A Brief Introduction to Nerve Agents 27 Feb 2021
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Malathion General Fact Sheet
MALATHION GENERAL FACT SHEET What is malathion Malathion is an insecticide in the chemical family known as organophosphates. Products containing malathion are used outdoors to control a wide variety of insects in agricultural settings and around people’s homes. Malathion has also been used in public health mosquito control and fruit fly eradication programs. Malathion may also be found in some special shampoos for treating lice. Malathion was first registered for use in the United States in 1956. What are some products that contain malathion Products containing malathion may be liquids, dusts, wettable powders, or emulsions. There are thousands of products contain- ing malathion registered for use in the United States. Always follow label instructions and take steps to avoid exposure. If any exposures occur, be sure to follow the First Aid instructions on the product label carefully. For additional treatment advice, contact the Poison Control Center at 1-800-222-1222. If you wish to discuss a pesticide problem, please call 1-800-858-7378. How does malathion work Malathion kills insects by preventing their nervous system from working properly. When healthy nerves send signals to each other, a special chemical messenger travels from one nerve to another to continue the message. The nerve signal stops when an enzyme is released into the space between the nerves. Malathion binds to the enzyme and prevents the nerve signal from stopping. This causes the nerves to signal each other without stopping. The constant nerve signals make it so the insects can’t move or breathe normally and they die. People, pets and other animals can be affected the same way as insects if they are exposed to enough malathion. -

US EPA, Pesticide Product Label, ORTHO MALATHION 50 INSECT
23V 73^ f UNITED STATES ENVIRONMENTAL PROTECTION AGENCY WASHINGTON, D.C. 20460 Jff***^ OFFICE OF WAV 00 Onit CHEMICAL SAFETY AND MAY 4 0 201] POLLUTION PREVENTION James Wallace The Scotts Company d/b/a The Ortho Group P.O.Box 190 Marysville, OH 43040 Subject: Submission of amended labeling per May 2009 Malathion RED Label Table EPA Reg. No. 239-739 Submissions dated March 5, 2010 Dear Mr. Wallace: The proposed labeling referred to above, submitted in connection with registration under the Federal Insecticide, Fungicide, and Rodenticide Act (FIFRA), is acceptable under FIFRA § 3(c)(5) provided that you submit two copies of your final printed label before you release the product for shipment. Products shipped after 12 months from the date of this amendment or the next printing of the label whichever occurs first, must bear the new revised label. Your release for shipment of the product bearing the amended label constitutes acceptance of these conditions. If these conditions are not complied with, the registration will be subject to cancellation in accordance with FIFRA sec. 6(e). The Basic Confidential Statement of Formula (CSF) dated 2/19/10 is acceptable. All other CSF's including Alternates have been superseded by this new CSF. This has been added to your record. Your product has met and complied with the risk mitigation provisions of the Malathion RED and therefore your product is reregistered pursuant to FIFRA § 4(g)(2)(C). But note, EPA is currently reassessing the cumulative impacts of organophosphates and that review may impact your product once complete. -

Malathion) Lotion, 0.5% Rx Only
NDC 51672-5276-4 Ovide® (malathion) Lotion, 0.5% Rx Only For topical use only. Not for oral or ophthalmic use. DESCRIPTION OVIDE Lotion contains 0.005 g of malathion per mL in a vehicle of isopropyl alcohol (78%), terpineol, dipentene, and pine needle oil. The chemical name of malathion is (±) - [(dimethoxyphosphinothioyl) - thio] butanedioic acid diethyl ester. Malathion has a molecular weight of 330.36, represented by C10H19O6PS2, and has the following chemical structure: CLINICAL PHARMACOLOGY Malathion is an organophosphate agent which acts as a pediculicide by inhibiting cholinesterase activity in vivo. Inadvertent transdermal absorption of malathion has occurred from its agricultural use. In such cases, acute toxicity was manifested by excessive cholinergic activity, i.e., increased sweating, salivary and gastric secretion, gastrointestinal and uterine motility, and bradycardia (see OVERDOSAGE). Because the potential for transdermal absorption of malathion from OVIDE Lotion is not known at this time, strict adherence to the dosing instructions regarding its use in children, method of application, duration of exposure, and frequency of application is required. INDICATIONS AND USAGE OVIDE Lotion is indicated for patients infected with Pediculus humanus capitis (head lice and their ova) of the scalp hair. CONTRAINDICATIONS OVIDE Lotion is contraindicated for neonates and infants because their scalps are more permeable and may have increased absorption of malathion. OVIDE Lotion should also not be used on individuals known to be sensitive to malathion or any of the ingredients in the vehicle. WARNINGS 1. OVIDE Lotion is flammable. The lotion and wet hair should not be exposed to open flames or electric heat sources, including hair dryers and electric curlers. -

