Chlamydia Trachomatis
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Chlamydia Trachomatis Infection Is Driven by Nonprotective Immune Cells That Are Distinct from Protective Populations
Pathology after Chlamydia trachomatis infection is driven by nonprotective immune cells that are distinct from protective populations Rebeccah S. Lijeka,b,1, Jennifer D. Helblea, Andrew J. Olivea,c, Kyra W. Seigerb, and Michael N. Starnbacha,1 aDepartment of Microbiology and Immunobiology, Harvard Medical School, Boston, MA 02115; bDepartment of Biological Sciences, Mount Holyoke College, South Hadley, MA 01075; and cDepartment of Microbiology and Physiological Systems, University of Massachusetts Medical School, Worcester, MA 01605 Edited by Rafi Ahmed, Emory University, Atlanta, GA, and approved December 27, 2017 (received for review June 23, 2017) Infection with Chlamydia trachomatis drives severe mucosal immu- sequence identity, Chlamydia muridarum, the extent to which the nopathology; however, the immune responses that are required for molecular pathogenesis of C. muridarum represents that of Ct is mediating pathology vs. protection are not well understood. Here, unknown (6). Ct serovar L2 (Ct L2) is capable of infecting the we employed a mouse model to identify immune responses re- mouse upper genital tract when inoculated across the cervix into quired for C. trachomatis-induced upper genital tract pathology the uterus (7, 8) but it does not induce robust immunopathology. and to determine whether these responses are also required for This is consistent with the human disease phenotype caused by Ct L2, bacterial clearance. In mice as in humans, immunopathology was which disseminates to the lymph nodes causing lymphogranuloma characterized by extravasation of leukocytes into the upper genital venereum (LGV) and is not a major cause of mucosal immunopa- thology in the female upper genital tract (uterus and ovaries). tract that occluded luminal spaces in the uterus and ovaries. -

Global Burden of Norovirus and Prospects for Vaccine Development
Global Burden of Norovirus and Prospects for Vaccine Development Primary author Ben Lopman Centers for Disease Control and Prevention Contributors and Reviewers Robert Atmar, Baylor College of Medicine Ralph Baric, University of North Carolina Mary Estes, Baylor College of Medicine Kim Green, NIH; National Institute of Allergy and Infectious Diseases Roger Glass, NIH; Fogarty International Center Aron Hall, Centers for Disease Control and Prevention Miren Iturriza-Gómara, University of Liverpool Cherry Kang, Christian Medical College Bruce Lee, Johns Hopkins University Umesh Parashar, Centers for Disease Control and Prevention Mark Riddle, Naval Medical Research Center Jan Vinjé, Centers for Disease Control and Prevention The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention, or the US Department of Health and Human Services. This work was funded in part by a grant from the Bill & Melinda Gates Foundation to the CDC Foundation. GLOBAL BURDEN OF NOROVIRUS AND PROSPECTS FOR VACCINE DEVELOPMENT | 1 Table of Contents 1. Executive summary ....................................................................3 2. Burden of disease and epidemiology 7 a. Burden 7 i. Global burden and trends of diarrheal disease in children and adults 7 ii. The role of norovirus 8 b. Epidemiology 9 i. Early childhood infections 9 ii. Risk factors, modes and settings of transmission 10 iii. Chronic health consequences associated with norovirus infection? 11 c. Challenges in attributing disease to norovirus 12 3. Norovirus biology, diagnostics and their interpretation for field studies and clinical trials..15 a. Norovirus virology 15 i. Genetic diversity, evolution and related challenges for diagnosis 15 ii. -

African Meningitis Belt
WHO/EMC/BAC/98.3 Control of epidemic meningococcal disease. WHO practical guidelines. 2nd edition World Health Organization Emerging and other Communicable Diseases, Surveillance and Control This document has been downloaded from the WHO/EMC Web site. The original cover pages and lists of participants are not included. See http://www.who.int/emc for more information. © World Health Organization This document is not a formal publication of the World Health Organization (WHO), and all rights are reserved by the Organization. The document may, however, be freely reviewed, abstracted, reproduced and translated, in part or in whole, but not for sale nor for use in conjunction with commercial purposes. The views expressed in documents by named authors are solely the responsibility of those authors. The mention of specific companies or specific manufacturers' products does no imply that they are endorsed or recommended by the World Health Organization in preference to others of a similar nature that are not mentioned. CONTENTS CONTENTS ................................................................................... i PREFACE ..................................................................................... vii INTRODUCTION ......................................................................... 1 1. MAGNITUDE OF THE PROBLEM ........................................................3 1.1 REVIEW OF EPIDEMICS SINCE THE 1970S .......................................................................................... 3 Geographical distribution -

