Reactions of Amines
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Reduction of Organic Functional Groups Using Hypophosphites Rim Mouselmani
Reduction of Organic Functional Groups Using Hypophosphites Rim Mouselmani To cite this version: Rim Mouselmani. Reduction of Organic Functional Groups Using Hypophosphites. Other. Univer- sité de Lyon; École Doctorale des Sciences et de Technologie (Beyrouth), 2018. English. NNT : 2018LYSE1241. tel-02147583v2 HAL Id: tel-02147583 https://tel.archives-ouvertes.fr/tel-02147583v2 Submitted on 5 Jun 2019 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. THESE de DOCTORAT DE L’UNIVERSITE DE LYON EN COTUTELLE AVEC L'UNIVERSITÉ LIBANAISE opérée au sein de l’Université Claude Bernard Lyon 1 École Doctorale de Chimie-École Doctorale des Sciences et Technologies Discipline : Chimie Soutenue publiquement le 07/11/2018, par Rim MOUSELMANI Reduction of Organic Functional Groups Using Hypophosphites Devant le jury composé de Mme. Micheline DRAYE Université Savoie Mont Blanc Rapporteure M. Mohammad ELDAKDOUKI Université Arabe de Beyrouth Rapporteur Mme. Emmanuelle SCHULZ Université Paris 11 examinatrice M. Abderrahmane AMGOUNE Université Lyon 1 Président M. Mahmoud FARAJ Université Internationale Libanaise examinateur Mme. Estelle MÉTAY Université Lyon 1 Directrice de thèse M. Ali HACHEM Université Libanaise Directeur de thèse M. Marc LEMAIRE Université Lyon 1 Membre invité M. -

Chapter 18 Amines in Order for a Drug to Be Effective Orally, It Generally
Chapter 18 Amines In order for a drug to be effective orally, it generally has to be reasonable soluble in water so that it can be transported through the blood. Since amines are weak bases, they are often converted to salts with some acid and therefore may oral drugs have amine salts as part of their structure. One reason for their presence is that they confer some water solubility to the drug. The three-dimensional models show the shapes of amine molecules; notice the lone pair of electrons on nitrogen is not shown but affects the geometry about the nitrogen Primary, secondary and tertiary amines have 1, 2 or 3 alkyl groups attached to nitrogen. In these cases the alkyl group is the methyl In the IUPAC system, STEP 1 Name the longest carbon chain bonded to the N atoms as alkanamines by replacing e of the alkane name with amine. STEP 2 Number the carbon chain to locate the amine group and any substituents. N,N-Dimethylethanamine aminoethane 2-aminopropane 2-(N,N-dimethylamino)ethane 1-(N-methylamino)propane 2-(N-methylamino)butane Amines can also be names as groups attached to a hydrocarbon Aminobenzene is called aniline NH2 C H H C C C C H H C H NH2 NH2 NH CH3 Cl aniline 3-chloroaniline N-methylaniline aminobenzene 3-chloroaminobenzene N-methylaminobenzene Properties of amines The boiling points of amines are higher than alkanes of similar mass lower than alcohols of similar mass Amines are soluble in water if they have 1 to 5 carbon atoms; the N atom forms hydrogen bonds with the polar O—H bond in water An amine salt forms when an amine -

In This Handout, All of Our Functional Groups Are Presented As Condensed Line Formulas, 2D and 3D Formulas and with Nomenclature Prefixes and Suffixes (If Present)
In this handout, all of our functional groups are presented as condensed line formulas, 2D and 3D formulas and with nomenclature prefixes and suffixes (if present). Organic names are built on a foundation of alkanes, alkenes and alkynes. Those examples are presented first and you need to know those rules. The strategies can be found in Chapter 4 of our textbook (alkanes: pages 93-98, cycloalkanes 102-104, alkenes: pages 104-110, alkynes: pages 112-113 and combinations of all of them 113-115). After introducing examples of alkanes, alkenes, alkynes and combinations of them, the functional groups are presented in order of priority. A few nomenclature examples are provided for each of the functional groups. Examples of the various functional groups are presented on pages 115-135 in the textbook. Two overview pages are on pages 136-137. Some functional groups have a suffix name when they are the highest priority functional group and a prefix name when they are not the highest priority group, and these are added to the skeletal names with identifying numbers and stereochemistry terms (E and Z for alkenes, R and S for chiral centers and cis and trans for rings). Several low priority functional groups only have a prefix name. A few additional special patterns are shown on pages 98-102. The only way to learn this topic is practice (over and over). The best practice approach is to actually write out the names (on an extra piece of paper or on a white board, and then do it again). The same functional groups are used throughout the entire course. -

