Hovey Et Al. (45) Date of Patent: *May 26, 2015
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Comparing the Effects of Combined Oral Contraceptives Containing Progestins with Low Androgenic and Antiandrogenic Activities on the Hypothalamic-Pituitary-Gonadal Axis In
JMIR RESEARCH PROTOCOLS Amiri et al Review Comparing the Effects of Combined Oral Contraceptives Containing Progestins With Low Androgenic and Antiandrogenic Activities on the Hypothalamic-Pituitary-Gonadal Axis in Patients With Polycystic Ovary Syndrome: Systematic Review and Meta-Analysis Mina Amiri1,2, PhD, Postdoc; Fahimeh Ramezani Tehrani2, MD; Fatemeh Nahidi3, PhD; Ali Kabir4, MD, MPH, PhD; Fereidoun Azizi5, MD 1Students Research Committee, School of Nursing and Midwifery, Department of Midwifery and Reproductive Health, Shahid Beheshti University of Medical Sciences, Tehran, Islamic Republic Of Iran 2Reproductive Endocrinology Research Center, Research Institute for Endocrine Sciences, Shahid Beheshti University of Medical Sciences, Tehran, Islamic Republic Of Iran 3School of Nursing and Midwifery, Department of Midwifery and Reproductive Health, Shahid Beheshti University of Medical Sciences, Tehran, Islamic Republic Of Iran 4Minimally Invasive Surgery Research Center, Iran University of Medical Sciences, Tehran, Islamic Republic Of Iran 5Endocrine Research Center, Shahid Beheshti University of Medical Sciences, Tehran, Islamic Republic Of Iran Corresponding Author: Fahimeh Ramezani Tehrani, MD Reproductive Endocrinology Research Center Research Institute for Endocrine Sciences Shahid Beheshti University of Medical Sciences 24 Parvaneh Yaman Street, Velenjak, PO Box 19395-4763 Tehran, 1985717413 Islamic Republic Of Iran Phone: 98 21 22432500 Email: [email protected] Abstract Background: Different products of combined oral contraceptives (COCs) can improve clinical and biochemical findings in patients with polycystic ovary syndrome (PCOS) through suppression of the hypothalamic-pituitary-gonadal (HPG) axis. Objective: This systematic review and meta-analysis aimed to compare the effects of COCs containing progestins with low androgenic and antiandrogenic activities on the HPG axis in patients with PCOS. -

Fluoxymesterone-Dea Schedule Iii Androfluorene
Cat. No. F7751 FLUOXYMESTERONE--DEA SCHEDULE III ANDROFLUORENE Androgen. OH Mol. Formula: C20H29FO3 HO H3C CH3 Mol. Wt.: 336.45 (anhyd.) F H3C m.p.: 288-290°C CAS Registry No.: 76-43-7 O Chemical Name: 4-Androsten-9a-fluoro-17a-methyl-11b,17b-diol-3-one 22 Physical Properties: White photosensitive solid. [a] D = +102.34° (c = 0.47, EtOH). Caution: Due care should be exercised to prevent skin contact, ingestion or inhalation of this compound. Storage: Store tightly sealed at room temperature, protected from exposure to light. Solubility: Soluble in ethanol (7.3 mg/ml) or water (< 0.5 mg/ml). Solubility in 45% aqueous (w/v) 2-hydroxypropyl-b- cyclodextrin (Cat. No. H-107): 6.4 mg/ml. Disposal: Dissolve or mix the compound with a combustible solvent and burn in a chemical incinerator equipped with an afterburner and scrubber. This product is controlled by the Drug Enforcement Administration. Appropriate security must be maintained until the substance is destroyed. Records must be kept which detail the ultimate disposition of the material. References: 1. Merck Index 11th Ed., No. 4113. 