Study Protocol and Statistical Analysis Plan
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Risk of Breast Cancer After Stopping Menopausal Hormone Therapy In
Risk of breast cancer after stopping menopausal hormone therapy in the E3N cohort Agnès Fournier, Sylvie Mesrine, Laure Dossus, Marie-Christine Boutron-Ruault, Françoise Clavel-Chapelon, Nathalie Chabbert-Buffet To cite this version: Agnès Fournier, Sylvie Mesrine, Laure Dossus, Marie-Christine Boutron-Ruault, Françoise Clavel- Chapelon, et al.. Risk of breast cancer after stopping menopausal hormone therapy in the E3N cohort. Breast Cancer Research and Treatment, Springer Verlag, 2014, 145 (2), pp.535-43. 10.1007/s10549- 014-2934-6. inserm-01319982 HAL Id: inserm-01319982 https://www.hal.inserm.fr/inserm-01319982 Submitted on 23 May 2016 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. TITLE PAGE Risk of breast cancer after stopping menopausal hormone therapy in the E3N cohort Authors : Agnès Fournier1,2,3, Sylvie Mesrine1,2,3, Laure Dossus1,2,3, Marie-Christine Boutron- Ruault1,2,3, Françoise Clavel-Chapelon1,2,3, Nathalie Chabbert-Buffet4 Affiliations: 1. Inserm, Center for research in Epidemiology and Population Health, U1018, Nutrition, Hormones and Women’s Health team, F-94807, Villejuif, France 2. Univ Paris-Sud, UMRS 1018, F-94807, Villejuif, France 3. Institut Gustave Roussy, F-94805, Villejuif, France 4. -

CASODEX (Bicalutamide)
HIGHLIGHTS OF PRESCRIBING INFORMATION • Gynecomastia and breast pain have been reported during treatment with These highlights do not include all the information needed to use CASODEX 150 mg when used as a single agent. (5.3) CASODEX® safely and effectively. See full prescribing information for • CASODEX is used in combination with an LHRH agonist. LHRH CASODEX. agonists have been shown to cause a reduction in glucose tolerance in CASODEX® (bicalutamide) tablet, for oral use males. Consideration should be given to monitoring blood glucose in Initial U.S. Approval: 1995 patients receiving CASODEX in combination with LHRH agonists. (5.4) -------------------------- RECENT MAJOR CHANGES -------------------------- • Monitoring Prostate Specific Antigen (PSA) is recommended. Evaluate Warnings and Precautions (5.2) 10/2017 for clinical progression if PSA increases. (5.5) --------------------------- INDICATIONS AND USAGE -------------------------- ------------------------------ ADVERSE REACTIONS ----------------------------- • CASODEX 50 mg is an androgen receptor inhibitor indicated for use in Adverse reactions that occurred in more than 10% of patients receiving combination therapy with a luteinizing hormone-releasing hormone CASODEX plus an LHRH-A were: hot flashes, pain (including general, back, (LHRH) analog for the treatment of Stage D2 metastatic carcinoma of pelvic and abdominal), asthenia, constipation, infection, nausea, peripheral the prostate. (1) edema, dyspnea, diarrhea, hematuria, nocturia, and anemia. (6.1) • CASODEX 150 mg daily is not approved for use alone or with other treatments. (1) To report SUSPECTED ADVERSE REACTIONS, contact AstraZeneca Pharmaceuticals LP at 1-800-236-9933 or FDA at 1-800-FDA-1088 or ---------------------- DOSAGE AND ADMINISTRATION ---------------------- www.fda.gov/medwatch The recommended dose for CASODEX therapy in combination with an LHRH analog is one 50 mg tablet once daily (morning or evening). -

COVID-19—The Potential Beneficial Therapeutic Effects of Spironolactone During SARS-Cov-2 Infection
