Hydrophobicity, Hydrophilicity
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Heterogeneous Impacts of Protein-Stabilizing Osmolytes On
bioRxiv preprint doi: https://doi.org/10.1101/328922; this version posted May 23, 2018. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Heterogeneous Impacts of Protein-Stabilizing Osmolytes on Hydrophobic Interaction Mrinmoy Mukherjee and Jagannath Mondal⇤ Tata Institute of Fundamental Research Hyderabad, 500107 India E-mail: [email protected],+914020203091 Abstract Osmolytes’ mechanism of protecting proteins against denaturation is a longstanding puzzle, further complicated by the complex diversities inherent in protein sequences. An emergent approach in understanding osmolytes’ mechanism of action towards biopoly- mer has been to investigate osmolytes’ interplay with hydrophobic interaction, the ma- jor driving force of protein folding. However, the crucial question is whether all these protein-stabilizing osmolytes display a single unified mechanism towards hydrophobic interactions. By simulating the hydrophobic collapse of a macromolecule in aqueous solutions of two such osmoprotectants, Glycine and Trimethyl N-oxide (TMAO), both of which are known to stabilize protein’s folded conformation, we here demonstrate that these two osmolytes can impart mutually contrasting effects towards hydropho- bic interaction. While TMAO preserves its protectant nature across diverse range of polymer-osmolyte interactions, glycine is found to display an interesting cross-over from being a protectant at weaker polymer-osmolyte interaction to a denaturant of hydrophobicity at stronger polymer-osmolyte interactions. A preferential-interaction analysis reveals that a subtle balance of conformation-dependent exclusion/binding of ⇤To whom correspondence should be addressed 1 bioRxiv preprint doi: https://doi.org/10.1101/328922; this version posted May 23, 2018. -

Protein Structure & Folding
6 Protein Structure & Folding To understand protein folding In the last chapter we learned that proteins are composed of amino acids Goal as a chemical equilibrium. linked together by peptide bonds. We also learned that the twenty amino acids display a wide range of chemical properties. In this chapter we will see Objectives that how a protein folds is determined by its amino acid sequence and that the three-dimensional shape of a folded protein determines its function by After this chapter, you should be able to: the way it positions these amino acids. Finally, we will see that proteins fold • describe the four levels of protein because doing so minimizes Gibbs free energy and that this minimization structure and the thermodynamic involves both making the most favorable bonds and maximizing disorder. forces that stabilize them. • explain how entropy (S) and enthalpy Proteins exhibit four levels of structure (H) contribute to Gibbs free energy. • use the equation ΔG = ΔH – TΔS The structure of proteins can be broken down into four levels of to determine the dependence of organization. The first is primary structure, the linear sequence of amino the favorability of a reaction on acids in the polypeptide chain. By convention, the primary sequence is temperature. written in order from the amino acid at the N-terminus (by convention • explain the hydrophobic effect and its usually on the left) to the amino acid at the C-terminus. The second level role in protein folding. of protein structure, secondary structure, is the local conformation adopted by stretches of contiguous amino acids. -

Substrate Wettability Guided Oriented Self Assembly of Janus Particles
www.nature.com/scientificreports OPEN Substrate wettability guided oriented self assembly of Janus particles Meneka Banik1, Shaili Sett2, Chirodeep Bakli3, Arup Kumar Raychaudhuri2, Suman Chakraborty4* & Rabibrata Mukherjee1* Self-assembly of Janus particles with spatial inhomogeneous properties is of fundamental importance in diverse areas of sciences and has been extensively observed as a favorably functionalized fuidic interface or in a dilute solution. Interestingly, the unique and non-trivial role of surface wettability on oriented self-assembly of Janus particles has remained largely unexplored. Here, the exclusive role of substrate wettability in directing the orientation of amphiphilic metal-polymer Bifacial spherical Janus particles, obtained by topo-selective metal deposition on colloidal Polymestyere (PS) particles, is explored by drop casting a dilute dispersion of the Janus colloids. While all particles orient with their polymeric (hydrophobic) and metallic (hydrophilic) sides facing upwards on hydrophilic and hydrophobic substrates respectively, they exhibit random orientation on a neutral substrate. The substrate wettability guided orientation of the Janus particles is captured using molecular dynamic simulation, which highlights that the arrangement of water molecules and their local densities near the substrate guide the specifc orientation. Finally, it is shown that by spin coating it becomes possible to create a hexagonal close-packed array of the Janus colloids with specifc orientation on diferential wettability substrates. -