Efficient Synthesis of Novel Pyridine-Based Derivatives Via
molecules Article Efficient Synthesis of Novel Pyridine-Based Derivatives via Suzuki Cross-Coupling Reaction of Commercially Available 5-Bromo-2-methylpyridin-3-amine: Quantum Mechanical Investigations and Biological Activities Gulraiz Ahmad 1, Nasir Rasool 1,*, Hafiz Mansoor Ikram 1, Samreen Gul Khan 1, Tariq Mahmood 2, Khurshid Ayub 2, Muhammad Zubair 1, Eman Al-Zahrani 3, Usman Ali Rana 4, Muhammad Nadeem Akhtar 5 and Noorjahan Banu Alitheen 6,* 1 Department of Chemistry, Government College University Faisalabad, Faisalabad 38000, Pakistan; [email protected] (G.A.); [email protected] (H.M.I.); [email protected] (S.G.K.); [email protected] (M.Z.) 2 Department of Chemistry, COMSATS Institute of Information Technology, University Road, Tobe Camp, 22060 Abbottabad, Pakistan; [email protected] (T.M.); [email protected] (K.A.) 3 Chemistry Department, Faculty of Science, Taif University, 888-Taif, Saudi Arabia; [email protected] 4 Sustainable Energy Technologies Center, College of Engineering, P.O. Box 800, King Saud University, Riyadh 11421, Saudi Arabia; [email protected] 5 Faculty of Industrial Sciences & Technology, University Malaysia Pahang, Lebuhraya Tun, Razak 26300, Kuantan Pahang, Malaysia; [email protected] 6 Faculty of Biotechnology and Biomolecular Sciences, University Putra Malaysia, Serdang, Selangor Darul Ehsan 43400, Malaysia * Correspondence: [email protected] (N.R.); [email protected] (N.B.A.); Tel.: +92-332-7491790 (N.R.); +60-3-8946-7471 (N.B.A.); Fax: +92-41-9201032 (N.R.); +60-3-8946-7510 (N.B.A.) Academic Editor: Diego Muñoz-Torrero Received: 20 November 2016; Accepted: 6 January 2017; Published: 27 January 2017 Abstract: The present study describes palladium-catalyzed one pot Suzuki cross-coupling reaction to synthesize a series of novel pyridine derivatives 2a–2i, 4a–4i. -

United States Patent Office Patented Dec
3,114,704 United States Patent Office Patented Dec. 17, 1963 2 This invention is based in part on the discovery that 3,114,704 by hydrogenating the nitrile pitch and combining the ORE FLOTATION COLLECTOR AND ORE hydrogenated pitch in certain proportions with primary FOTAEON PROCESS fatty monomaines that an ore flotation collector can be Robert S. Kaufman, Paios Heights, Robert E. Baarson, 5 produced which is of equal or greater effectiveness to the Chicago, and Harold B. Treweek, Park Forest, ii., assignors to Armour and Corinpasay, Chicago, I., a straight primary amines. Since the hydrogenated nitrile corporation of Delaware pitch may comprise as much as 40 to 60% of the col No Drawing. Filed Apr. 20, 1961, Ser. No. 104,242 lector, the cost of the collector is greatly reduced. Fur 6 Claims. (C. 299-166) thermore, when the preferred proportions are employed, O the collector will not only be of substantially lower cost, This invention relates to an ore flotation collector but it will also have improved selectivity and/or collector which has particular utility in the froth flotation of silica strength. containing phosphate ores, and therefore the invention The nitrile pitch as produced, for example, by the pro also relates to a phosphate ore flotation process. cedures described in U.S. Patent Nos. 2,314,894 and At the present time primary fatty monoamines are 5 2,808,426, will contain nitriles as the principal component widely used in the froth flotation of silica containing and Will be composed of compounds and polymers there phosphate ores. -

