Heteromyidae)
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Socioeconomic and Ecological Aspects of Field Rat Control in Tropical and Subtropical Countries Bans Kurylas
SOCIOECONOMIC AND ECOLOGICAL ASPECTS OF FIELD RAT CONTROL IN TROPICAL AND SUBTROPICAL COUNTRIES BANS KURYLAS. Thai-German Rodent Control Project. German Agency for Technical Cooperation (GTZ), P.O. Box 9-34, Bangkok. Thailand. ABSTRACT: The vital question. as to the cause of the pennanent increase in field rat populations throughout most tropical and subtropical areas. has been the subject of researchers and fielcM!en during the past years, in the hope of finding an answer to this problem. Man has made his way through history wherein he was gradually able to renounce nature and establish his own man-made cultural frame. Unlike other marrmals. man has no natural instincts to guide him through life. Brain and spirit have to compensate for lack of. physical capabilities and instincts. Man was forced to change his natural surroundings in order to serve his special and ever-growing needs . Survival meant not only using nature but more so changing it in order to develop his culture. The field rat has become man's "cultural treader"! Myomorpha is the largest suborder of the Order Rodentia. Of the 1700 rodents known to man. more than 1100 belong to this group. The greater part of the Myomorpha species are found within two families, which are Cricetidae with about 567 species and Muridae with about 475 species. Most of these rodents live either in trees or under the ground. They rarely collide with man's interest. Only a few rodent species have become "field rats" and seem to dwell in and utilize man's cultural steppe. Some species have even become cosmopolitan and are found in Europe. -

Molecular Systematics of Spiny Pocket Mice (Subfamily Heteromyinae) Inferred from Mitochondrial and Nuclear Sequence Data
Brigham Young University BYU ScholarsArchive Theses and Dissertations 2009-04-17 Molecular Systematics of Spiny Pocket Mice (Subfamily Heteromyinae) Inferred from Mitochondrial and Nuclear Sequence Data Melina Crystal Williamson Brigham Young University - Provo Follow this and additional works at: https://scholarsarchive.byu.edu/etd Part of the Biology Commons BYU ScholarsArchive Citation Williamson, Melina Crystal, "Molecular Systematics of Spiny Pocket Mice (Subfamily Heteromyinae) Inferred from Mitochondrial and Nuclear Sequence Data" (2009). Theses and Dissertations. 1718. https://scholarsarchive.byu.edu/etd/1718 This Thesis is brought to you for free and open access by BYU ScholarsArchive. It has been accepted for inclusion in Theses and Dissertations by an authorized administrator of BYU ScholarsArchive. For more information, please contact [email protected], [email protected]. MOLECULAR SYSTEMATICS OF SPINY POCKET MICE (SUBFAMILY HETEROMYINAE) INFERRED FROM MITOCHONDRIAL AND NUCLEAR SEQUENCE DATA by Melina C. Williamson A thesis submitted to the faculty of Brigham Young University in partial fulfillment of the requirements for the degree of Master of Science Department of Biology Brigham Young University August 2009 BRIGHAM YOUNG UNIVERSITY GRADUATE COMMITTEE APPROVAL of a thesis submitted by Melina C. Williamson This thesis has been read by each member of the following graduate committee and by majority vote has been found to be satisfactory. Date Duke S. Rogers, Chair Date Leigh A. Johnson Date Jack W. Sites, Jr. BRIGHAM YOUNG UNIVERSITY As chair of the candidate’s graduate committee, I have read the thesis of Melina C. Williamson in its final form and have found that (1) its format, citations, and bibliographical style are consistent and acceptable and fulfill university and department style requirements; (2) its illustrative materials including figures, tables, and charts are in place; and (3) the final manuscript is satisfactory to the graduate committee and is ready for submission to the university library. -
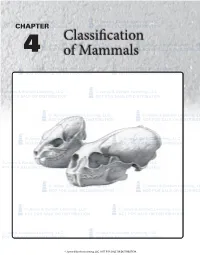
Classification of Mammals 61
© Jones & Bartlett Learning, LLC © Jones & Bartlett Learning, LLC NOT FORCHAPTER SALE OR DISTRIBUTION NOT FOR SALE OR DISTRIBUTION Classification © Jones & Bartlett Learning, LLC © Jones & Bartlett Learning, LLC 4 NOT FORof SALE MammalsOR DISTRIBUTION NOT FOR SALE OR DISTRIBUTION © Jones & Bartlett Learning, LLC © Jones & Bartlett Learning, LLC NOT FOR SALE OR DISTRIBUTION NOT FOR SALE OR DISTRIBUTION © Jones & Bartlett Learning, LLC © Jones & Bartlett Learning, LLC NOT FOR SALE OR DISTRIBUTION NOT FOR SALE OR DISTRIBUTION © Jones & Bartlett Learning, LLC © Jones & Bartlett Learning, LLC NOT FOR SALE OR DISTRIBUTION NOT FOR SALE OR DISTRIBUTION © Jones & Bartlett Learning, LLC © Jones & Bartlett Learning, LLC NOT FOR SALE OR DISTRIBUTION NOT FOR SALE OR DISTRIBUTION © Jones & Bartlett Learning, LLC © Jones & Bartlett Learning, LLC NOT FOR SALE OR DISTRIBUTION NOT FOR SALE OR DISTRIBUTION © Jones & Bartlett Learning, LLC © Jones & Bartlett Learning, LLC NOT FOR SALE OR DISTRIBUTION NOT FOR SALE OR DISTRIBUTION © Jones & Bartlett Learning, LLC © Jones & Bartlett Learning, LLC NOT FOR SALE OR DISTRIBUTION NOT FOR SALE OR DISTRIBUTION © Jones & Bartlett Learning, LLC © Jones & Bartlett Learning, LLC NOT FOR SALE OR DISTRIBUTION NOT FOR SALE OR DISTRIBUTION © Jones & Bartlett Learning, LLC. NOT FOR SALE OR DISTRIBUTION. 2ND PAGES 9781284032093_CH04_0060.indd 60 8/28/13 12:08 PM CHAPTER 4: Classification of Mammals 61 © Jones Despite& Bartlett their Learning,remarkable success, LLC mammals are much less© Jones stress & onBartlett the taxonomic Learning, aspect LLCof mammalogy, but rather as diverse than are most invertebrate groups. This is probably an attempt to provide students with sufficient information NOT FOR SALE OR DISTRIBUTION NOT FORattributable SALE OR to theirDISTRIBUTION far greater individual size, to the high on the various kinds of mammals to make the subsequent energy requirements of endothermy, and thus to the inabil- discussions of mammalian biology meaningful. -

