Viropharma Failure Leaves CMV Pipeline Looking Thin
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Uniqure N.V. Paasheuvelweg 25A 1105BP Amsterdam the Netherlands +1-339-970-7000
uniQure N.V. Paasheuvelweg 25a 1105BP Amsterdam The Netherlands +1-339-970-7000 NOTICE OF EXTRAORDINARY GENERAL MEETING OF SHAREHOLDERS To be held on September 14, 2017 To the Shareholders of uniQure N.V.: Notice is hereby given that an Extraordinary General Meeting of Shareholders (the “Extraordinary Meeting”) of uniQure N.V., a public company with limited liability ( naamloze vennootschap ) under the laws of the Netherlands (the “Company,” “uniQure,” and “we”), will be held on September 14, 2017, at 9:30 a.m., Central European Summer Time, at the Company’s principal executive offices located at Paasheuvelweg 25a, 1105BP Amsterdam, the Netherlands, for the following purposes: I. Opening and announcements; II. Appointment of Jeremy P. Springhorn, Ph.D. as a non-executive director (voting proposal no. 1); III. Appointment of Madhavan Balachandran as a non-executive director (voting proposal no. 2); IV Any other business that may properly come before the meeting or any adjournment of the meeting; and V. Closing of the meeting. Each person authorized to attend the Extraordinary Meeting may inspect the Agenda at the office of uniQure. Our Board of Directors (our “Board”) recommends that you vote “FOR” each of the voting proposals noted above. The record date is set at the close of business on August 17, 2017 EST and, therefore, only the Company’s shareholders of record at the close of business on August 17, 2017 EST are entitled to receive this notice (this “Notice”) and to vote at the Extraordinary Meeting and any adjournment thereof. Only shareholders who have given notice in writing to the Company by September 12, 2017 of their intention to attend the Extraordinary Meeting in person are entitled to attend the Extraordinary Meeting in person. -

Corporate Governance
Strategic report Governance and remuneration Financial statements Investor information Corporate Governance In this section Chairman’s Governance statement 78 The Board 80 Corporate Executive Team 83 Board architecture 85 Board roles and responsibilities 86 Board activity and principal decisions 87 Our purpose, values and culture 90 The Board’s approach to engagement 91 Board performance 94 Board Committee information 96 Our Board Committee reports 97 Section 172 statement 108 Directors’ report 109 GSK Annual Report 2020 77 Chairman’s Governance statement In last year’s Governance statement, I explained that our primary Education and focus on Science objective for 2020 was to ensure there was clarity between the Given the critical importance of strengthening the pipeline, Board and management on GSK’s execution of strategy and its the Board has benefitted from devoting a higher proportion of operational priorities. We have aligned our long-term priorities its time in understanding the science behind our strategy and of Innovation, Performance and Trust powered by culture testing its application. It is important that the Board has a and agreed on the metrics to measure delivery against them. working understanding of the key strategic themes upon The Board’s annual cycle of meetings ensures that all major which our R&D strategy is based. These themes have been components of our strategy are reviewed over the course complemented by Board R&D science thematic deep dives. of the year. Our focus was on the fundamentals of our strategy: human The COVID-19 pandemic impacted and dominated all our genetics, the immune system and AI and ML, as well as to lives for the majority of 2020. -

PEDIARIX Is a Vaccine