Chlorpyrifos (Dursban) Ddvp (Dichlorvos) Diazinon Malathion Parathion
CHLORPYRIFOS (DURSBAN) DDVP (DICHLORVOS) DIAZINON MALATHION PARATHION Method no.: 62 Matrix: Air Procedure: Samples are collected by drawing known volumes of air through specially constructed glass sampling tubes, each containing a glass fiber filter and two sections of XAD-2 adsorbent. Samples are desorbed with toluene and analyzed by GC using a flame photometric detector (FPD). Recommended air volume and sampling rate: 480 L at 1.0 L/min except for Malathion 60 L at 1.0 L/min for Malathion Target concentrations: 1.0 mg/m3 (0.111 ppm) for Dichlorvos (PEL) 0.1 mg/m3 (0.008 ppm) for Diazinon (TLV) 0.2 mg/m3 (0.014 ppm) for Chlorpyrifos (TLV) 15.0 mg/m3 (1.11 ppm) for Malathion (PEL) 0.1 mg/m3 (0.008 ppm) for Parathion (PEL) Reliable quantitation limits: 0.0019 mg/m3 (0.21 ppb) for Dichlorvos (based on the RAV) 0.0030 mg/m3 (0.24 ppb) for Diazinon 0.0033 mg/m3 (0.23 ppb) for Chlorpyrifos 0.0303 mg/m3 (2.2 ppb) for Malathion 0.0031 mg/m3 (0.26 ppb) for Parathion Standard errors of estimate at the target concentration: 5.3% for Dichlorvos (Section 4.6.) 5.3% for Diazinon 5.3% for Chlorpyrifos 5.6% for Malathion 5.3% for Parathion Status of method: Evaluated method. This method has been subjected to the established evaluation procedures of the Organic Methods Evaluation Branch. Date: October 1986 Chemist: Donald Burright Organic Methods Evaluation Branch OSHA Analytical Laboratory Salt Lake City, Utah 1 of 27 T-62-FV-01-8610-M 1. -

Malathion Human Health and Ecological Risk Assessment Final Report
SERA TR-052-02-02c Malathion Human Health and Ecological Risk Assessment Final Report Submitted to: Paul Mistretta, COR USDA/Forest Service, Southern Region 1720 Peachtree RD, NW Atlanta, Georgia 30309 USDA Forest Service Contract: AG-3187-C-06-0010 USDA Forest Order Number: AG-43ZP-D-06-0012 SERA Internal Task No. 52-02 Submitted by: Patrick R. Durkin Syracuse Environmental Research Associates, Inc. 5100 Highbridge St., 42C Fayetteville, New York 13066-0950 Fax: (315) 637-0445 E-Mail: [email protected] Home Page: www.sera-inc.com May 12, 2008 Table of Contents Table of Contents............................................................................................................................ ii List of Figures................................................................................................................................. v List of Tables ................................................................................................................................. vi List of Appendices ......................................................................................................................... vi List of Attachments........................................................................................................................ vi ACRONYMS, ABBREVIATIONS, AND SYMBOLS ............................................................... vii COMMON UNIT CONVERSIONS AND ABBREVIATIONS.................................................... x CONVERSION OF SCIENTIFIC NOTATION .......................................................................... -