Special Publications Museum of Texas Tech University Number 63 18 September 2014
Special Publications Museum of Texas Tech University Number 63 18 September 2014 List of Recent Land Mammals of Mexico, 2014 José Ramírez-Pulido, Noé González-Ruiz, Alfred L. Gardner, and Joaquín Arroyo-Cabrales.0 Front cover: Image of the cover of Nova Plantarvm, Animalivm et Mineralivm Mexicanorvm Historia, by Francisci Hernández et al. (1651), which included the first list of the mammals found in Mexico. Cover image courtesy of the John Carter Brown Library at Brown University. SPECIAL PUBLICATIONS Museum of Texas Tech University Number 63 List of Recent Land Mammals of Mexico, 2014 JOSÉ RAMÍREZ-PULIDO, NOÉ GONZÁLEZ-RUIZ, ALFRED L. GARDNER, AND JOAQUÍN ARROYO-CABRALES Layout and Design: Lisa Bradley Cover Design: Image courtesy of the John Carter Brown Library at Brown University Production Editor: Lisa Bradley Copyright 2014, Museum of Texas Tech University This publication is available free of charge in PDF format from the website of the Natural Sciences Research Laboratory, Museum of Texas Tech University (nsrl.ttu.edu). The authors and the Museum of Texas Tech University hereby grant permission to interested parties to download or print this publication for personal or educational (not for profit) use. Re-publication of any part of this paper in other works is not permitted without prior written permission of the Museum of Texas Tech University. This book was set in Times New Roman and printed on acid-free paper that meets the guidelines for per- manence and durability of the Committee on Production Guidelines for Book Longevity of the Council on Library Resources. Printed: 18 September 2014 Library of Congress Cataloging-in-Publication Data Special Publications of the Museum of Texas Tech University, Number 63 Series Editor: Robert J. -