Topological Analysis of the Metal-Metal Bond: a Tutorial Review Christine Lepetit, Pierre Fau, Katia Fajerwerg, Myrtil L
Topological analysis of the metal-metal bond: A tutorial review Christine Lepetit, Pierre Fau, Katia Fajerwerg, Myrtil L. Kahn, Bernard Silvi To cite this version: Christine Lepetit, Pierre Fau, Katia Fajerwerg, Myrtil L. Kahn, Bernard Silvi. Topological analysis of the metal-metal bond: A tutorial review. Coordination Chemistry Reviews, Elsevier, 2017, 345, pp.150-181. 10.1016/j.ccr.2017.04.009. hal-01540328 HAL Id: hal-01540328 https://hal.sorbonne-universite.fr/hal-01540328 Submitted on 16 Jun 2017 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Topological analysis of the metal-metal bond: a tutorial review Christine Lepetita,b, Pierre Faua,b, Katia Fajerwerga,b, MyrtilL. Kahn a,b, Bernard Silvic,∗ aCNRS, LCC (Laboratoire de Chimie de Coordination), 205, route de Narbonne, BP 44099, F-31077 Toulouse Cedex 4, France. bUniversité de Toulouse, UPS, INPT, F-31077 Toulouse Cedex 4, i France cSorbonne Universités, UPMC, Univ Paris 06, UMR 7616, Laboratoire de Chimie Théorique, case courrier 137, 4 place Jussieu, F-75005 Paris, France Abstract This contribution explains how the topological methods of analysis of the electron density and related functions such as the electron localization function (ELF) and the electron localizability indicator (ELI-D) enable the theoretical characterization of various metal-metal (M-M) bonds (multiple M-M bonds, dative M-M bonds). -
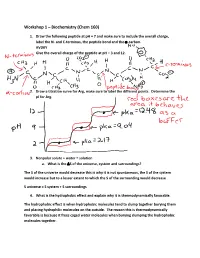
Workshop 1 – Biochemistry (Chem 160)
Workshop 1 – Biochemistry (Chem 160) 1. Draw the following peptide at pH = 7 and make sure to include the overall charge, label the N- and C-terminus, the peptide bond and the -carbon. AVDKY Give the overall charge of the peptide at pH = 3 and 12. 2. Draw a titration curve for Arg, make sure to label the different points. Determine the pI for Arg. 3. Nonpolar solute + water = solution a. What is the S of the universe, system and surroundings? The S of the universe would decrease this is why it is not spontaneous, the S of the system would increase but to a lesser extent to which the S of the surrounding would decrease S universe = S system + S surroundings 4. What is the hydrophobic effect and explain why it is thermodynamically favorable. The hydrophobic effect is when hydrophobic molecules tend to clump together burying them and placing hydrophilic molecules on the outside. The reason this is thermodynamically favorable is because it frees caged water molecules when burying clumping the hydrophobic molecules together. 5. Urea dissolves very readily in water, but the solution becomes very cold as the urea dissolves. How is this possible? Urea dissolves in water because when dissolving there is a net increase in entropy of the universe. The heat exchange, getting colder only reflects the enthalpy (H) component of the total energy change. The entropy change is high enough to offset the enthalpy component and to add up to an overall -G 6. A mutation that changes an alanine residue in the interior of a protein to valine is found to lead to a loss of activity. -