Leptospirosis: a Waterborne Zoonotic Disease of Global Importance
August 2006 volume 22 number 08 Leptospirosis: A waterborne zoonotic disease of global importance INTRODUCTION syndrome has two phases: a septicemic and an immune phase (Levett, 2005). Leptospirosis is considered one of the most common zoonotic diseases It is in the immune phase that organ-specific damage and more severe illness globally. In the United States, outbreaks are increasingly being reported is seen. See text box for more information on the two phases. The typical among those participating in recreational water activities (Centers for Disease presenting signs of leptospirosis in humans are fever, headache, chills, con- Control and Prevention [CDC], 1996, 1998, and 2001) and sporadic cases are junctival suffusion, and myalgia (particularly in calf and lumbar areas) often underdiagnosed. With the onset of warm temperatures, increased (Heymann, 2004). Less common signs include a biphasic fever, meningitis, outdoor activities, and travel, Georgia may expect to see more leptospirosis photosensitivity, rash, and hepatic or renal failure. cases. DIAGNOSIS OF LEPTOSPIROSIS Leptospirosis is a zoonosis caused by infection with the bacterium Leptospira Detecting serum antibodies against leptospira interrogans. The disease occurs worldwide, but it is most common in temper- • Microscopic Agglutination Titers (MAT) ate regions in the late summer and early fall and in tropical regions during o Paired serum samples which show a four-fold rise in rainy seasons. It is not surprising that Hawaii has the highest incidence of titer confirm the diagnosis; a single high titer in a per- leptospirosis in the United States (Levett, 2005). The reservoir of pathogenic son clinically suspected to have leptospirosis is highly leptospires is the renal tubules of wild and domestic animals. -

Gonorrhea, Chlamydia, and Syphilis
2019 GONORRHEA, CHLAMYDIA, AND SYPHILIS AND CHLAMYDIA, GONORRHEA, Dedication TAG would like to thank the National Coalition of STD Directors for funding and input on the report. THE PIPELINE REPORT Pipeline for Gonorrhea, Chlamydia, and Syphilis By Jeremiah Johnson Introduction The current toolbox for addressing gonorrhea, chlamydia, and syphilis is inadequate. At a time where all three epidemics are dramatically expanding in locations all around the globe, including record-breaking rates of new infections in the United States, stakeholders must make do with old tools, inadequate systems for addressing sexual health, and a sparse research pipeline of new treatment, prevention, and diagnostic options. Lack of investment in sexual health research has left the field with inadequate prevention options, and limited access to infrastructure for testing and treatment have allowed sexually transmitted infections (STIs) to flourish. The consequences of this underinvestment are large: according to the World Health Organization (WHO), in 2012 there were an estimated 357 million new infections (roughly 1 million per day) of the four curable STIs: gonorrhea, chlamydia, syphilis, and trichomoniasis.1 In the United States, the three reportable STIs that are the focus of this report—gonorrhea, chlamydia, and syphilis—are growing at record paces. In 2017, a total of 30,644 cases of primary and secondary (P&S) syphilis—the most infectious stages of the disease—were reported in the United States. Since reaching a historic low in 2000 and 2001, the rate of P&S syphilis has increased almost every year, increasing 10.5% during 2016–2017. Also in 2017, 555,608 cases of gonorrhea were reported to the U.S. -
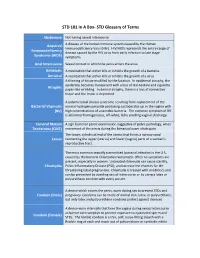
STD Glossary of Terms
STD 101 In A Box- STD Glossary of Terms Abstinence Not having sexual intercourse Acquired A disease of the human immune system caused by the Human Immunodeficiency Virus (HIV). HIV/AIDS represents the entire range of Immunodeficiency disease caused by the HIV virus from early infection to late stage Syndrome (AIDS) symptoms. Anal Intercourse Sexual contact in which the penis enters the anus. Antibiotic A medication that either kills or inhibits the growth of a bacteria. Antiviral A medication that either kills or inhibits the growth of a virus. A thinning of tissue modified by the location. In epidermal atrophy, the epidermis becomes transparent with a loss of skin texture and cigarette Atrophic paper-like wrinkling. In dermal atrophy, there is a loss of connective tissue and the lesion is depressed. A polymicrobial clinical syndrome resulting from replacement of the Bacterial Vaginosis normal hydrogen peroxide producing Lactobacillus sp. in the vagina with (BV) high concentrations of anaerobic bacteria. The common symptom of BV is abnormal homogeneous, off-white, fishy smelling vaginal discharge. Cervical Motion A sign found on pelvic examination suggestive of pelvic pathology; when Tenderness (CMT) movement of the cervix during the bimanual exam elicits pain. The lower, cylindrical end of the uterus that forms a narrow canal Cervix connecting the upper (uterus) and lower (vagina) parts of a woman's reproductive tract. The most common sexually transmitted bacterial infection in the U.S., caused by the bacteria Chlamydia trachomatis. Often no symptoms are present, especially in women. Untreated chlamydia can cause sterility, Chlamydia Pelvic Inflammatory Disease (PID), and increase the chances for life- threatening tubal pregnancies. -