pharmaceuticals Review COVID-19—The Potential Beneficial Therapeutic Effects of Spironolactone during SARS-CoV-2 Infection Katarzyna Kotfis 1,* , Kacper Lechowicz 1 , Sylwester Drozd˙ zal˙ 2 , Paulina Nied´zwiedzka-Rystwej 3 , Tomasz K. Wojdacz 4, Ewelina Grywalska 5 , Jowita Biernawska 6, Magda Wi´sniewska 7 and Miłosz Parczewski 8 1 Department of Anesthesiology, Intensive Therapy and Acute Intoxications, Pomeranian Medical University in Szczecin, 70-111 Szczecin, Poland; [email protected] 2 Department of Pharmacokinetics and Monitored Therapy, Pomeranian Medical University, 70-111 Szczecin, Poland; [email protected] 3 Institute of Biology, University of Szczecin, 71-412 Szczecin, Poland; [email protected] 4 Independent Clinical Epigenetics Laboratory, Pomeranian Medical University, 71-252 Szczecin, Poland; [email protected] 5 Department of Clinical Immunology and Immunotherapy, Medical University of Lublin, 20-093 Lublin, Poland; [email protected] 6 Department of Anesthesiology and Intensive Therapy, Pomeranian Medical University in Szczecin, 71-252 Szczecin, Poland; [email protected] 7 Clinical Department of Nephrology, Transplantology and Internal Medicine, Pomeranian Medical University, 70-111 Szczecin, Poland; [email protected] 8 Department of Infectious, Tropical Diseases and Immune Deficiency, Pomeranian Medical University in Szczecin, 71-455 Szczecin, Poland; [email protected] * Correspondence: katarzyna.kotfi[email protected]; Tel.: +48-91-466-11-44 Abstract: In March 2020, coronavirus disease 2019 (COVID-19) caused by SARS-CoV-2 was declared Citation: Kotfis, K.; Lechowicz, K.; a global pandemic by the World Health Organization (WHO). The clinical course of the disease is Drozd˙ zal,˙ S.; Nied´zwiedzka-Rystwej, unpredictable but may lead to severe acute respiratory infection (SARI) and pneumonia leading to P.; Wojdacz, T.K.; Grywalska, E.; acute respiratory distress syndrome (ARDS). -

Hormonal Treatment Strategies Tailored to Non-Binary Transgender Individuals
Journal of Clinical Medicine Review Hormonal Treatment Strategies Tailored to Non-Binary Transgender Individuals Carlotta Cocchetti 1, Jiska Ristori 1, Alessia Romani 1, Mario Maggi 2 and Alessandra Daphne Fisher 1,* 1 Andrology, Women’s Endocrinology and Gender Incongruence Unit, Florence University Hospital, 50139 Florence, Italy; [email protected] (C.C); jiska.ristori@unifi.it (J.R.); [email protected] (A.R.) 2 Department of Experimental, Clinical and Biomedical Sciences, Careggi University Hospital, 50139 Florence, Italy; [email protected]fi.it * Correspondence: fi[email protected] Received: 16 April 2020; Accepted: 18 May 2020; Published: 26 May 2020 Abstract: Introduction: To date no standardized hormonal treatment protocols for non-binary transgender individuals have been described in the literature and there is a lack of data regarding their efficacy and safety. Objectives: To suggest possible treatment strategies for non-binary transgender individuals with non-standardized requests and to emphasize the importance of a personalized clinical approach. Methods: A narrative review of pertinent literature on gender-affirming hormonal treatment in transgender persons was performed using PubMed. Results: New hormonal treatment regimens outside those reported in current guidelines should be considered for non-binary transgender individuals, in order to improve psychological well-being and quality of life. In the present review we suggested the use of hormonal and non-hormonal compounds, which—based on their mechanism of action—could be used in these cases depending on clients’ requests. Conclusion: Requests for an individualized hormonal treatment in non-binary transgender individuals represent a future challenge for professionals managing transgender health care. For each case, clinicians should balance the benefits and risks of a personalized non-standardized treatment, actively involving the person in decisions regarding hormonal treatment. -