Biological Notes and Descriptions of Two New Moths Phoretic on Spiny Pocket Mice in Costa Rica (Lepidoptera, Tineoidea)
PROC. ENTOMOl. SOC. WASH. 88(1). 1986, pp. 98-109 NEOTROPICAL TlNEIDAE, U: BIOLOGICAL NOTES AND DESCRIPTIONS OF TWO NEW MOTHS PHORETIC ON SPINY POCKET MICE IN COSTA RICA (LEPIDOPTERA, TINEOIDEA) DoNALD R. DAVIS, DALE H . CLAYTON, DANIEL H . J ANZEN, AND ANNE P. BROOKE (DRD) Department of Entomology, National Museum of Natural History, Smithsonian Institution, Washington, D. C. 20560; (DHC) Committee on Evo lutionary Biology, University of Chicago, Chicago, lIIinois 60637; (DHJ) De partment of Biology, University of Pennsylvania, Philadelphia, Pennsylvania 191 04; (APB) Museum of Vertebrate Zoology, Uni versity of Cali fomi a, Berkeley, California 94720. Abstract. - Two new species of tineid moths discovered riding on the backs of two species of spiny pocket mice (Heteromyidae) in Costa Rica are described. Amydria selvae. new species, was found on Heteromys desmarestianus Gray in the rainforest at Finca La Selva and Ptilopsaltis santarosae. new species, occurred on Liomys salvini (Thomas) in the dry forest at Santa Rosa National Park. Only female moths were observed and collected. Biological observations on both moths and their hosts in phoresy are summarized. Recent live-trapping of spiny pocket mice in Costa Rican forests has revealed the association of three species of ph ore tic moths of the family Tineidae. The life histories of the moths are still poorly understood and only the females are known to be phoretic. No males of any of the three species have been discovered. Exca vations of the burrows of the two species of spiny mice are now underway, which we hope will reveal not only the presence of males with associated immatures but also the larval biology. -

Learning About Mammals
Learning About Mammals The mammals (Class Mammalia) includes everything from mice to elephants, bats to whales and, of course, man. The amazing diversity of mammals is what has allowed them to live in any habitat from desert to arctic to the deep ocean. They live in trees, they live on the ground, they live underground, and in caves. Some are active during the day (diurnal), while some are active at night (nocturnal) and some are just active at dawn and dusk (crepuscular). They live alone (solitary) or in great herds (gregarious). They mate for life (monogamous) or form harems (polygamous). They eat meat (carnivores), they eat plants (herbivores) and they eat both (omnivores). They fill every niche imaginable. Mammals come in all shapes and sizes from the tiny pygmy shrew, weighing 1/10 of an ounce (2.8 grams), to the blue whale, weighing more than 300,000 pounds! They have a huge variation in life span from a small rodent living one year to an elephant living 70 years. Generally, the bigger the mammal, the longer the life span, except for bats, which are as small as rodents, but can live for up to 20 years. Though huge variation exists in mammals, there are a few physical traits that unite them. 1) Mammals are covered with body hair (fur). Though marine mammals, like dolphins and whales, have traded the benefits of body hair for better aerodynamics for traveling in water, they do still have some bristly hair on their faces (and embryonically - before birth). Hair is important for keeping mammals warm in cold climates, protecting them from sunburn and scratches, and used to warn off others, like when a dog raises the hair on its neck. -

Genetic Structure of the North American Porcupine (Erethizon Dorsatum) Across Western Texas
GENETIC STRUCTURE OF THE NORTH AMERICAN PORCUPINE (ERETHIZON DORSATUM) ACROSS WESTERN TEXAS by Erica D. Thomas A Thesis Submitted in Partial Fulfillment Of the Requirements for the Degree MASTER OF SCIENCE Major Subject: Biology West Texas A&M University Canyon, Texas December 2017 Approved: Rocky Ward, PhD Date Chairman, Thesis Committee W. David Sissom, PhD Date Member, Thesis Committee William P. Johnson, M.S. Date Member, Thesis Committee W. David Sissom, PhD Date Department Head Dean, Academic College Date Angela N. Spaulding Date Dean, Graduate School ii ABSTRACT The North American porcupine (Erethizon dorsatum) is a highly mobile, generalist species with an extensive geographical distribution in North America. The porcupine was first documented in southwestern Texas in the early 20th century, but today occurs in most of the western two-thirds of the state. This species is relatively unstudied within the Great Plains ecoregion of North America, with no genetic studies having been conducted for this species in Texas. The objectives of this study were to describe population genetic metrics of porcupines across 3 ecoregions in western Texas by examining variation in 17 polymorphic microsatellites, and to confirm the applicability of the zinc finger protein sequencing method to identify sex in a population of North American porcupines. Tissue samples from 106 porcupines were collected from the High Plains, Rolling Plains, and Edwards Plateau ecoregions of western Texas. Sex was accurately identified for 92 porcupine tissue samples by directly sequencing a short portion (195 base pairs) of the zinc finger protein gene. Sixteen base pair substitutions between Zfx and Zfy chromosomes denoted the sex of individuals; heterozygous sequence for males (Zfx and Zfy), homozygous sequence for females (Zfx only). -