HIGHLIGHTS OF PRESCRIBING INFORMATION • If Guillain-Barré syndrome occurs within 6 weeks of receipt of a prior These highlights do not include all the information needed to use vaccine containing tetanus toxoid, the decision to give PEDIARIX should PEDIARIX safely and effectively. See full prescribing information for be based on potential benefits and risks. (5.2) PEDIARIX. • The tip caps of the prefilled syringes contain natural rubber latex which may cause allergic reactions. (5.3) PEDIARIX [Diphtheria and Tetanus Toxoids and Acellular Pertussis • Syncope (fainting) can occur in association with administration of Adsorbed, Hepatitis B (Recombinant) and Inactivated Poliovirus injectable vaccines, including PEDIARIX. Procedures should be in place Vaccine], Suspension for Intramuscular Injection to avoid falling injury and to restore cerebral perfusion following Initial U.S. Approval: 2002 syncope. (5.4) • If temperature ≥105°F, collapse or shock-like state, or persistent, ----------------------------- INDICATIONS AND USAGE ---------------------------- PEDIARIX is a vaccine indicated for active immunization against diphtheria, inconsolable crying lasting ≥3 hours have occurred within 48 hours after tetanus, pertussis, infection caused by all known subtypes of hepatitis B virus, receipt of a pertussis-containing vaccine, or if seizures have occurred and poliomyelitis. PEDIARIX is approved for use as a 3-dose series in infants within 3 days after receipt of a pertussis-containing vaccine, the decision born of hepatitis B surface antigen (HBsAg)-negative mothers. PEDIARIX to give PEDIARIX should be based on potential benefits and risks. (5.5) may be given as early as 6 weeks of age through 6 years of age (prior to the • For children at higher risk for seizures, an antipyretic may be 7th birthday). -
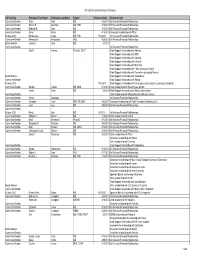
CB2013 Committee and Board Disclosureauditlist 9.10.13
2013-2014 Conflict of Interest Disclosure AST Activity Participant First Name Participant Last Name Degree DisclosureDate Disclsoure Item Committee Member Reza Abdi MD 4/22/2013 No Relevant Financial Relationships Committee Member Sameh R. Abul-Ezz MD, PhD 4/16/2013 No Relevant Financial Relationships Committee Member Deborah B. Adey MD 4/15/2013 No Relevant Financial Relationships Committee Member Enver Akalin MD 4/16/2013 Consultant relationship with Pfizer Fellows 2013 Maria-Luisa Alegre MD, PhD 7/12/2013 No Relevant Financial Relationships Committee Member Alessandrini Alessandro PhD 4/29/2013 No Relevant Financial Relationships Board Member James S. Allan MD 5/3/2013 Committee Member No Relevant Financial Relationships Rita R. Alloway PharmD, FCCP Grant Support relationship with Astellas Grant Support relationship with BMS Grant Support relationship with Novartis Grant Support relationship with Lifecycle Grant Support relationship with Millenium Grant Support relationship with Pfizer (previously Wyeth) Grant Support relationship with Genentech (previously Roche) Board Member Grant Support relationship with Viropharma Committee Member Grant Support relationship with Alexion Fellows 2013 7/18/2013 Grant Support relationship with Sanofi (previously Genzyme, previously Sangstat) Committee Member Sandra Amaral MD, MHS 6/18/2013 Other relationship with Bristol Myers-Squibb Hatem Amer MD 5/6/2013 Grant Support relationship with Abbott Laborartories. Committee Member Other relationship with Massacheussetts Medical Society. Committee Member William Applegate No Relevant Financial Relationships Committee Member Alexander Aussi BSN, RN, MBA 4/22/2013 Consultant relationship with Total Transplant Advantage, LLC Committee Member Jamil Azzi MD 4/30/2013 No Relevant Financial Relationships Committee Member Fellows 2013 Mark L. -

Final Attendee List