Chlamydia Trachomatis and Chlamydia Pneumoniae Interaction with the Host: Latest Advances and Future Prospective
microorganisms Review Chlamydia trachomatis and Chlamydia pneumoniae Interaction with the Host: Latest Advances and Future Prospective Marisa Di Pietro 1,* , Simone Filardo 1 , Silvio Romano 2 and Rosa Sessa 1 1 Department of Public Health and Infectious Diseases, Section of Microbiology, University of Rome “Sapienza”, 00185 Rome, Italy; simone.fi[email protected] (S.F.); [email protected] (R.S.) 2 Cardiology, Department of Life, Health and Environmental Sciences, University of L’Aquila, 67100 L’Aquila, Italy; [email protected] * Correspondence: [email protected] Received: 15 April 2019; Accepted: 14 May 2019; Published: 16 May 2019 Abstract: Research in Chlamydia trachomatis and Chlamydia pneumoniae has gained new traction due to recent advances in molecular biology, namely the widespread use of the metagenomic analysis and the development of a stable genomic transformation system, resulting in a better understanding of Chlamydia pathogenesis. C. trachomatis, the leading cause of bacterial sexually transmitted diseases, is responsible of cervicitis and urethritis, and C. pneumoniae, a widespread respiratory pathogen, has long been associated with several chronic inflammatory diseases with great impact on public health. The present review summarizes the current evidence regarding the complex interplay between C. trachomatis and host defense factors in the genital micro-environment as well as the key findings in chronic inflammatory diseases associated to C. pneumoniae. Keywords: Chlamydia trachomatis; Chlamydia pneumoniae; host-pathogen interaction 1. Introduction Currently, there is a renewed research interest in Chlamydiae that cause a broad spectrum of pathologies of varying severity in human, mainly Chlamydia trachomatis and Chlamydia pneumoniae [1,2]. Advances in molecular biology and, in particular, the recent advent of metagenomic analysis as well as the development of a stable genomic transformation system in Chlamydiae have significantly contributed to expanding our understanding of Chlamydia pathogenesis [3–5]. -