The Beaver's Phylogenetic Lineage Illuminated by Retroposon Reads
www.nature.com/scientificreports OPEN The Beaver’s Phylogenetic Lineage Illuminated by Retroposon Reads Liliya Doronina1,*, Andreas Matzke1,*, Gennady Churakov1,2, Monika Stoll3, Andreas Huge3 & Jürgen Schmitz1 Received: 13 October 2016 Solving problematic phylogenetic relationships often requires high quality genome data. However, Accepted: 25 January 2017 for many organisms such data are still not available. Among rodents, the phylogenetic position of the Published: 03 March 2017 beaver has always attracted special interest. The arrangement of the beaver’s masseter (jaw-closer) muscle once suggested a strong affinity to some sciurid rodents (e.g., squirrels), placing them in the Sciuromorpha suborder. Modern molecular data, however, suggested a closer relationship of beaver to the representatives of the mouse-related clade, but significant data from virtually homoplasy- free markers (for example retroposon insertions) for the exact position of the beaver have not been available. We derived a gross genome assembly from deposited genomic Illumina paired-end reads and extracted thousands of potential phylogenetically informative retroposon markers using the new bioinformatics coordinate extractor fastCOEX, enabling us to evaluate different hypotheses for the phylogenetic position of the beaver. Comparative results provided significant support for a clear relationship between beavers (Castoridae) and kangaroo rat-related species (Geomyoidea) (p < 0.0015, six markers, no conflicting data) within a significantly supported mouse-related clade (including Myodonta, Anomaluromorpha, and Castorimorpha) (p < 0.0015, six markers, no conflicting data). Most of an organism’s phylogenetic history is fossilized in their heritable genomic material. Using data from genome sequencing projects, particularly informative regions of this material can be extracted in sufficient num- bers to resolve the deepest history of speciation. -

Plant and Rodent Communities of Organ Pipe Cactus National Monument
Plant and rodent communities of Organ Pipe Cactus National Monument Item Type text; Thesis-Reproduction (electronic) Authors Warren, Peter Lynd Publisher The University of Arizona. Rights Copyright © is held by the author. Digital access to this material is made possible by the University Libraries, University of Arizona. Further transmission, reproduction or presentation (such as public display or performance) of protected items is prohibited except with permission of the author. Download date 29/09/2021 16:51:51 Link to Item http://hdl.handle.net/10150/566520 PLANT AND RODENT COMMUNITIES OF ORGAN PIPE CACTUS NATIONAL.MONUMENT by Peter Lynd Warren A Thesis Submitted to the Faculty of the DEPARTMENT OF ECOLOGY AND EVOLUTIONARY BIOLOGY In Partial Fulfillment of the Requirements For the Degree of MASTER OF SCIENCE In the Graduate College THE UNIVERSITY OF ARIZONA 1 9 7 9 STATEMENT BY AUTHOR This thesis has been submitted in partial fulfillment of re quirements for an advanced degree at The University of Arizona and is deposited in the University Library to be made available to borrowers under rules of the Library. Brief quotations from this thesis are allowable without special permission, provided that accurate acknowledgment of source is made. Requests for permission for extended quotation from or reproduction of this manuscript in whole or in part may be granted by the head of the major department or the Dean of the Graduate College when in his judg ment the proposed use of the material is in the interests of scholar ship. In all other instances, however, permission must be obtained from the author. -

Dipodomys Ingens)
Species Status Assessment Report for the Giant Kangaroo Rat (Dipodomys ingens) Photo by Elizabeth Bainbridge Version 1.0 August 2020 Prepared by the U.S. Fish and Wildlife Service August 2020 GKR SSA Report – August 2020 EXECUTIVE SUMMARY The U.S. Fish and Wildlife Service listed the giant kangaroo rat (Dipodomys ingens) as endangered under the Endangered Species Act in 1987 due to the threats of habitat loss and widespread rodenticide use (Service 1987, entire). The giant kangaroo rat is the largest species in the genus that contains all kangaroo rats. The giant kangaroo rat is found only in south-central California, on the western slopes of the San Joaquin Valley, the Carrizo and Elkhorn Plains, and the Cuyama Valley. The preferred habitat of the giant kangaroo rat is native, sloping annual grasslands with sparse vegetation (Grinnell, 1932; Williams, 1980). This report summarizes the results of a species status assessment (SSA) that the U. S. Fish and Wildlife Service (Service) completed for the giant kangaroo rat. To assess the species’ viability, we used the three conservation biology principles of resiliency, redundancy, and representation (together, the 3Rs). These principles rely on assessing the species at an individual, population, and species level to determine whether the species can persist into the future and avoid extinction by having multiple resilient populations distributed widely across its range. Giant kangaroo rats remain in fragmented habitat patches throughout their historical range. However, some areas where giant kangaroo rats once existed have not had documented occurrences for 30 years or more. The giant kangaroo rat is found in six geographic areas (units), representing the northern, middle, and southern portions of the range. -