Classification and Pharmacology of Progestins
Maturitas 46S1 (2003) S7–S16 Classification and pharmacology of progestins Adolf E. Schindler a,∗, Carlo Campagnoli b, René Druckmann c, Johannes Huber d, Jorge R. Pasqualini e, Karl W. Schweppe f, Jos H. H. Thijssen g a Institut für Medizinische Forschung und Fortbildung, Universitätsklinikum, Hufelandstr. 55, Essen 45147, Germany b Ospedale Ginecologico St. Anna, Corso Spezia 60, 10126 Torino, Italy c Ameno-Menopause-Center, 12, Rue de France, 06000 Nice, France d Abt. für Gynäkologische Endokrinologie, AKH Wien, Währingergürtel 18-20, 1090 Wien, Austria e Institute de Puériculture26, Boulevard Brune, 75014 Paris, France f Abt. für Gynäkologie und Geburtshilfe, Ammerland Klinik, Langestr.38, 26622 Westerstede, Germany g Department of Endocrinology, Universitair Medisch Centrum Utrecht, P.O. Box 85090, 3508 AB Utrecht, The Netherlands Abstract Besides the natural progestin, progesterone, there are different classes of progestins, such as retroprogesterone (i.e. dydroges- terone), progesterone derivatives (i.e. medrogestone) 17␣-hydroxyprogesterone derivatives (i.e. chlormadinone acetate, cypro- terone acetate, medroxyprogesterone acetate, megestrol acetate), 19-norprogesterone derivatives (i.e. nomegestrol, promege- stone, trimegestone, nesterone), 19-nortestosterone derivatives norethisterone (NET), lynestrenol, levonorgestrel, desogestrel, gestodene, norgestimate, dienogest) and spironolactone derivatives (i.e. drospirenone). Some of the synthetic progestins are prodrugs, which need to be metabolized to become active compounds. Besides -

Epigenetic Therapy with Panobinostat Combined with Bicalutamide Rechallenge in Castration- Resistant Prostate Cancer Anna C
Published OnlineFirst January 3, 2019; DOI: 10.1158/1078-0432.CCR-18-1589 Clinical Trials: Targeted Therapy Clinical Cancer Research Epigenetic Therapy with Panobinostat Combined with Bicalutamide Rechallenge in Castration- Resistant Prostate Cancer Anna C. Ferrari1, Joshi J. Alumkal2, Mark N. Stein3, Mary-Ellen Taplin4, James Babb5, Ethan S. Barnett6, Alejandro Gomez-Pinillos5, Xiaomei Liu5, Dirk Moore6, Robert DiPaola7, and Tomasz M. Beer2 Abstract Purpose: This study assesses the action of panobino- Results: In the model, panobinostat/bicalutamide demon- stat, a histone deacetylase inhibitor (HDACI), in restor- strated synergistic antitumor effect while reducing AR activity. ing sensitivity to bicalutamide in a castration-resistant The dose-limiting toxicity was not reached. The probabilities of prostate cancer (CRPC) model and the efficacy and safety remaining rPF were 47.5% in the A arm and 38.5% in the B arm, of the panobinostat/bicalutamide combination in CRPC exceeding the protocol-specified threshold of 35%. The A arm patients resistant to second-line antiandrogen therapy but not the B arm exceeded expectations for times (medians) to (2ndLAARx). rP (33.9 and 10 weeks), and from PSA progression to rP (24 and Patients and Methods: The CWR22PC xenograft and 5.9 weeks). A arm/B arm events included: adverse events (AE), isogenic cell line were tested for drug interactions on 62%/19%; treatment stopped for AEs, 27.5%/11.5%; dose tumor cell growth and on the androgen receptor (AR), reduction required, 41%/4%. The principal A-arm grade 3 AR-splice variant7, and AR targets. A phase I trial had a AEs were thrombocytopenia (31%) and fatigue (14%). -

Comparing the Effects of Combined Oral Contraceptives Containing Progestins with Low Androgenic and Antiandrogenic Activities on the Hypothalamic-Pituitary-Gonadal Axis In