Aldicarb by Caroline Cox Pulses Across the Junctions Between Nerves
Aldicarb By Caroline Cox pulses across the junctions between nerves. (given over a one-year period). The high- This causes loss of muscular coordination, est dose at which no adverse effects were In 1966, four years before the insecticide convulsions, and ultimately death.5 The observed (called the NOEL) was 0.02 mg/ 11 aldicarb was registered for use in the United AChE inhibition is said to be reversible be- kg/day, about one-tenth of the LD50. States, researchers were using the chemical cause the aldicarb disassociates from the There is evidence that people may suf- on an experimental basis.1 One researcher AChE within several hours. This occurs even fer acute symptoms when exposed to even brought a small amount home and his wife if death has occurred. Organophosphate in- lower levels of aldicarb. In humans who applied it to the soil under a backyard rose secticides (malathion and diazinon, for ex- consumed aldicarb-contaminated water- bush. Over three weeks later she ate a sprig ample) have the same mode of action ex- melons, clinical signs of aldicarb poison- of mint from a plant growing nearby. Within cept that the AChE inhibition is not as ing were found in individuals consuming half an hour she was suffering from vomit- readily reversible.7 as little as 0.002 mg/kg of the aldicarb ing, diarrhea, and involuntary urination. Her Aldicarb is a systemic insecticide. It is metabolite aldicarb sulfoxide. This pupils closed to pinpoints, her muscles applied as granules below the soil surface amount is one-tenth the NOEL in the twitched, and her breathing was difficult. -

Environmental Health Criteria 63 ORGANOPHOSPHORUS
Environmental Health Criteria 63 ORGANOPHOSPHORUS INSECTICIDES: A GENERAL INTRODUCTION Please note that the layout and pagination of this web version are not identical with the printed version. Organophophorus insecticides: a general introduction (EHC 63, 1986) INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY ENVIRONMENTAL HEALTH CRITERIA 63 ORGANOPHOSPHORUS INSECTICIDES: A GENERAL INTRODUCTION This report contains the collective views of an international group of experts and does not necessarily represent the decisions or the stated policy of the United Nations Environment Programme, the International Labour Organisation, or the World Health Organization. Published under the joint sponsorship of the United Nations Environment Programme, the International Labour Organisation, and the World Health Organization World Health Orgnization Geneva, 1986 The International Programme on Chemical Safety (IPCS) is a joint venture of the United Nations Environment Programme, the International Labour Organisation, and the World Health Organization. The main objective of the IPCS is to carry out and disseminate evaluations of the effects of chemicals on human health and the quality of the environment. Supporting activities include the development of epidemiological, experimental laboratory, and risk-assessment methods that could produce internationally comparable results, and the development of manpower in the field of toxicology. Other activities carried out by the IPCS include the development of know-how for coping with chemical accidents, coordination -

Pesticide Residues : Maximum Residue Limits
THAI AGRICULTURAL STANDARD TAS 9002-2013 PESTICIDE RESIDUES : MAXIMUM RESIDUE LIMITS National Bureau of Agricultural Commodity and Food Standards Ministry of Agriculture and Cooperatives ICS 67.040 ISBN UNOFFICAL TRANSLATION THAI AGRICULTURAL STANDARD TAS 9002-2013 PESTICIDE RESIDUES : MAXIMUM RESIDUE LIMITS National Bureau of Agricultural Commodity and Food Standards Ministry of Agriculture and Cooperatives 50 Phaholyothin Road, Ladyao, Chatuchak, Bangkok 10900 Telephone (662) 561 2277 Fascimile: (662) 561 3357 www.acfs.go.th Published in the Royal Gazette, Announcement and General Publication Volume 131, Special Section 32ง (Ngo), Dated 13 February B.E. 2557 (2014) (2) Technical Committee on the Elaboration of the Thai Agricultural Standard on Maximum Residue Limits for Pesticide 1. Mrs. Manthana Milne Chairperson Department of Agriculture 2. Mrs. Thanida Harintharanon Member Department of Livestock Development 3. Mrs. Kanokporn Atisook Member Department of Medical Sciences, Ministry of Public Health 4. Mrs. Chuensuke Methakulawat Member Office of the Consumer Protection Board, The Prime Minister’s Office 5. Ms. Warunee Sensupa Member Food and Drug Administration, Ministry of Public Health 6. Mr. Thammanoon Kaewkhongkha Member Office of Agricultural Regulation, Department of Agriculture 7. Mr. Pisan Pongsapitch Member National Bureau of Agricultural Commodity and Food Standards 8. Ms. Wipa Thangnipon Member Office of Agricultural Production Science Research and Development, Department of Agriculture 9. Ms. Pojjanee Paniangvait Member Board of Trade of Thailand 10. Mr. Charoen Kaowsuksai Member Food Processing Industry Club, Federation of Thai Industries 11. Ms. Natchaya Chumsawat Member Thai Agro Business Association 12. Mr. Sinchai Swasdichai Member Thai Crop Protection Association 13. Mrs. Nuansri Tayaputch Member Expert on Method of Analysis 14. -

