56 TYMS 18p11.32 thymidylate synthetase ENOSF1 18p11.32 enolase superfamily member 1 YES1 18p11.31-p11.21 v-yes-1 Yamaguchi sarcoma viral oncogene homolog 1 LOC645053 18p11.32 similar to BolA-like protein 2 isoform a similar to 26S proteasome non-ATPase regulatory LOC441806 18p11.32 subunit 8 (26S proteasome regulatory subunit S14) (p31) ADCYAP1 18p11 adenylate cyclase activating polypeptide 1 (pituitary) LOC100130247 18p11.32 similar to cytochrome c oxidase subunit VIc LOC100129774 18p11.32 hypothetical LOC100129774 LOC100128360 18p11.32 hypothetical LOC100128360 METTL4 18p11.32 methyltransferase like 4 LOC100128926 18p11.32 hypothetical LOC100128926 NDC80 homolog, kinetochore complex component (S. NDC80 18p11.32 cerevisiae) LOC100130608 18p11.32 hypothetical LOC100130608 structural maintenance -

Single-Cell Sequencing Reveals Clonally Expanded Plasma Cells During Chronic Viral Infection Produce Virus-Specific and Cross-Reactive Antibodies
bioRxiv preprint doi: https://doi.org/10.1101/2021.01.29.428852; this version posted January 31, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Single-cell sequencing reveals clonally expanded plasma cells during chronic viral infection produce virus-specific and cross-reactive antibodies Daniel Neumeier1, Alessandro Pedrioli2 , Alessandro Genovese2, Ioana Sandu2, Roy Ehling1, Kai-Lin Hong1, Chrysa Papadopoulou1, Andreas Agrafiotis1, Raphael Kuhn1, Damiano Robbiani1, Jiami Han1, Laura Hauri1, Lucia Csepregi1, Victor Greiff3, Doron Merkler4,5, Sai T. Reddy1,*, Annette Oxenius2,*, Alexander Yermanos1,2,4,* 1Department of Biosystems Science and Engineering, ETH Zurich, Basel, Switzerland 2Institute of Microbiology, ETH Zurich, Zurich, Switzerland 3Department of Immunology, University of Oslo, Oslo, Norway 4Department of Pathology and Immunology, University of Geneva, Geneva, Switzerland 5Division of Clinical Pathology, Geneva University Hospital, Geneva, Switzerland *Correspondence: [email protected] ; [email protected] ; [email protected] Graphical abstract. Single-cell sequencing reveals clonally expanded plasma cells during chronic viral infection produce virus-specific and cross-reactive antibodies. bioRxiv preprint doi: https://doi.org/10.1101/2021.01.29.428852; this version posted January 31, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Neumeier et al., Abstract Plasma cells and their secreted antibodies play a central role in the long-term protection against chronic viral infection.