2. Saborido, A., Vila, J., Molano, F., Megias, A. “Effect of steroids on mitochondria and sarcotubular system of skeletal muscle.” J. Appl. Physiol. 70(3), 1038 (1991). 3. Fernandez, L., Chirino, R., Boada, L.D., Navarro, D., Cabrera, N., del Rio, I., Diaz-Chico, B.N. “Stanozolol and danazol, unlike natural androgens, interact with the low affinity glucocorticoid-binding sites from male rat liver microsomes.” Endocrinology 134(3), 1401 (1994). CONTROLLED SUBSTANCE - DEA LICENSE REQUIRED (III) Rev. -

Ketoconazole (Systemic) | Memorial Sloan Kettering Cancer Center
PATIENT & CAREGIVER EDUCATION Ketoconazole (Systemic) This information from Lexicomp® explains what you need to know about this medication, including what it’s used for, how to take it, its side effects, and when to call your healthcare provider. Brand Names: Canada APO-Ketoconazole; Ketoconazole-200; TEVA-Ketoconazole Warning This drug is not for use to treat certain types of fungal infections. This includes fungal infections of the skin, nails, or brain. Talk with the doctor. This drug must only be used when other drugs cannot be used or have not worked. Talk with your doctor to be sure that the benefits of this drug are more than the risks. Very bad and sometimes deadly liver problems like the need for a liver transplant have happened with this drug. Some people did not have a raised chance of liver problems before taking this drug. Most of the time, but not always, liver problems have gone back to normal after this drug was stopped. Call your doctor right away if you have signs of liver problems like dark urine, feeling tired, not hungry, upset stomach or stomach pain, light-colored stools, throwing up, or yellow skin or eyes. Blood tests will be needed to watch for any liver problems. Talk with your doctor. Taking this drug with certain other drugs may raise the chance of very bad and sometimes deadly heart problems like a heartbeat that is not normal. Do not take this drug if you are taking any of these drugs: Cisapride, disopyramide, dofetilide, dronedarone, methadone, pimozide, quinidine, or ranolazine. Ketoconazole (Systemic) 1/6 What is this drug used for? It is used to treat fungal infections. -

Treatment of Peripheral Precocious Puberty
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by IUPUIScholarWorks Treatment of Peripheral Precocious Puberty Melissa Schoelwer, MD and Erica A Eugster, MD Section of Pediatric Endocrinology, Department of Pediatrics, Riley Hospital for Children, Indiana University School of Medicine, Indianapolis, Indiana Send correspondence to: 705 Riley Hospital Drive, Room 5960 Indianapolis, IN 46202 Phone: 317-944-3889 Fax: 317-944-3882 Email: [email protected] __________________________________________________________________________________________ This is the author's manuscript of the article published in final edited form as: Schoelwer, M., & Eugster, E. A. (2016). Treatment of Peripheral Precocious Puberty. In Puberty from Bench to Clinic (Vol. 29, pp. 230-239). Karger Publishers. http://dx.doi.org/10.1159/000438895 Peripheral Precocious Puberty Abstract There are many etiologies of peripheral precocious puberty (PPP) with diverse manifestations resulting from exposure to androgens, estrogens, or both. The clinical presentation depends on the underlying process and may be acute or gradual. The primary goals of therapy are to halt pubertal development and restore sex steroids to prepubertal values. Attenuation of linear growth velocity and rate of skeletal maturation in order to maximize height potential are additional considerations for many patients. McCune-Albright syndrome (MAS) and Familial Male-Limited Precocious Puberty (FMPP) represent rare causes of PPP that arise from activating mutations in GNAS1 and the LH receptor gene, respectively. Several different therapeutic approaches have been investigated for both conditions with variable success. Experience to date suggests that the ideal therapy for precocious puberty secondary to MAS in girls remains elusive. In contrast, while the number of treated patients remains small, several successful therapeutic options for FMPP are available. -

CYP3A4 Mediated Pharmacokinetics Drug Interaction Potential of Maha
www.nature.com/scientificreports OPEN CYP3A4 mediated pharmacokinetics drug interaction potential of Maha‑Yogaraj Gugglu and E, Z guggulsterone Sarvesh Sabarathinam1, Satish Kumar Rajappan Chandra2 & Vijayakumar Thangavel Mahalingam1* Maha yogaraja guggulu (MYG) is a classical herbomineral polyherbal formulation being widely used since centuries. The aim of this study was to investigate the efect of MYG formulation and its major constituents E & Z guggulsterone on CYP3A4 mediated metabolism. In vitro inhibition of MYG and Guggulsterone isomers on CYP3A4 was evaluated by high throughput fuorometric assay. Eighteen Adult male Sprague–Dawley rats (200 ± 25 g body weight) were randomly divided into three groups. Group A, Group B and Group C were treated with placebo, MYG and Standard E & Z guggulsterone for 14 days respectively by oral route. On 15th day, midazolam (5 mg/kg) was administered orally to all rats in each group. Blood samples (0.3 mL) were collected from the retro orbital vein at 0.25, 0.5, 0.75, 1, 2, 4, 6, 12 and 24 h of each rat were collected. The fndings from the in vitro & in vivo study proposed that the MYG tablets and its guggulsterone isomers have drug interaction potential when consumed along with conventional drugs which are CYP3A4 substrates. In vivo pharmacokinetic drug interaction study of midazolam pointed out that the MYG tablets and guggulsterone isomers showed an inhibitory activity towards CYP3A4 which may have leads to clinically signifcant interactions. Te use of alternative medicine such as herbal medicines, phytonutrients, ayurvedic products and nutraceuticals used widely by the majority of the patients for their primary healthcare needs. -