Pesticide Resistance of Agricultural Pests in Japan
Pesticide Resistance of Agricultural Pests in Japan By TOSHIKAZU IWATA Chief, 3rd Laboratory of Insect Control Investigation, Department of Phytopathology and Entomology, National Institute of Agricultural Sciences A quarter century has passed since DDT other organophosphorus chemicals such as was firstly applied to control agricultural in methyl paration. As the result the parathion sect pests. The amount of synthesized organic resistant borer was prevented from spreading insecticides applied in fields increased rapidly by changing insecticide to be applied for the since 1952 and the rate of increase was about control. 10 per cent a year. In 1960 an evident report Control of the rice stem borer by BHC dust was made on insecticide resistance of insects, was established in about 1950, and application so 10 years have passed following the first of fine dust or granular formulation of BHC appearance of insecticide-resistant insects. to paddy fields became very popular in 1961 Today, pest species developed or suspected of or 1962. In 1964, declination of susceptibility resistance totals 11 species of insects and 6 of the borers to BHC was found in Kagawa species of mites as shown in Table 1. The Prefecture. After that, similar phenomena author reviews the circumstances of occur were reported in some other prefectures. The rence and some results of studies on major amount of BHC applied to paddy fields has instances of resistant pests. been decreased for its residue in rice plants. Consequently, BHC resistance of rice stem Rice stem borer, Chilo suppressalis borer may not become a serious problem in Walker future. Recently, declination of susceptibility of The rice stem borer is one of the most im the borer to fenitrothion is suspected in a portant pests of rice plants in Japan. -

United States Patent (19) 11 Patent Number: 5,703,064 Yokoi Et Al
US005703064A United States Patent (19) 11 Patent Number: 5,703,064 Yokoi et al. 45) Date of Patent: Dec. 30, 1997 54 PESTICIDAL COMBINATIONS FOREIGN PATENT DOCUMENTS 75 Inventors: Shinji Yokoi; Akira Nishida, both of 0 196524 10/1986 European Pat. Of.. Shiga-ken; Tokio Obata; Kouichi Golka, both of Ube, all of Japan OTHER PUBLICATIONS 73) Assignees: Sankyo Company, Limited, Tokyo; Worthing et al, The Pesticide Manual, 9th Ed. (1991), pp. Ube industries Ltd., Ube, both of 747 and 748. Japan L.C. Gaughan et al., "Pesticide interactions: effects of orga nophosphorus pesticides on the metabolism, toxicity, and 21 Appl. No.: 405,795 persistence of selected pyrethroid insecticides". Chemical Abstracts, vol. 94, No. 9, 1981, No. 59740k of Pestic. 22 Filed: Mar 16, 1995 Biochem. Physio., vol. 14, No. 1, 1980, pp. 81-85. 30 Foreign Application Priority Data I. Ishaaya et al., "Cypermethrin synergism by pyrethroid esterase inhibitors in adults of the whitefly Bemisia tabaci". Mar 16, 1994 JP Japan ............................ HE6045.405 Chemical Abstracts, vol. 107, No. 9, 1987, No. 72818y of (51) Int. Cl................. A01N 43/54; A01N 57/00 Pestic Biochem. Physiol., vol. 28, No. 2, 1987, pp. 155-162. 52 U.S. C. ......................................... 51480; 514/256 (58) Field of Search ..................................... 514/80, 256 Primary Examiner Allen J. Robinson Attorney, Agent, or Firm-Frishauf, Holtz, Goodman, 56 References Cited Langer & Chick, P.C. U.S. PATENT DOCUMENTS 57 ABSTRACT 4,374,833 2/1983 Badmin et al. ...................... 424/225 Combinations of the known compound pyrimidifen with 4,845,097 7/1989 Matsumoto et al... 514/234.2 phosphorus-containing pesticides have a synergistic pesti 4,935,516 6/1990 Ataka et al. -