JMIR RESEARCH PROTOCOLS Amiri et al Review Comparing the Effects of Combined Oral Contraceptives Containing Progestins With Low Androgenic and Antiandrogenic Activities on the Hypothalamic-Pituitary-Gonadal Axis in Patients With Polycystic Ovary Syndrome: Systematic Review and Meta-Analysis Mina Amiri1,2, PhD, Postdoc; Fahimeh Ramezani Tehrani2, MD; Fatemeh Nahidi3, PhD; Ali Kabir4, MD, MPH, PhD; Fereidoun Azizi5, MD 1Students Research Committee, School of Nursing and Midwifery, Department of Midwifery and Reproductive Health, Shahid Beheshti University of Medical Sciences, Tehran, Islamic Republic Of Iran 2Reproductive Endocrinology Research Center, Research Institute for Endocrine Sciences, Shahid Beheshti University of Medical Sciences, Tehran, Islamic Republic Of Iran 3School of Nursing and Midwifery, Department of Midwifery and Reproductive Health, Shahid Beheshti University of Medical Sciences, Tehran, Islamic Republic Of Iran 4Minimally Invasive Surgery Research Center, Iran University of Medical Sciences, Tehran, Islamic Republic Of Iran 5Endocrine Research Center, Shahid Beheshti University of Medical Sciences, Tehran, Islamic Republic Of Iran Corresponding Author: Fahimeh Ramezani Tehrani, MD Reproductive Endocrinology Research Center Research Institute for Endocrine Sciences Shahid Beheshti University of Medical Sciences 24 Parvaneh Yaman Street, Velenjak, PO Box 19395-4763 Tehran, 1985717413 Islamic Republic Of Iran Phone: 98 21 22432500 Email: [email protected] Abstract Background: Different products of combined oral contraceptives (COCs) can improve clinical and biochemical findings in patients with polycystic ovary syndrome (PCOS) through suppression of the hypothalamic-pituitary-gonadal (HPG) axis. Objective: This systematic review and meta-analysis aimed to compare the effects of COCs containing progestins with low androgenic and antiandrogenic activities on the HPG axis in patients with PCOS. -
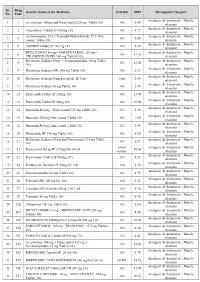
PMBJP Product.Pdf
Sr. Drug Generic Name of the Medicine Unit Size MRP Therapeutic Category No. Code Analgesic & Antipyretic / Muscle 1 1 Aceclofenac 100mg and Paracetamol 325 mg Tablet 10's 10's 8.00 relaxants Analgesic & Antipyretic / Muscle 2 2 Aceclofenac Tablets IP 100mg 10's 10's 4.37 relaxants Acetaminophen 325 + Tramadol Hydrochloride 37.5 film Analgesic & Antipyretic / Muscle 3 4 10's 8.00 coated Tablet 10's relaxants Analgesic & Antipyretic / Muscle 4 5 ASPIRIN Tablets IP 150 mg 14's 14's 2.70 relaxants DICLOFENAC 50 mg+ PARACETAMOL 325 mg+ Analgesic & Antipyretic / Muscle 5 6 10's 11.30 CHLORZOXAZONE 500 mg Tablets 10's relaxants Diclofenac Sodium 50mg + Serratiopeptidase 10mg Tablet Analgesic & Antipyretic / Muscle 6 8 10's 12.00 10's relaxants Analgesic & Antipyretic / Muscle 7 9 Diclofenac Sodium (SR) 100 mg Tablet 10's 10's 6.12 relaxants Analgesic & Antipyretic / Muscle 8 10 Diclofenac Sodium 25mg per ml Inj. IP 3 ml 3 ml 2.00 relaxants Analgesic & Antipyretic / Muscle 9 11 Diclofenac Sodium 50 mg Tablet 10's 10's 2.90 relaxants Analgesic & Antipyretic / Muscle 10 12 Etoricoxilb Tablets IP 120mg 10's 10's 33.00 relaxants Analgesic & Antipyretic / Muscle 11 13 Etoricoxilb Tablets IP 90mg 10's 10's 25.00 relaxants Analgesic & Antipyretic / Muscle 12 14 Ibuprofen 400 mg + Paracetamol 325 mg Tablet 10's 15's 5.50 relaxants Analgesic & Antipyretic / Muscle 13 15 Ibuprofen 200 mg film coated Tablet 10's 10's 1.80 relaxants Analgesic & Antipyretic / Muscle 14 16 Ibuprofen 400 mg film coated Tablet 10's 15's 3.50 relaxants Analgesic & Antipyretic -