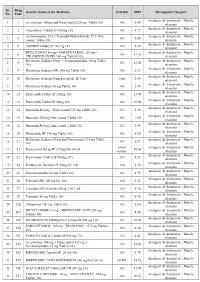
PMBJP Product.Pdf
Sr. Drug Generic Name of the Medicine Unit Size MRP Therapeutic Category No. Code Analgesic & Antipyretic / Muscle 1 1 Aceclofenac 100mg and Paracetamol 325 mg Tablet 10's 10's 8.00 relaxants Analgesic & Antipyretic / Muscle 2 2 Aceclofenac Tablets IP 100mg 10's 10's 4.37 relaxants Acetaminophen 325 + Tramadol Hydrochloride 37.5 film Analgesic & Antipyretic / Muscle 3 4 10's 8.00 coated Tablet 10's relaxants Analgesic & Antipyretic / Muscle 4 5 ASPIRIN Tablets IP 150 mg 14's 14's 2.70 relaxants DICLOFENAC 50 mg+ PARACETAMOL 325 mg+ Analgesic & Antipyretic / Muscle 5 6 10's 11.30 CHLORZOXAZONE 500 mg Tablets 10's relaxants Diclofenac Sodium 50mg + Serratiopeptidase 10mg Tablet Analgesic & Antipyretic / Muscle 6 8 10's 12.00 10's relaxants Analgesic & Antipyretic / Muscle 7 9 Diclofenac Sodium (SR) 100 mg Tablet 10's 10's 6.12 relaxants Analgesic & Antipyretic / Muscle 8 10 Diclofenac Sodium 25mg per ml Inj. IP 3 ml 3 ml 2.00 relaxants Analgesic & Antipyretic / Muscle 9 11 Diclofenac Sodium 50 mg Tablet 10's 10's 2.90 relaxants Analgesic & Antipyretic / Muscle 10 12 Etoricoxilb Tablets IP 120mg 10's 10's 33.00 relaxants Analgesic & Antipyretic / Muscle 11 13 Etoricoxilb Tablets IP 90mg 10's 10's 25.00 relaxants Analgesic & Antipyretic / Muscle 12 14 Ibuprofen 400 mg + Paracetamol 325 mg Tablet 10's 15's 5.50 relaxants Analgesic & Antipyretic / Muscle 13 15 Ibuprofen 200 mg film coated Tablet 10's 10's 1.80 relaxants Analgesic & Antipyretic / Muscle 14 16 Ibuprofen 400 mg film coated Tablet 10's 15's 3.50 relaxants Analgesic & Antipyretic -

)&F1y3x PHARMACEUTICAL APPENDIX to THE
)&f1y3X PHARMACEUTICAL APPENDIX TO THE HARMONIZED TARIFF SCHEDULE )&f1y3X PHARMACEUTICAL APPENDIX TO THE TARIFF SCHEDULE 3 Table 1. This table enumerates products described by International Non-proprietary Names (INN) which shall be entered free of duty under general note 13 to the tariff schedule. The Chemical Abstracts Service (CAS) registry numbers also set forth in this table are included to assist in the identification of the products concerned. For purposes of the tariff schedule, any references to a product enumerated in this table includes such product by whatever name known. Product CAS No. Product CAS No. ABAMECTIN 65195-55-3 ACTODIGIN 36983-69-4 ABANOQUIL 90402-40-7 ADAFENOXATE 82168-26-1 ABCIXIMAB 143653-53-6 ADAMEXINE 54785-02-3 ABECARNIL 111841-85-1 ADAPALENE 106685-40-9 ABITESARTAN 137882-98-5 ADAPROLOL 101479-70-3 ABLUKAST 96566-25-5 ADATANSERIN 127266-56-2 ABUNIDAZOLE 91017-58-2 ADEFOVIR 106941-25-7 ACADESINE 2627-69-2 ADELMIDROL 1675-66-7 ACAMPROSATE 77337-76-9 ADEMETIONINE 17176-17-9 ACAPRAZINE 55485-20-6 ADENOSINE PHOSPHATE 61-19-8 ACARBOSE 56180-94-0 ADIBENDAN 100510-33-6 ACEBROCHOL 514-50-1 ADICILLIN 525-94-0 ACEBURIC ACID 26976-72-7 ADIMOLOL 78459-19-5 ACEBUTOLOL 37517-30-9 ADINAZOLAM 37115-32-5 ACECAINIDE 32795-44-1 ADIPHENINE 64-95-9 ACECARBROMAL 77-66-7 ADIPIODONE 606-17-7 ACECLIDINE 827-61-2 ADITEREN 56066-19-4 ACECLOFENAC 89796-99-6 ADITOPRIM 56066-63-8 ACEDAPSONE 77-46-3 ADOSOPINE 88124-26-9 ACEDIASULFONE SODIUM 127-60-6 ADOZELESIN 110314-48-2 ACEDOBEN 556-08-1 ADRAFINIL 63547-13-7 ACEFLURANOL 80595-73-9 ADRENALONE -