Jennifer Abnet GlaxoSmithKline I GlaxoSmithKline I Jane Arboleda APRN Guardant I Molly Benson Sanjiv Agarwala MD Puma Biotechnology I Cancer Expert Now I Joe Arminger AbbVie I Seth Berkowitz LCSW, Manmeet Ahluwalia MD, CCLS MBA Patricia Armstrong The Leukemia & Lymphoma Miami Cancer Institute I Novartis I Society V Steve Albers Kim Arnold APRN CPNP Barry Berman MD, MS Alexion V CPHON Florida Cancer Specialists I Servier Pharmaceuticals I Maritza Alencar DNP, Tizano Bernard MBA, APRN, BMTCN Sheila Arrington MSN, Cancer Care Centers of Miami Cancer Institute V APRN, NP-C, AOCN Brevard I Puma Biotechnology I Carmen Allen MSIT Ana Mari Bernardini Pharmavoxx V Shannon Ashmon Novartis Oncology I Eisai, Inc I Luly Almeida Bernard Berry MBA Incyte Corporation I Sarah Ashton MS AstraZeneca Pharmaceuticals Guardant Health I I Talat Almukhtar MD Orlando Health Cancer Melissa Austin Jason Bever Institute I Cancer Care Centers of Oncopeptides I Brevard I Beatrice Alvarado Roberts Amy Bignon MD Garland Avera Doyle Caris Life Sciences I University of Florida I Jazz Pharmaceuticals I Nadeem Bilani MD Tadeu Ambros MD Francie Babcock Cleveland Clinic I FCS I AMAG Pharmaceuticals/Covis I Angela Bilik RN BSN Douglas Anderson AstraZeneca I Incyte Corporation I Craig Bailey Astra Zeneca I Jamie Bilsky BS Blesson Andrews Genentech V Genentech I Kevin Barr Daiichi Sankyo I Rohit Bishnoi MD Ollie Annum PharmD University of Florida I BHMCR I Leonard Bennett PharmD EMD Serono I Brady Blackman Susmitha Apuri MD MorphoSys I Florida Cancer Specialists I Michael Bennett As of 4-22-21 Kimberly Blandon RN MSN Rick Breitenstein Memorial Cancer Institute I Apellis Pharmaceuticals I Denise Capo Karyopharm Therapeutics I Taryn Boiteau GlaxoSmithKline I Amanda Bridges Florida Society of Clinical Heidi Caravetta Tracy Bonds RN, BSN, Oncology I Exelixis, Inc. -

SAMHD1 . . . and Viral Ways Around It
viruses Review SAMHD1 . and Viral Ways around It Janina Deutschmann and Thomas Gramberg * Institute of Clinical and Molecular Virology, Friedrich-Alexander University Erlangen-Nürnberg, 91054 Erlangen, Germany; [email protected] * Correspondence: [email protected] Abstract: The SAM and HD domain-containing protein 1 (SAMHD1) is a dNTP triphosphohydrolase that plays a crucial role for a variety of different cellular functions. Besides balancing intracellular dNTP concentrations, facilitating DNA damage repair, and dampening excessive immune responses, SAMHD1 has been shown to act as a major restriction factor against various virus species. In addition to its well-described activity against retroviruses such as HIV-1, SAMHD1 has been identified to reduce the infectivity of different DNA viruses such as the herpesviruses CMV and EBV, the poxvirus VACV, or the hepadnavirus HBV. While some viruses are efficiently restricted by SAMHD1, others have developed evasion mechanisms that antagonize the antiviral activity of SAMHD1. Within this review, we summarize the different cellular functions of SAMHD1 and highlight the countermeasures viruses have evolved to neutralize the restriction factor SAMHD1. Keywords: SAMHD1; restriction factor; viral antagonism; HIV; herpesviruses; viral kinases; Vpx; dNTP hydrolase; viral interference 1. The dNTPase SAMHD1 The SAM and HD domain-containing protein 1 (SAMHD1) is a ubiquitously expressed Citation: Deutschmann, J.; deoxynucleotide triphosphohydrolase (dNTPase) of 626 amino acids (Figure1). In general, Gramberg, T. SAMHD1 . and Viral sterile alpha motif (SAM) domains have been shown to mediate protein–protein interaction Ways around It. Viruses 2021, 13, 395. or nucleic acid binding; however, its function in SAMHD1 is still unclear. The enzymatically https://doi.org/10.3390/v13030395 active HD domain, defined by two pairs of histidine and aspartate residues in its active center, on the other hand is essential for retroviral restriction and tetramerization of the Academic Editor: Sébastien Nisole protein [1–3]. -