Introduction to Aromaticity
Introduction to Aromaticity Historical Timeline:1 Spotlight on Benzene:2 th • Early 19 century chemists derive benzene formula (C6H6) and molecular mass (78). • Carbon to hydrogen ratio of 1:1 suggests high reactivity and instability. • However, benzene is fairly inert and fails to undergo reactions that characterize normal alkenes. - Benzene remains inert at room temperature. - Benzene is more resistant to catalytic hydrogenation than other alkenes. Possible (but wrong) benzene structures:3 Dewar benzene Prismane Fulvene 2,4- Hexadiyne - Rearranges to benzene at - Rearranges to - Undergoes catalytic - Undergoes catalytic room temperature. Faraday’s benzene. hydrogenation easily. hydrogenation easily - Lots of ring strain. - Lots of ring strain. - Lots of ring strain. 1 Timeline is computer-generated, compiled with information from pg. 594 of Bruice, Organic Chemistry, 4th Edition, Ch. 15.2, and from Chemistry 14C Thinkbook by Dr. Steven Hardinger, Version 4, p. 26 2 Chemistry 14C Thinkbook, p. 26 3 Images of Dewar benzene, prismane, fulvene, and 2,4-Hexadiyne taken from Chemistry 14C Thinkbook, p. 26. Kekulé’s solution: - “snake bites its own tail” (4) Problems with Kekulé’s solution: • If Kekulé’s structure were to have two chloride substituents replacing two hydrogen atoms, there should be a pair of 1,2-dichlorobenzene isomers: one isomer with single bonds separating the Cl atoms, and another with double bonds separating the Cl atoms. • These isomers were never isolated or detected. • Rapid equilibrium proposed, where isomers interconvert so quickly that they cannot be isolated or detected. • Regardless, Kekulé’s structure has C=C’s and normal alkene reactions are still expected. - But the unusual stability of benzene still unexplained. -

Symmetry of Three-Center, Four-Electron Bonds†‡
Chemical Science EDGE ARTICLE View Article Online View Journal | View Issue Symmetry of three-center, four-electron bonds†‡ b a c Cite this: Chem. Sci., 2020, 11,7979 Ann Christin Reiersølmoen, § Stefano Battaglia, § Sigurd Øien-Ødegaard, Arvind Kumar Gupta, d Anne Fiksdahl,b Roland Lindh a and Mate Erdelyi *a All publication charges for this article ´ ´ ´ have been paid for by the Royal Society of Chemistry Three-center, four-electron bonds provide unusually strong interactions; however, their nature remains ununderstood. Investigations of the strength, symmetry and the covalent versus electrostatic character of three-center hydrogen bonds have vastly contributed to the understanding of chemical bonding, whereas the assessments of the analogous three-center halogen, chalcogen, tetrel and metallic s^-type long bonding are still lagging behind. Herein, we disclose the X-ray crystallographic, NMR spectroscopic and computational investigation of three-center, four-electron [D–X–D]+ bonding for a variety of cations (X+ ¼ H+,Li+,Na+,F+,Cl+,Br+,I+,Ag+ and Au+) using a benchmark bidentate model system. Formation of a three-center bond, [D–X–D]+ is accompanied by an at least 30% shortening of the D–X Received 11th April 2020 bonds. We introduce a numerical index that correlates symmetry to the ionic size and the electron Accepted 19th June 2020 affinity of the central cation, X+. Providing an improved understanding of the fundamental factors DOI: 10.1039/d0sc02076a Creative Commons Attribution 3.0 Unported Licence. determining bond symmetry on a comprehensive level is expected to facilitate future developments and rsc.li/chemical-science applications of secondary bonding and hypervalent chemistry. -

Inverse Gas Chromatographic Examination of Polymer Composites
Open Chem., 2015; 13: 893–900 Invited Paper Open Access Adam Voelkel*, Beata Strzemiecka, Kasylda Milczewska, Zuzanna Okulus Inverse Gas Chromatographic Examination of Polymer Composites DOI: 10.1515/chem-2015-0104 received December 30, 2014; accepted April 1, 2015. of composite components and/or interactions between them, and behavior during technological processes. This paper reviews is the examination of Abstract: Inverse gas chromatographic characterization various polymer-containing systems by inverse gas of resins and resin based abrasive materials, polymer- chromatography. polymer and polymer-filler systems, as well as dental restoratives is reviewed. Keywords: surface activity, polymer-polymer interactions, 2 Discussion adhesion, dental restoratives, inverse gas chromatography 2.1 Surface energy and adhesion 1 Introduction IGC is useful for surface energy determination of solid polymers and fillers. Solid surface energy of consists of Inverse gas chromatography (IGC) was introduced in 1967 dispersive ( , from van der Waals forces) and specific by Kiselev [1], developed by Smidsrød and Guillet [2], and ( , from acid-base interactions) components: is still being improved. Its popularity is due to its simplicity D and user friendliness. Only a standard gas chromatograph = + (1) is necessary [3] although more sophisticated equipment has been advised. “Inverse” relates to the aim of the experiment. for solids can be calculated according to several It is not separation as in classical GC, but examination of methods [7]; one of the most often used is that of Schultz- the stationary phase properties. Test compounds with Lavielle [8-12]: known properties are injected onto the column containing dd the material to be examined. Retention times and peak RT××ln VN =× 2 N ×× aggsl × + C (2) profiles determine parameters describing the column filling. -