NMP-Free Formulations of Neonicotinoids
(19) & (11) EP 2 266 400 A1 (12) EUROPEAN PATENT APPLICATION (43) Date of publication: (51) Int Cl.: 29.12.2010 Bulletin 2010/52 A01N 43/40 (2006.01) A01N 43/86 (2006.01) A01N 47/40 (2006.01) A01N 51/00 (2006.01) (2006.01) (2006.01) (21) Application number: 09305544.0 A01P 7/00 A01N 25/02 (22) Date of filing: 15.06.2009 (84) Designated Contracting States: (72) Inventors: AT BE BG CH CY CZ DE DK EE ES FI FR GB GR • Gasse, Jean-Jacques HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL 27600 Saint-Aubin-Sur-Gaillon (FR) PT RO SE SI SK TR • Duchamp, Guillaume Designated Extension States: 92230 Gennevilliers (FR) AL BA RS • Cantero, Maria 92230 Gennevilliers (FR) (71) Applicant: NUFARM 92233 Gennevelliers (FR) (74) Representative: Cabinet Plasseraud 52, rue de la Victoire 75440 Paris Cedex 09 (FR) (54) NMP-free formulations of neonicotinoids (57) The invention relates to NMP-free liquid formulation comprising at least one nicotinoid and at least one aprotic polar component selected from the group comprising the compounds of formula I, II or III below, and mixtures thereof, wherein R1 and R2 independently represent H or an alkyl group having less than 5 carbons, preferably a methyl group, and n represents an integer ranging from 0 to 5, and to their applications. EP 2 266 400 A1 Printed by Jouve, 75001 PARIS (FR) EP 2 266 400 A1 Description Technical Field of the invention 5 [0001] The invention relates to novel liquid formulations of neonicotinoids and to their use for treating plants, for protecting plants from pests and/or for controlling pests infestation. -
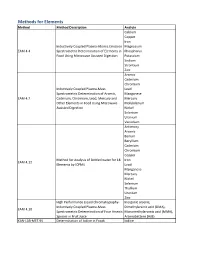
Method Description
Methods for Elements Method Method Description Analyte Calcium Copper Iron Inductively Coupled Plasma-Atomic Emission Magnesium EAM 4.4 Spectrometric Determination of Elements in Phosphorus Food Using Microwave Assisted Digestion Potassium Sodium Strontium Zinc Arsenic Cadmium Chromium Inductively Coupled Plasma-Mass Lead Spectrometric Determination of Arsenic, Manganese EAM 4.7 Cadmium, Chromium, Lead, Mercury and Mercury Other Elements in Food Using Microwave Molybdenum Assisted Digestion Nickel Selenium Uranium Vanadium Antimony Arsenic Barium Beryllium Cadmium Chromium Copper Method for Analysis of Bottled water for 18 Iron EAM 4.12 Elements by ICPMS Lead Manganese Mercury Nickel Selenium Thallium Uranium Zinc High Performance Liquid Chromatography- Inorganic arsenic, Inductively Coupled Plasma-Mass Dimethylarsinic acid (DMA), EAM 4.10 Spectrometric Determination of Four Arsenic Monomethylarsonic acid (MMA), Species in Fruit Juice Arsenobetaine (AsB) KAN-LAB-MET.95 Determination of Iodine in Foods Iodine Methods for Radionuclides Method Method Description Analyte Determination of Strontium-90 in Foods by WEAC.RN.METHOD.2.0 Strontium-90 Internal Gas-Flow Proportional Counting Americium-241 Cesium-134 Cesium-137 Determination of Gamma-Ray Emitting Cobalt-60 WEAC.RN.METHOD.3.0 Radionuclides in Foods by High-Purity Potassium-40 Germanium Spectrometry Radium-226 Ruthenium-103 Ruthenium-106 Thorium-232 Methods for Pesticides/Industrial Chemicals Method Method Description Analyte Extraction Method: Analysis of Pesticides KAN-LAB-PES.53 and