DMD #9209 1 N-Methylpurine DNA Glycosylase and 8-Oxoguanine
DMD Fast Forward. Published on March 24, 2006 as DOI: 10.1124/dmd.105.009209 DMD FastThis article Forward. has not beenPublished copyedited on and March formatted. 24, The 2006 final version as doi:10.1124/dmd.105.009209 may differ from this version. DMD #9209 N-methylpurine DNA glycosylase and 8-oxoguanine DNA glycosylase metabolize the antiviral nucleoside 2-bromo-5,6-dichloro-1-(β-D-ribofuranosyl)benzimidazole Philip L. Lorenzi1, Christopher P. Landowski2, Andrea Brancale, Xueqin Song, Leroy B. Townsend, John C. Drach and Gordon L. Amidon. Department of Pharmaceutical Sciences (P.L.L., C.P.L., X.S., G.L.A.) and Medicinal Chemistry (L.B.T., J.C.D.), College of Pharmacy, and Department of Biologic and Downloaded from Materials Sciences (J.C.D.), School of Dentistry, University of Michigan, Ann Arbor, Michigan, USA; and Welsh School of Pharmacy (A.B.), Cardiff University, Wales, UK. dmd.aspetjournals.org at ASPET Journals on September 24, 2021 1 Copyright 2006 by the American Society for Pharmacology and Experimental Therapeutics. DMD Fast Forward. Published on March 24, 2006 as DOI: 10.1124/dmd.105.009209 This article has not been copyedited and formatted. The final version may differ from this version. DMD #9209 Running Title: Nucleoside drug metabolism by DNA repair enzymes Text pages: 19 Tables: 4 Figures: 3 References: 43 Words in Abstract: 180 Words in Introduction: 464 Words in Discussion: 1433 Abbreviations: BDCRB, 2-bromo-5,6-dichloro-1-(β-D-ribofuranosyl)benzimidazole; TCRB, 2,5,6-trichloro-1-(β-D-ribofuranosyl)benzimidazole; -

Surescripts, Llc As Amicus Curiae in Support of Petitioners in No
Nos. 19-508 and 19-825 In the Supreme Court of the United States ———————————— AMG CAPITAL MANAGEMENT, LLC, ET AL., Petitioners, v. FEDERAL TRADE COMMISSION, Respondent. ———————————— FEDERAL TRADE COMMISSION, Petitioner, v. CREDIT BUREAU CENTER, LLC, ET AL., Respondents. ———————————— ON WRITS OF CERTIORARI TO THE UNITED STATES COURTS OF APPEALS FOR THE SEVENTH AND NINTH CIRCUITS ———————————— BRIEF OF SURESCRIPTS, LLC AS AMICUS CURIAE IN SUPPORT OF PETITIONERS IN NO. 19-508 AND RESPONDENTS IN NO. 19-825 ———————————— ALFRED C. PFEIFFER, JR. ROMAN MARTINEZ LATHAM & WATKINS LLP Counsel of Record 505 Montgomery Street AMANDA P. REEVES Suite 2000 ALLYSON M. MALTAS San Francisco, CA 94111 DOUGLAS C. TIFFT BLAKE E. STAFFORD JAMES A. TOMBERLIN* LATHAM & WATKINS LLP 555 Eleventh Street, NW Suite 1000 Washington, DC 20004 (202) 637-2200 [email protected] Counsel for Amicus Curiae Surescripts, LLC TABLE OF CONTENTS Page TABLE OF AUTHORITIES ...................................... ii INTEREST OF AMICUS CURIAE ............................1 SUMMARY OF ARGUMENT .....................................3 ARGUMENT ...............................................................5 I. The FTC Has Increasingly Wielded Section 13(b) To Obtain Monetary Relief In Antitrust Cases ................................................5 II. The FTC’s Antitrust Authority Confirms That Section 13(b) Does Not Authorize Monetary Relief ..................................................22 CONCLUSION ..........................................................32 ii TABLE OF AUTHORITIES Page(s) CASES Apple Inc. v. Pepper, 139 S. Ct. 1514 (2019) .......................................... 13 Armstrong v. Exceptional Child Center, Inc., 575 U.S. 320 (2015) .............................................. 23 Bell Atlantic Corp. v. Twombly, 550 U.S. 544 (2007) .............................................. 26 In re Cardinal Health, Inc., No. 101-0006, 2015 WL 1849040 (F.T.C. Apr. 17, 2015) ........................ 19, 20, 28, 30 Credit Suisse Securities (USA) LLC v. Billing, 551 U.S. -