Catalytic Carbonylation of Amines And
CATALYTIC CARBONYLATION OF AMINES AND DIAMINES AS AN ALTERNATIVE TO PHOSGENE DERIVATIVES: APPLICATION TO SYNTHESES OF THE CORE STRUCTURE OF DMP 323 AND DMP 450 AND OTHER FUNCTIONALIZED UREAS By KEISHA-GAY HYLTON A DISSERTATION PRESENTED TO THE GRADUATE SCHOOL OF THE UNIVERSITY OF FLORIDA IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF DOCTOR OF PHILOSOPHY UNIVERSITY OF FLORIDA 2004 Copyright 2004 by Keisha-Gay Hylton Dedicated to my father Alvest Hylton; he never lived to celebrate any of my achievements but he is never forgotten. ACKNOWLEDGMENTS A number of special individuals have contributed to my success. I thank my mother, for her never-ending support of my dreams; and my grandmother, for instilling integrity, and for her encouragement. Special thanks go to my husband Nemanja. He is my confidant, my best friend, and the love of my life. I thank him for providing a listening ear when I needed to “discuss” my reactions; and for his support throughout these 5 years. To my advisor (Dr. Lisa McElwee-White), I express my gratitude for all she has taught me over the last 4 years. She has shaped me into the chemist I am today, and has provided a positive role model for me. I am eternally grateful. I, of course, could never forget to mention my group members. I give special mention to Corey Anthony, for all the free coffee and toaster strudels; and for helping to keep the homesickness at bay. I thank Daniel for all the good gossip and lessons about France. I thank Yue Zhang for carbonylation discussions, and lessons about China. -

Lecture 7 Imines, Hydrazones and Oximes
Lecture 7 Imines, Hydrazones and Oximes Objectives: By the end of this lecture you will be able to: • draw the mechanism for imine formation; • account for the decreased electrophilicity of C=N compounds; • use frontier molecular orbitals to account for the increased nucleophilicity of hydroxylamines and hydrazines over alkoxides and amines; • draw the mechanism for oxime and hydrazone formation; • use 2,4-dinitrophenylhydrazones for characterising aldehydes and ketones; • use the Beckmann rearrangement for preparing amides from oximes. Introduction The carbonyl group (C=O) is the most important C=X functional group. However X can be other heteroatoms such as N and S. In this lecture we will examine the formation of a variety of C=N functional groups and compare their reactivity with their oxygen analogues. Imines The reaction of primary amines with aldehydes and ketones under dehydrating conditions generates the corresponding imine. The mechanism is a variation of the nucleophilic addition- elimination reaction that we have discussed for carboxylic acid derivatives. • You should compare the first few steps with those in the hydration of aldehydes and ketones; the mechanisms are identical. • The reaction is normally acid-catalysed (Lewis or Brønsted). Whilst primary amines are relatively good nucleophiles, without some activation of the carbonyl group, nucleophilic addition is generally slow. + • Note that primary amines are also much more basic than the carbonyl oxygen (pKa(RNH3 ) ~ + 11; pKa(R(C=OH) R) ~ -6) so for the most part the amine will be protonated making it non- nucleophilic). • Under acidic conditions, the rate-determining step is not surprisingly the addition reaction to form the first tetrahedral intermediate. -