Megestrol Acetate/Melengestrol Acetate 2115 Adverse Effects and Precautions Megestrol Acetate Is Also Used in the Treatment of Ano- 16
Megestrol Acetate/Melengestrol Acetate 2115 Adverse Effects and Precautions Megestrol acetate is also used in the treatment of ano- 16. Mwamburi DM, et al. Comparing megestrol acetate therapy with oxandrolone therapy for HIV-related weight loss: similar As for progestogens in general (see Progesterone, rexia and cachexia (see below) in patients with cancer results in 2 months. Clin Infect Dis 2004; 38: 895–902. p.2125). The weight gain that may occur with meges- or AIDS. The usual dose is 400 to 800 mg daily, as tab- 17. Grunfeld C, et al. Oxandrolone in the treatment of HIV-associ- ated weight loss in men: a randomized, double-blind, placebo- trol acetate appears to be associated with an increased lets or oral suspension. A suspension of megestrol ace- controlled study. J Acquir Immune Defic Syndr 2006; 41: appetite and food intake rather than with fluid reten- tate that has an increased bioavailability is also availa- 304–14. tion. Megestrol acetate may have glucocorticoid ef- ble (Megace ES; Par Pharmaceutical, USA) and is Hot flushes. Megestrol has been used to treat hot flushes in fects when given long term. given in a dose of 625 mg in 5 mL daily for anorexia, women with breast cancer (to avoid the potentially tumour-stim- cachexia, or unexplained significant weight loss in pa- ulating effects of an oestrogen—see Malignant Neoplasms, un- Effects on carbohydrate metabolism. Megestrol therapy der Precautions of HRT, p.2075), as well as in men with hot 1-3 4 tients with AIDS. has been associated with hyperglycaemia or diabetes mellitus flushes after orchidectomy or anti-androgen therapy for prostate in AIDS patients being treated for cachexia. -

Drug-Induced Sexual Dysfunction in Men and Women
VOLUME 36 : NUMBER 2 : APRIL 2013 ARTICLE Drug-induced sexual dysfunction in men and women Helen M Conaglen whether the clinician is willing to ask about sexual Clinical psychologist and SUMMARY issues and does so in a sensitive way.7,8 Senior research fellow Many medical conditions and their treatments Patients on long-term medications may not be John V Conaglen aware that their sexual problems have developed Endocrinologist and contribute to sexual dysfunction. as a result of their treatment. Conversely some may Associate professor in Commonly implicated drugs include Medicine blame their drugs for sexual problems which are due Sexual Health Research Unit antihypertensives, antidepressants, to relationship difficulties or other stressors. Some Waikato Clinical School antipsychotics and antiandrogens. doctors consider that asking patients if they had Faculty of Medical and Understanding the potential for drug-induced noticed any sexual adverse effects from their drugs Health Sciences University of Auckland sexual problems and their negative impact may ‘suggest’ them to the patient, and possibly result on adherence to treatment will enable the in non-adherence. Patients attributing their sexual clinician to tailor treatments for the patient problems to their drugs are less likely to continue the Key words treatment even when necessary for their health.9 The antidepressants, and his or her partner. consultation should include discussion of the patient’s antihypertensives, Encouraging a discussion with the patient sexual issues so these can be considered in treatment antipsychotics, arousal, about sexual function and providing erectile dysfunction, decisions. hypoactive sexual desire strategies to manage the problem are critical disorder, male impotence, to good clinical care. -

U.S. Medical Eligibility Criteria for Contraceptive Use, 2016