PROCUR Why Procur Has Been Prescribed for You
Consumer Medicine Information Ask your doctor if you have any questions about PROCUR why Procur has been prescribed for you. Cyproterone acetate 50 mg and 100 mg tablets This medicine is available only with a doctor's prescription. What is in this leaflet Before you take Procur Please read this leaflet carefully before you start taking Procur When you must not take it This leaflet answers some common questions about Procur. It does not contain all the available Do not take Procur if you have an allergy to: information. It does not take the place of talking • any medicine containing cyproterone acetate to your doctor or pharmacist. • any of the ingredients listed at the end of this leaflet All medicines have risks and benefits. Your doctor has weighed the risks of you taking Procur against Some of the symptoms of an allergic reaction may the benefits they expect it will have for you. include: • difficulty in breathing or wheezing If you have any concerns about taking this • shortness of breath medicine, ask your doctor or pharmacist. • swelling of the face, tongue, lips, or other parts of the body Keep this leaflet with the medicine. You may • hives on the skin, rash, or itching need to read it again. Do not take Procur if: What Procur is used for • you are allergic to cyproterone acetate or any other ingredient listed at the end of this leaflet Procur tablets contain the active ingredient • you are pregnant cyproterone acetate. Cyproterone acetate is an • you are breastfeeding antiandrogen. It works by blocking the actions of • you suffer from liver diseases (including sex hormones (androgens) that are produced previous or existing liver tumours, Dubin- mainly in men but also, to a lesser extent in Johnson syndrome or Rotor syndrome) women. -

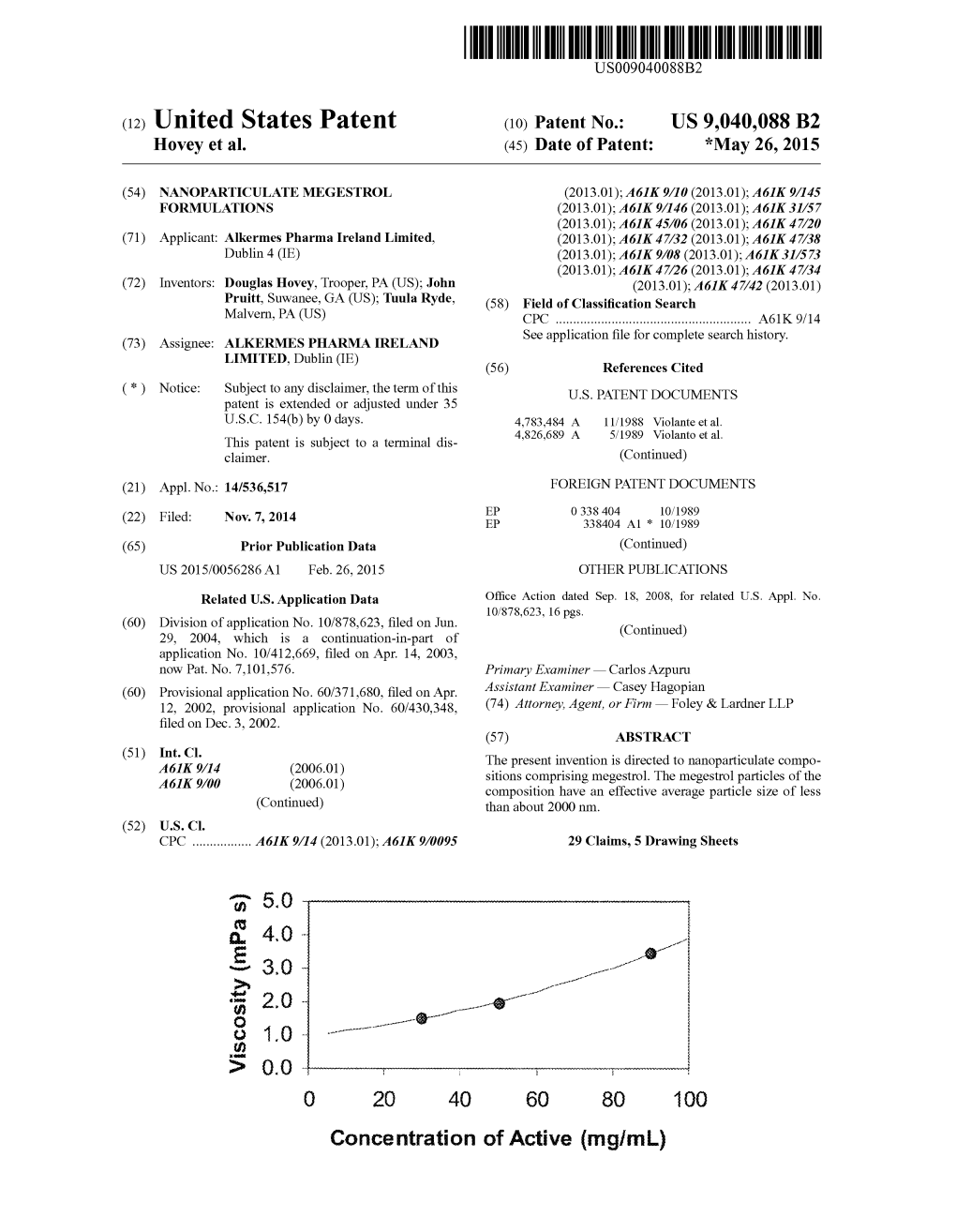
Megestrol Acetate/Melengestrol Acetate 2115 Adverse Effects and Precautions Megestrol Acetate Is Also Used in the Treatment of Ano- 16
Megestrol Acetate/Melengestrol Acetate 2115 Adverse Effects and Precautions Megestrol acetate is also used in the treatment of ano- 16. Mwamburi DM, et al. Comparing megestrol acetate therapy with oxandrolone therapy for HIV-related weight loss: similar As for progestogens in general (see Progesterone, rexia and cachexia (see below) in patients with cancer results in 2 months. Clin Infect Dis 2004; 38: 895–902. p.2125). The weight gain that may occur with meges- or AIDS. The usual dose is 400 to 800 mg daily, as tab- 17. Grunfeld C, et al. Oxandrolone in the treatment of HIV-associ- ated weight loss in men: a randomized, double-blind, placebo- trol acetate appears to be associated with an increased lets or oral suspension. A suspension of megestrol