Novel Therapeutics for Epstein–Barr Virus
molecules Review Novel Therapeutics for Epstein–Barr Virus Graciela Andrei *, Erika Trompet and Robert Snoeck Laboratory of Virology and Chemotherapy, Department of Microbiology and Immunology, Rega Institute for Medical Research, KU Leuven, 3000 Leuven, Belgium; [email protected] (E.T.); [email protected] (R.S.) * Correspondence: [email protected]; Tel.: +32-16-321-915 Academic Editor: Stefano Aquaro Received: 15 February 2019; Accepted: 4 March 2019; Published: 12 March 2019 Abstract: Epstein–Barr virus (EBV) is a human γ-herpesvirus that infects up to 95% of the adult population. Primary EBV infection usually occurs during childhood and is generally asymptomatic, though the virus can cause infectious mononucleosis in 35–50% of the cases when infection occurs later in life. EBV infects mainly B-cells and epithelial cells, establishing latency in resting memory B-cells and possibly also in epithelial cells. EBV is recognized as an oncogenic virus but in immunocompetent hosts, EBV reactivation is controlled by the immune response preventing transformation in vivo. Under immunosuppression, regardless of the cause, the immune system can lose control of EBV replication, which may result in the appearance of neoplasms. The primary malignancies related to EBV are B-cell lymphomas and nasopharyngeal carcinoma, which reflects the primary cell targets of viral infection in vivo. Although a number of antivirals were proven to inhibit EBV replication in vitro, they had limited success in the clinic and to date no antiviral drug has been approved for the treatment of EBV infections. We review here the antiviral drugs that have been evaluated in the clinic to treat EBV infections and discuss novel molecules with anti-EBV activity under investigation as well as new strategies to treat EBV-related diseases. -

GLAXOSMITHKLINE LLC V. TEVA PHARMACEUTICALS USA, INC
Case: 18-1976 Document: 111 Page: 1 Filed: 10/02/2020 United States Court of Appeals for the Federal Circuit ______________________ GLAXOSMITHKLINE LLC, SMITHKLINE BEECHAM (CORK) LIMITED, Plaintiffs-Appellants v. TEVA PHARMACEUTICALS USA, INC., Defendant-Cross-Appellant ______________________ 2018-1976, 2018-2023 ______________________ Appeals from the United States District Court for the District of Delaware in No. 1:14-cv-00878-LPS-CJB, Chief Judge Leonard P. Stark. ______________________ Decided: October 2, 2020 ______________________ JUANITA ROSE BROOKS, Fish & Richardson, PC, San Diego, CA, argued for plaintiffs-appellants. Also represented by MICHAEL ARI AMON, CRAIG E. COUNTRYMAN, JONATHAN ELLIOT SINGER; ELIZABETH M. FLANAGAN, MICHAEL J. KANE, WILLIAM WOODFORD, Minneapolis, MN; DOUGLAS E. MCCANN, Wilmington, DE. WILLIAM M. JAY, Goodwin Procter LLP, Washington, DC, argued for defendant-cross-appellant. Also represented by JAIME ANN SANTOS; ELAINE BLAIS, J. Case: 18-1976 Document: 111 Page: 2 Filed: 10/02/2020 2 GLAXOSMITHKLINE LLC v. TEVA PHARMACEUTICALS USA, INC. ANTHONY DOWNS, ROBERT FREDERICKSON, III, CHRISTOPHER T. HOLDING, ALEXANDRA LU, LANA S. SHIFERMAN, DARYL L. WIESEN, Boston, MA; IRA J. LEVY, New York, NY. HANSJORG SAUER, Biotechnology Innovation Organization, Washington, DC, for amicus curiae Biotechnology Innovation Organization. Also represented by MELISSA A. BRAND; BRIAN PAUL BARRETT, Eli Lilly and Company, Indianapolis, IN. MICHAEL N. KENNEDY, Covington & Burling LLP, Washington, DC, for amicus curiae Pharmaceutical Research and Manufacturers of America. Also represented by STEVEN JOHN WINKELMAN; DAVID EVAN KORN, Pharmaceutical Research and Manufacturers Association of America, Washington, DC. ANDREW CURTIS NICHOLS, Winston & Strawn LLP, Washington, DC, for amicus curiae Association for Accessible Medicines. Also represented by GEORGE C. -