Nanoparticles Improving the Wetting Ability of Biological Liquids
dynam mo ic Torrisi and Scolaro, J Thermodyn Catal 2017, 8:1 er s h & T f C DOI: 10.4172/2157-7544.1000184 o a t l a a l Journal of y n r s i u s o J ISSN: 2157-7544 Thermodynamics & Catalysis Research Article Open Access Nanoparticles Improving the Wetting Ability of Biological Liquids Torrisi L* and Scolaro C Dottorato di Ricerca in Fisica and Dipartimento di Scienze Fisiche MIFT, Università di Messina, V.le F.S. D’Alcontres 31, 98166 S. Agata (ME), Italy Abstract The contact angle of some biological liquids was investigated on different biocompatible surfaces and nanoparticles were employed to improve their wetting ability. Biocompatible nanoparticles based on titanium, silver and gold were prepared using the technique of the pulsed laser ablation in liquids. The nanoparticles were characterized in size, shape, coalescence and concentration in solutions. Solid biocompatible substrates based on polyethylene, hydroxyapatite and titanium were employed as simulation of prosthetic surfaces. Measurements of contact angle were performed in different biological solution, including the human blood, with respect to the distilled water, without and with the use of the metallic nanoparticles at concentration of the order of 80-370 µg/ml. Keywords: Nanoparticles; Laser ablation; Contact angle; viscosity, thermal and electric conduction, vapour pressure, the PH Biocompatibility; Interfaces and other parameters [5,9]. The wetting ability of the liquid on a given surface changes introducing specific nanoparticles at the interface. This Introduction aspect is of special interest in Biology and Medicine, especially due More and more the nanotechnology is becoming important to the impact on the protein absorption and the cellular response in and promising in different fields of the science. -

2.#Water;#Acid.Base#Reac1ons
8/24/15 BIOCH 755: Biochemistry I Fall 2015 2.#Water;#Acid.base#reac1ons# Jianhan#Chen# Office#Hour:#M#1:30.2:30PM,#Chalmers#034# Email:#[email protected]# Office:#785.2518# 2.1#Physical#Proper1es#of#Water# • Key$Concepts$2.1$ – Water#molecules,#which#are#polar,#can#form#hydrogen#bonds#with# other#molecules.# – In#ice,#water#molecules#are#hydrogen#bonded#in#a#crystalline#array,#but# in#liquid#water,#hydrogen#bonds#rapidly#break#and#re.form#in#irregular# networks.# – The#aTrac1ve#forces#ac1ng#on#biological#molecules#include#ionic# interac1ons,#hydrogen#bonds,#and#van#der#Waals#interac1ons.# – Polar#and#ionic#substances#can#dissolve#in#water.# (c)#Jianhan#Chen# 2# 1 8/24/15 2.1#Physical#Proper1es#of#Water# • Key$Concepts$2.1$ – The#hydrophobic#effect#explains#the#exclusion#of#nonpolar#groups#as#a# way#to#maximize#the#entropy#of#water#molecules.# – Amphiphilic#substances#form#micelles#or#bilayers#that#hide#their# hydrophobic#groups#while#exposing#their#hydrophilic#groups#to#water.# – Molecules#diffuse#across#membranes#which#are#permeable#to#them# from#regions#of#higher#concentra1on#to#regions#of#lower# concentra1on.# – In#dialysis,#solutes#diffuse#across#a#semipermeable#membrane#from# regions#of#higher#concentra1on#to#regions#of#lower#concentra1on.# (c)#Jianhan#Chen# 3# Human#Body#Mass#Composi1on# (c)#Jianhan#Chen# 4 2 8/24/15 Structure#of#Water# (c)#Jianhan#Chen# 5 Water#Hydrogen#Bonding# ~1.8 Å, 180o Acceptor Donor (c)#Jianhan#Chen# 6# 3 8/24/15 Typical#Bond#Energies# (c)#Jianhan#Chen# 7# Hydrogen#bond#networks#of#water/ice# (c)#Jianhan#Chen# -