ace- controlled study. J Acquir Immune Defic Syndr 2006; 41: appetite and food intake rather than with fluid reten- tate that has an increased bioavailability is also availa- 304–14. tion. Megestrol acetate may have glucocorticoid ef- ble (Megace ES; Par Pharmaceutical, USA) and is Hot flushes. Megestrol has been used to treat hot flushes in fects when given long term. given in a dose of 625 mg in 5 mL daily for anorexia, women with breast cancer (to avoid the potentially tumour-stim- cachexia, or unexplained significant weight loss in pa- ulating effects of an oestrogen—see Malignant Neoplasms, un- Effects on carbohydrate metabolism. Megestrol therapy der Precautions of HRT, p.2075), as well as in men with hot 1-3 4 tients with AIDS. has been associated with hyperglycaemia or diabetes mellitus flushes after orchidectomy or anti-androgen therapy for prostate in AIDS patients being treated for cachexia. -

Determination of 17 Hormone Residues in Milk by Ultra-High-Performance Liquid Chromatography and Triple Quadrupole Mass Spectrom
No. LCMSMS-065E Liquid Chromatography Mass Spectrometry Determination of 17 Hormone Residues in Milk by Ultra-High-Performance Liquid Chromatography and Triple Quadrupole No. LCMSMS-65E Mass Spectrometry This application news presents a method for the determination of 17 hormone residues in milk using Shimadzu Ultra-High-Performance Liquid Chromatograph (UHPLC) LC-30A and Triple Quadrupole Mass Spectrometer LCMS- 8040. After sample pretreatment, the compounds in the milk matrix were separated using UPLC LC-30A and analyzed via Triple Quadrupole Mass Spectrometer LCMS-8040. All 17 hormones displayed good linearity within their respective concentration range, with correlation coefficient in the range of 0.9974 and 0.9999. The RSD% of retention time and peak area of 17 hormones at the low-, mid- and high- concentrations were in the range of 0.0102-0.161% and 0.563-6.55% respectively, indicating good instrument precision. Method validation was conducted and the matrix spike recovery of milk ranged between 61.00-110.9%. The limit of quantitation was 0.14-0.975 g/kg, and it meets the requirement for detection of hormones in milk. Keywords: Hormones; Milk; Solid phase extraction; Ultra performance liquid chromatograph; Triple quadrupole mass spectrometry ■ Introduction Since 2008’s melamine-tainted milk scandal, the With reference to China’s national standard GB/T adulteration of milk powder has become a major 21981-2008 "Hormone Multi-Residue Detection food safety concern. In recent years, another case of Method for Animal-derived Food - LC-MS Method", dairy product safety is suspected to cause "infant a method utilizing solid phase extraction, ultra- sexual precocity" (also known as precocious puberty) performance liquid chromatography and triple and has become another major issue challenging the quadrupole mass spectrometry was developed for dairy industry in China. -

Part I Biopharmaceuticals