Declarations of Interest Thorax: First Published As 10.1136/Thoraxjnl-2016-209333.476 on 15 November 2016
Declarations of interest Thorax: first published as 10.1136/thoraxjnl-2016-209333.476 on 15 November 2016. Downloaded from KF Rabe reports financial for support for consultancy work P297 LUNG-FUNCTION PROFILE BEFORE AND AFTER THE from AstraZeneca, Chiesi, Novartis, MSD and GlaxoSmithKline, FIRST MODERATE TO SEVERE EXACERBATION DURING and grants from Altana, Novartis, AstraZeneca, MSD and THE WISDOM STUDY Nycomed. F Maltais reports financial support from Boehringer Ingelheim 10.1136/thoraxjnl-2016-209333.476 during the conduct of the study and, outside of the submitted work, financial support from Boehringer Ingelheim and GlaxoS- E Wouters, outside of the submitted work, reports personal fees mithKline for attendance at advisory boards, financial support from AstraZeneca, GlaxoSmithKline, Novartis, Takeda, Pfizer from Boehringer Ingelheim, GlaxoSmithKline, AstraZeneca and and Boehringer Ingelheim, and grants from AstraZeneca, GlaxoS- Novartis for speaking activities, and research support from Boeh- mithKline and Novartis. ringer Ingelheim, GlaxoSmithKline, AstraZeneca, Nycomed and H Magnussen received funding from Almirall, AstraZeneca, Novartis. Boehringer Ingelheim, Chiesi, GlaxoSmithKline, Merck, Novar- tis, Takeda and Roche to conduct clinical studies, and has spoken at meetings and is a member of advisory boards for these and P295 EFFICACY AND SAFETY OF TIOTROPIUM/OLODATEROL other companies. IN PATIENTS WITH COPD BY ATS CATEGORY R Rodriguez-Roisin reports personal fees from AstraZeneca, Boehringer Ingelheim, Mylan, Pearl Therapeutics, Takeda, Teva 10.1136/thoraxjnl-2016-209333.474 and Ferrer, and grants and personal fees from Menarini and Almirall. F Maltais reports financial support from Boehringer Ingelheim K Tetzlaff and S Bell are employees of Boehringer Ingelheim during the conduct of the study and, outside of the submitted Pharma GmbH & Co. -

Federal Register/Vol. 84, No. 232/Tuesday, December 3, 2019
Federal Register / Vol. 84, No. 232 / Tuesday, December 3, 2019 / Notices 66191 the Assistant Attorney General, patterns, devices, manufacturing filed with and accepted, subject to final developed the HSR Rules and the processes, or customer names. approval, by the Commission, has been corresponding Notification and Report placed on the public record for a period Form. Heather Hippsley, of thirty (30) days. The following On September 11, 2019, the Deputy General Counsel. Analysis to Aid Public Comment Commission sought comment on the [FR Doc. 2019–26075 Filed 12–2–19; 8:45 am] describes the terms of the consent reporting requirements associated with BILLING CODE 6750–01–P agreement and the allegations in the the HSR Rules and corresponding complaint. An electronic copy of the Notification and Report Form. 84 FR full text of the consent agreement 47951. No relevant comments were FEDERAL TRADE COMMISSION package can be obtained from the FTC received. Pursuant to the OMB [File No. 191 0061] Home Page (for November 15, 2019), on regulations, 5 CFR part 1320, that the World Wide Web, at https:// implement the PRA, 44 U.S.C. 3501 et Bristol-Myers Squibb Company and www.ftc.gov/news-events/commission- seq., the FTC is providing this second Celgene Corporation; Analysis of actions. opportunity for public comment while Agreement Containing Consent Orders You can file a comment online or on seeking OMB approval to renew the pre- To Aid Public Comment paper. For the Commission to consider existing clearance for those information your comment, we must receive it on or collection requirements.