Highly Regioselective Hydroaminomethylation of Long Chain Alkenes Catalyzed by Rh–BISBIS in a Two-Phase System
http://www.paper.edu.cn Catalysis Communications 7 (2006) 979–981 www.elsevier.com/locate/catcom Highly regioselective hydroaminomethylation of long chain alkenes catalyzed by Rh–BISBIS in a two-phase system Yingyong Wang, Junhua Chen, Meiming Luo *, Hua Chen, Xianjun Li Key Laboratory of Green Chemistry and Technology of Ministry of Education at Sichuan University, College of Chemistry, Sichuan University, Chengdu, Sichuan 610064, PR China Received 5 October 2005; received in revised form 17 January 2006; accepted 12 April 2006 Available online 22 April 2006 Abstract The hydroaminomethylation of long chain alkenes with secondary amines in aqueous/organic two-phase system catalyzed by rho- dium catalyst precursor and water-soluble diphosphine ligand BISBIS (sulfonated 2,20-bis(diphenylphosphinomethyl)-1,10-biphenyl) was investigated. The result showed that the use of BISBIS improved the reaction activity and especially regioselectivity for linear amine significantly compared with the monophosphine ligand TPPTS [P(m-C6H4SO3Na)3], the ratio of linear to branched amine was up to 83. Ó 2006 Elsevier B.V. All rights reserved. Keywords: Hydroaminomethylation; Long chain alkene; Rh complex; Aqueous two-phase catalysis 1. Introduction two-phase catalytic system [6]. We reported that the cata- lytic hydroaminomethylation in aqueous/organic two- Amines with long aliphatic chains are of importance as phase system could be extended to long chain alkenes by synthetic intermediates for surfactant, membrane compo- using water-soluble rhodium catalyst RhCl(CO)(TPPTS)2 nent and biologically active compounds [1]. Hydroami- in the presence of cationic surfactant cetyltrimethylammo- nomethylation, the one-pot efficient synthesis of amine nium bromide (CTAB) [7]. -
Chapter 6 Amines and Amides
Chapter 6 Amines and Amides Chapter 6 Amines and Amides Chapter Objectives: • Learn to recognize the amine and amide functional groups. • Learn the IUPAC system for naming amines and amides. • Learn the important physical properties of the amines and amides. • Learn the major chemical reactions of amines and amides, and learn how to predict the products of amide synthesis and hydrolysis reactions. • Learn some of the important properties of condensation polymers, especially the polyamides. Mr. Kevin A. Boudreaux Angelo State University CHEM 2353 Fundamentals of Organic Chemistry Organic and Biochemistry for Today (Seager & Slabaugh) www.angelo.edu/faculty/kboudrea Nitrogen-Containing Functional Groups • Nitrogen is in Group V of the periodic table, and in most of its compounds, it has three single bonds and one lone pair: N • In this chapter, we will take a look at two functional groups which contain nitrogen atoms connected to carbons: the amines and the amides. O RR''N RCN R' R' R" Amine Amide 2 Chapter 6 Amines and Amides Classification and Nomenclature of Amines 3 Amines • Amines and amides are abundant in nature. They are a major component of proteins and enzymes, nucleic acids, alkaloid drugs, etc. (Alkaloids are N- containing, weakly basic organic compounds; thousands of these substances are known.) • Amines are organic derivatives of ammonia, NH3, in which one or more of the three H’s is replaced by a carbon group. • Amines are classified as primary (1°), secondary (2°), or tertiary (3°), depending on how many carbon groups are connected to the nitrogen atom. HHN RHN RHN RR''N H H R' R' Ammonia 1° Amine 2° Amine 3° Amine 4 Chapter 6 Amines and Amides Examples: Classifying Amines • Classify the following amines as primary (1°), secondary (2°), or tertiary (3°). -