1 Part I Biopharmaceuticals Translational Medicine: Molecular Pharmacology and Drug Discovery First Edition. Edited by Robert A. Meyers. © 2018 Wiley-VCH Verlag GmbH & Co. KGaA. Published 2018 by Wiley-VCH Verlag GmbH & Co. KGaA. 3 1 Analogs and Antagonists of Male Sex Hormones Robert W. Brueggemeier The Ohio State University, Division of Medicinal Chemistry and Pharmacognosy, College of Pharmacy, Columbus, Ohio 43210, USA 1Introduction6 2 Historical 6 3 Endogenous Male Sex Hormones 7 3.1 Occurrence and Physiological Roles 7 3.2 Biosynthesis 8 3.3 Absorption and Distribution 12 3.4 Metabolism 13 3.4.1 Reductive Metabolism 14 3.4.2 Oxidative Metabolism 17 3.5 Mechanism of Action 19 4 Synthetic Androgens 24 4.1 Current Drugs on the Market 24 4.2 Therapeutic Uses and Bioassays 25 4.3 Structure–Activity Relationships for Steroidal Androgens 26 4.3.1 Early Modifications 26 4.3.2 Methylated Derivatives 26 4.3.3 Ester Derivatives 27 4.3.4 Halo Derivatives 27 4.3.5 Other Androgen Derivatives 28 4.3.6 Summary of Structure–Activity Relationships of Steroidal Androgens 28 4.4 Nonsteroidal Androgens, Selective Androgen Receptor Modulators (SARMs) 30 4.5 Absorption, Distribution, and Metabolism 31 4.6 Toxicities 32 Translational Medicine: Molecular Pharmacology and Drug Discovery First Edition. Edited by Robert A. Meyers. © 2018 Wiley-VCH Verlag GmbH & Co. KGaA. Published 2018 by Wiley-VCH Verlag GmbH & Co. KGaA. 4 Analogs and Antagonists of Male Sex Hormones 5 Anabolic Agents 32 5.1 Current Drugs on the Market 32 5.2 Therapeutic Uses and Bioassays -

INTEGRIS Formulary July 2017
INTEGRIS Formulary July 2017 Foreword FORMULARY EXCLUDED THERAPEUTIC DRUG This document represents the efforts of the MedImpact Healthcare Systems THERAPEUTIC DRUGS CLASS Pharmacy and Therapeutics (P & T) and Formulary Committees to provide ALTERNATIVES physicians and pharmacists with a method to evaluate the safety, efficacy and cost- clindamycin/tretinoin, ACNE AGENTS, effectiveness of commercially available drug products. A structured approach to the VELTIN drug selection process is essential in ensuring continuing patient access to rational ZIANA TOPICAL drug therapies. The ultimate goal of the MedPerform Formulary is to provide a morphine sulfate ER process and framework to support the dynamic evolution of this document to guide tablets, oxycodone ANALGESICS, KADIAN prescribing decisions that reflect the most current clinical consensus associated ER, NUCYNTA, NARCOTICS with drug therapy decisions. NUCYNTA ER ANALGESICS, This is accomplished through the auspices of the MedImpact P & T and Formulary BELBUCA BUTRANS PATCH Committees. These committees meet quarterly and more often as warranted to NARCOTICS ensure clinical relevancy of the Formulary. To accommodate changes to this ABSTRAL, document, updates are made accessible as necessary. FENTORA, fentanyl citrate ANALGESICS, LAZANDA, lozenge NARCOTICS As you use this Formulary, you are encouraged to review the information and ONSOLIS, provide your input and comments to the MedImpact P & T and Formulary SUBSYS Committees. immediate-release GRALISE ANTICONVULSANTS The MedImpact P & T