Paleontological Resources of the Upper and Middle San Pedro Valley
Paleontological Resources of the Upper and Middle San Pedro Valley Robert D. McCord Arizona Museum of Natural History Geological setting Regional extension causing block faulting – creation of the Basin and Range ~15Ma Poorly developed drainage results in lakes in valley bottom ?-3.4 Ma Drainage develops with flow to north, marshes, ponds and lakes significant from time to time Early Pleistocene Saint David Formation ? – 3.4 million lakes, few fossils Well developed paleomagnetic timeframe – a first for terrestrial sediments! Succession of faunas from ~3 to 1.5 Ma Blancan to ? Irvingtonian NALMA Plants diatoms charophytes Equisetum (scouring rush) Ostracoda (aquatic crustaceans) Cypridopsis cf. vidua Limnocythere cf. staplini Limnocythere sp. Candona cf. renoensis Candona sp. A Candona sp. B ?Candonlella sp. ?Heterocypris sp. ?Cycloypris sp. Potamocypris sp. Cyprideis sp. Darwinula sp. Snails and a Clam Pisidium casertanum (clam) Fossaria dalli (aquatic snail) Lymnaea caperata (aquatic snail) Lymnaea cf. elodes (aquatic snail) Bakerilymnaea bulimoides (aquatic snail) Gyraulus parvus (aquatic snail) Promenetus exacuous (aquatic snail) Promenetus umbilicatellus (aquatic snail) Physa virgata (aquatic snail) Gastrocopta cristata (terrestrial snail) Gastrocopta tappaniana (terrestrial snail) Pupoides albilabris (terrestrial snail) Vertigo milium (terrestrial snail) Vertigo ovata (terrestrial snail) cf. Succinea (terrestrial snail) Deroceras aenigma (slug) Hawaila minuscula (terrestrial snail) Fish and Amphibians indeterminate small fish Ambystoma tigrinum (tiger salamander) Scaphiopus hammondi (spadefoot toad) Bufo alvarius (toad) Hyla eximia (tree frog) Rana sp. (leopard frog) Turtles and Lizards Kinosternon arizonense (mud turtle) Terrapene cf. ornata (box turtle) Gopherus sp. (tortoise) Hesperotestudo sp. (giant tortoise) Eumeces sp. (skink) “Cnemidophorus” sp. (whiptail lizard) Crotaphytus sp. (collared lizard) Phrynosoma sp. (horned lizard) Sceloporus sp. -

Hispid Pocket Mouse Chaetodipus Hispidus
Wyoming Species Account Hispid Pocket Mouse Chaetodipus hispidus REGULATORY STATUS USFWS: No special status USFS R2: No special status USFS R4: No special status Wyoming BLM: No special status State of Wyoming: Nongame Wildlife CONSERVATION RANKS USFWS: No special status WGFD: NSSU (U), Tier III WYNDD: G5, S1S3 Wyoming Contribution: LOW IUCN: Least Concern STATUS AND RANK COMMENTS The Wyoming Natural Diversity Database has assigned Hispid Pocket Mouse (Chaetodipus hispidus) a state conservation rank ranging from S1 (Critically Imperiled) to S3 (Vulnerable) because of uncertainty about the proportion of range occupied and population trends for this species in Wyoming. NATURAL HISTORY Taxonomy: Historically, there were four recognized subspecies of Hispid Pocket Mouse, and only C. h. paradoxus was found in Wyoming 1-5. A recent DNA-based study determined that the previously accepted subspecies are neither morphologically nor genetically distinct and instead proposed new subspecies boundaries delineated by four geographically and ecologically disjunct mitochondrial clades 6. Following this taxonomic revision, Wyoming remains within the distribution of the newly defined subspecies C. h. paradoxus 6. Description: It is possible to identify Hispid Pocket Mouse in the field. It is the largest Wyoming pocket mouse species; adults weigh between 40–60 g and can reach total lengths of 200–223 mm 2. Tail, hind foot, and ear length ranges from 90–113 mm, 25–28 mm, and 12–13 mm, respectively 2. Hispid Pocket Mouse is named for its distinctly coarse dorsal pelage, which is buff to yellowish orange mixed with black hairs, thus leading to an overall brownish or even olive appearance 1, 2, 4, 5, 7.