Simulation and Optimization of Ethanol Amine Production Plant
Korean J. Chem. Eng., 26(6), 1504-1511 (2009) DOI: 10.1007/s11814-009-0254-z RAPID COMMUNICATION Simulation and optimization of ethanol amine production plant Gholamreza Zahedi*,†, Saeideh Amraei**, and Mazda Biglari*** *Process Systems Engineering Centre (PROSPECT), Faculty of Chemical and Natural Resources Engineering, Universiti Teknologi Malaysia, UTM Skudai, 81310 Johor Bahru, Johor, Malaysia **Simulation and Artificial Intelligence Research Center, Chemical Engineering, Department, Razi University, Iran ***Department of Chemical Engineering, Ryerson University, Toronto, Ontario, Canada M5B 2K3 (Received 6 April 2009 • accepted 7 April 2009) Abstract−An industrial Ethanol Amine (EA) production plant was simulated and optimized. Due to lack of accurate reaction rate information, the first step involved obtaining reliable kinetic data from the SRI (Stanford Research In- stitute) industrial database and calculation using error minimization method. In the next step, by implementing the ob- tained reaction kinetics the whole plant was simulated using Hysys software. Simulation results were compared with the SRI data and showed that there is acceptable agreement between simulation and the measured industrial data. In the next step of study by applying the gradient search (GS) optimization technique the plant was optimized using: feeding ammonia to ethylene oxide (EO) molar ratio, water flow rate in the feed stream, and reactor temperature as optimization variables. Employing process profit as objective function the optimal operating conditions were found to be: ammonia to EO ratio of 5 (mol/mol), water flow rate of 52.59 kg mol/hr and reactor temperature of 85 oC. Key words: Simulation, Optimization, Ethanol Amine Production, Ethylene Oxide, Ammonia INTRODUCTION sity and have ammonia-like odor. -
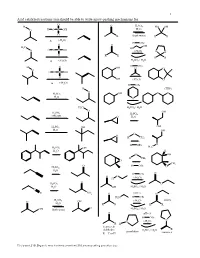
Acid Catalyzed Reactions You Should Be Able to Write Arrow-Pushing Mechanisms For
1 Acid catalyzed reactions you should be able to write arrow-pushing mechanisms for. O H O H2SO4 HO OH O H O S OH H2O O R R (both ways) R R ∆ (-H2O) H OTs O O OH H3C HO H O S OH O (-H2O) O O O R R R R ∆ (-H2O) H2SO4 / H2O H OTs H O OH O O O H O S OH O OH (-H2O) O ∆ (-H2O) H OTs H (THP) O O O H2SO4 OH H2O O H3C H2SO4 / H2O O H SO OH 2 4 H2SO4 CH OH 3 O H2O H2SO4 OH OH H2O OH O O H CH3 H OTs O H C H2SO4 OH 3 H2O H O OH H CH3 O CH3 O H OTs O H2SO4 H2O H OTs O O O (-H O) H 2 O H2SO4 O H2O OH H2SO4 / H2O C N NH2 O pH≈ 5 N H2N H OTs H2SO4 (-H O) imines O OH 2 H2O R R R R O OH (both ways) H2SO4 / H2O O pH≈ 5 H OTs N N (-H2O) R H ketones & R aldehydes H2SO4 / H2O pyrrolidine enamines R = C or H Z:\classes\316\Organic mechanisms overview\316 arrow pushing practice.doc 2 Examples of acyl substitution reactions, you should be able to write arrow-pushing mechanisms for. O O O O O O O N H OH N Cl O O O O O SH O O Cl S O O O AlCl3 Rriedel-Crafts OH reactions Cl O H O O O H Al H Li R N R H OH HO H N O Cl H O H B H O Na Li O R H very slow reaction Cu O Cl (cuprates) O Al O O O R 1. -

Chapter 16 – Amines and Amides
Chapter 16 – Amines and Amides 16.1 Occurrence, Names, and Physical Properties of Amines An amine is basically an ammonia molecule with one or more aliphatic and/or aromatic organic groups attached. Thus they have the generic formulas NH2R, NHR2, and NR3. If a carbonyl group lies between the nitrogen and R the compound is called an amide. Amines are typically named by naming the organic group as a substituent and using the suffix “-amine.” This is the old common system and we will use it for the rest of the course. If an -NH2 group is named as a substituent, it goes by “amino-”. For example: O CH3NH2 (CH3)2NH (CH3)2N(C2H5) CH3CNH2 acetamide methylamine dimethylamine ethyldimethylamine ethanamide Aromatic amines are called anilines. When other groups are also attached to the nitrogen in aniline, they are named as for amines, except that a capital “N” is placed in front to indicate the substituent group is bound to the nitrogen, rather than the aromatic ring. A couple of examples include: NH2 N(CH3)2 aniline N,N-dimethylaniline Aromatic amides are analogous to their aliphatic counterparts. Two amines/amides that you have heard of are: H O O N HO N N n H O H Acetaminophen Nylon-66 N-aceto-4-aminophenol poly(hexamethylene adipamide) 2 Many natural products chemicals (these are species extracted from living systems) contain amino groups. The alkaloids are a class of these materials. If you see a drug end in “-ine” there is a good chance of it containing an amino group. Examples include: codeine, morphine, and quinine (all have pictures in your book on p.