Hydrophobic Effect, Water Structure, and Heat Capacity Changes
J. Phys. Chem. B 1997, 101, 4343-4348 4343 Hydrophobic Effect, Water Structure, and Heat Capacity Changes Kim A. Sharp* and Bhupinder Madan Department of Biochemistry & Biophysics, UniVersity of PennsylVania, 3700 Hamilton Walk, Philadelphia, PennsylVania 19104-6059 ReceiVed: January 16, 1997; In Final Form: April 1, 1997X hyd The hydration heat capacity (∆Cp ) of nine solutes of varying hydrophobicity was studied using a combination of a random network model equation of water and Monte Carlo simulations. Nonpolar solutes cause a concerted decrease in the average length and angle of the water-water hydrogen bonds in the first hydration shell, while polar and ionic solutes have the opposite effect. This is due to changes in the amounts, relative to bulk water, of two populations of hydrogen bonds: one with shorter and more linear bonds and the other with longer and more bent bonds. Heat capacity changes were calculated from these changes in water structure using a random network model equation of state. The calculated changes account for observed hyd differences in ∆Cp for the various solutes. The simulations provide a unified picture of hydrophobic and hyd polar hydration, and both a structural and thermodynamic explanation of the opposite signs of ∆Cp observed for polar and nonpolar solutes. Introduction nonpolar groups means that at higher temperatures (T > 80 °C) their insolubility results from unfavorable changes in enthalpy, In recent years analysis of heat capacity changes has become not entropy. Second, the hydration of a majority of polar and of central importance in understanding the thermodynamics of ionic solutes is also accompanied by a decrease in entropy, and protein folding, protein-protein binding, protein-nucleic acid thus by some kind of structuring of water, but in this case binding, and the hydrophobic effect.1-7 There are several hyd 7,16-20 reasons for this. -

Basic Concepts of Chemical Bonding
Basic Concepts of Chemical Bonding Cover 8.1 to 8.7 EXCEPT 1. Omit Energetics of Ionic Bond Formation Omit Born-Haber Cycle 2. Omit Dipole Moments ELEMENTS & COMPOUNDS • Why do elements react to form compounds ? • What are the forces that hold atoms together in molecules ? and ions in ionic compounds ? Electron configuration predict reactivity Element Electron configurations Mg (12e) 1S 2 2S 2 2P 6 3S 2 Reactive Mg 2+ (10e) [Ne] Stable Cl(17e) 1S 2 2S 2 2P 6 3S 2 3P 5 Reactive Cl - (18e) [Ar] Stable CHEMICAL BONDSBONDS attractive force holding atoms together Single Bond : involves an electron pair e.g. H 2 Double Bond : involves two electron pairs e.g. O 2 Triple Bond : involves three electron pairs e.g. N 2 TYPES OF CHEMICAL BONDSBONDS Ionic Polar Covalent Two Extremes Covalent The Two Extremes IONIC BOND results from the transfer of electrons from a metal to a nonmetal. COVALENT BOND results from the sharing of electrons between the atoms. Usually found between nonmetals. The POLAR COVALENT bond is In-between • the IONIC BOND [ transfer of electrons ] and • the COVALENT BOND [ shared electrons] The pair of electrons in a polar covalent bond are not shared equally . DISCRIPTION OF ELECTRONS 1. How Many Electrons ? 2. Electron Configuration 3. Orbital Diagram 4. Quantum Numbers 5. LEWISLEWIS SYMBOLSSYMBOLS LEWISLEWIS SYMBOLSSYMBOLS 1. Electrons are represented as DOTS 2. Only VALENCE electrons are used Atomic Hydrogen is H • Atomic Lithium is Li • Atomic Sodium is Na • All of Group 1 has only one dot The Octet Rule Atoms gain, lose, or share electrons until they are surrounded by 8 valence electrons (s2 p6 ) All noble gases [EXCEPT HE] have s2 p6 configuration.