Morbidity and Mortality Weekly Report Recommendations and Reports / Vol. 65 / No. 3 July 29, 2016 U.S. Medical Eligibility Criteria for Contraceptive Use, 2016 U.S. Department of Health and Human Services Centers for Disease Control and Prevention Recommendations and Reports CONTENTS Introduction ............................................................................................................1 Methods ....................................................................................................................2 How to Use This Document ...............................................................................3 Keeping Guidance Up to Date ..........................................................................5 References ................................................................................................................8 Abbreviations and Acronyms ............................................................................9 Appendix A: Summary of Changes from U.S. Medical Eligibility Criteria for Contraceptive Use, 2010 ...........................................................................10 Appendix B: Classifications for Intrauterine Devices ............................. 18 Appendix C: Classifications for Progestin-Only Contraceptives ........ 35 Appendix D: Classifications for Combined Hormonal Contraceptives .... 55 Appendix E: Classifications for Barrier Methods ..................................... 81 Appendix F: Classifications for Fertility Awareness–Based Methods ..... 88 Appendix G: Lactational -

Dative Metabolism of Cortisol in Humans Kunihi
臨 床 薬 理JPn J Clin Pharmacol Ther 18 (3) Sept 1987 509 Comparison of the Inhibitory Effects of Famotidine and Cimetidine on Hepatic Oxi- dative Metabolism of Cortisol in Humans Kunihiko MORITA* Hiroki KONISHI* Takeshi ONO* and Harumi SHIMAKAWA* (Receivedon Dec.19, 1986) * Hospital Pharmacy, Shiga Universityof Medical Science, Tsukinowa-cho,Seta, Ohtsu520-21, Japan The inhibitory effect of famotidine, a new H2-receptor antagonist, on hepatic oxidative metabolism of cortisol in six healthy volunteers was compared with that of cimetidine by monitoring the change in urinary 6ƒÀ-hydroxycortisol (6ƒÀ-OHF), an oxidative metabolite of cortisol. The ratio of 6ƒÀ-OHF to 17-hydroxycorticosteroids (17-OHCS) in urine was measured before, during, and after treatment with famotidine and cimetidine for 3 days in a cross-over study. The ratio was decreased by 25% -35% of the original level after 1-3 days of oral treatment with cimetidine (800 mg, b. i. d.). The reduction vanished within 2 days after the last dose of cimetidine. The ratio was not significantly changed during oral treatment with famotidine (40 mg, b. i. d.). These findings indicate that famotidine, in contrast to cimetidine, does not affect the hepatic oxidative metabolism of cortisol in man, and it is suggested that famotidine does not affect the hepatic drug-metabolizing capacity in humans. Key words: famotidine, cimetidine, H2-receptor antangonist, 6ƒÀ-hydroxycortisol, enzyme inhibition been shown to inhibit the hepatic elimination of a Introduction number of drugs coadministred.1-4) A number of Cimetidine, a histamine H2-receptor antagonist studies have demonstrated that this inhibitory widely used for the treatment of peptic ulcer, has action of cimetidine is based on its high affinity for cytochrome P-450 in hepatic microsomes *滋 賀医科大学医学部附属病院薬剤部 arising from imidazole and cyano groups.5-7) 〓520-21滋 賀県大津市瀬田月輪町 Ranitidine, an H2-receptor antagonist developed 510 hospital pharmacists, aged 24 to 35 years old (mean 28), participated in the study.