Novel Risk Factors for Coronavirus Disease-Associated Mucormycosis (CAM): a Case Control Study During the Outbreak in India
medRxiv preprint doi: https://doi.org/10.1101/2021.07.24.21261040; this version posted July 26, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted medRxiv a license to display the preprint in perpetuity. It is made available under a CC-BY-NC-ND 4.0 International license . Novel risk factors for Coronavirus disease-associated mucormycosis (CAM): a case control study during the outbreak in India Umang Arora1, Megha Priyadarshi1, Varidh Katiyar2, Manish Soneja1, Prerna Garg1, Ishan Gupta1,- Vishwesh Bharadiya1, Parul Berry1, Tamoghna Ghosh1, Lajjaben Patel1 , Radhika Sarda1, Shreya Garg3, Shubham Agarwal1, Veronica Arora4, Aishwarya Ramprasad1, Amit Kumar1, Rohit Kumar Garg1, Parul Kodan1, Neeraj Nischal1, Gagandeep Singh5, Pankaj Jorwal1, Arvind Kumar1, Upendra Baitha1, Ved Prakash Meena1, Animesh Ray1, Prayas Sethi1, , Immaculata Xess5, Naval Vikram1, Sanjeev Sinha1, Ashutosh Biswas1,Alok Thakar3, Sushma Bhatnagar6, Anjan Trikha7, Naveet Wig1 1Department of Medicine, AIIMS, Delhi, India 2Department of Neurosurgery, AIIMS, Delhi, India 3Department of Otolaryngology & Head-Neck Surgery, AIIMS, Delhi, India 4Department of Medical Genetics, Sir Ganga Ram Hospital, Delhi, India 5Department of Microbiology, AIIMS, Delhi, India 6Department of Onco-anaesthesia and Palliative Medicine, AIIMS, Delhi, India 7Department of Anaesthesiology, Pain Medicine and Critical Care, AIIMS, Delhi, India 1. Umang Arora, MD (Medicine), Junior Resident, Department of Medicine, AIIMS, Delhi, India. E-mail ID: [email protected] 2. Megha Priyadarshi, DM (Infectious disease), Junior Resident, Department of Medicine, AIIMS, Delhi, India. E-mail ID: [email protected] 3. Varidh Katiyar, MCh (Neurosurgery), Chief Resident, Department of Neurosurgery AIIMS, Delhi, India. E-mail ID: [email protected] 4. -

Chlamydial Genital Infection(Chlamydia Trachomatis)
Chlamydial Genital Infection (Chlamydia trachomatis) February 2003 1) THE DISEASE AND ITS EPIDEMIOLOGY A. Etiologic Agent Chlamydial genital infection (CGI) is caused by the obligate, intracellular bacterium Chlamydia trachomatis immunotypes D through K. B. Clinical Description and Laboratory Diagnosis A sexually transmitted genital infection that manifests in males primarily as urethritis and in females as mucopurulent cervicitis. Clinical manifestations are difficult to distinguish from gonorrhea. Males may present with a mucopurulent discharges of scanty to moderate quantity, urethral itching and dysuria. Asymptomatic infection may be found in 1%-25% of sexually active men. Possible complications include epididymitis, infertility and Reiter syndrome. Anorectal intercourse may result in chlamydial proctitis. Women frequently present with a mucopurulent endocervical discharge including edema, erythema and easily induced endocervical bleeding. However, most women with endocervical or urethral infections are asymptomatic. Possible complications include salpingitis with subsequent risk of infertility and ectopic pregnancy. Asymptomatic chronic infections of the endometrium and fallopian tubes may lead to the same outcomes. Less frequent manifestations include bartholinitis, urethral syndrome with dysuria and pyuria, perihepatitis (Fitz-Hugh-Curtis syndrome), and proctitis. Infection during pregnancy may result in premature rupture of membranes and preterm delivery and conjunctival and pneumonic infection of the newborn. Laboratory diagnosis is based upon the identification of Chlamydia in intraurethral or endocervical smear by direct immunofluorescence test, enzyme immunoassay, DNA probe, and nucleic acid amplification test (NAAT) or cell culture. NAAT can be used with urine specimens. C. Vectors and Reservoirs Humans. D. Modes of Transmission By sexual contact and through perinatal exposure to the mother’s infected cervix. -

Chlamydia, Gonorrhoea, Trichomoniasis and Syphilis
Research Chlamydia, gonorrhoea, trichomoniasis and syphilis: global prevalence and incidence estimates, 2016 Jane Rowley,a Stephen Vander Hoorn,b Eline Korenromp,c Nicola Low,d Magnus Unemo,e Laith J Abu- Raddad,f R Matthew Chico,g Alex Smolak,f Lori Newman,h Sami Gottlieb,a Soe Soe Thwin,a Nathalie Brouteta & Melanie M Taylora Objective To generate estimates of the global prevalence and incidence of urogenital infection with chlamydia, gonorrhoea, trichomoniasis and syphilis in women and men, aged 15–49 years, in 2016. Methods For chlamydia, gonorrhoea and trichomoniasis, we systematically searched for studies conducted between 2009 and 2016 reporting prevalence. We also consulted regional experts. To generate estimates, we used Bayesian meta-analysis. For syphilis, we aggregated the national estimates generated by using Spectrum-STI. Findings For chlamydia, gonorrhoea and/or trichomoniasis, 130 studies were eligible. For syphilis, the Spectrum-STI database contained 978 data points for the same period. The 2016 global prevalence estimates in women were: chlamydia 3.8% (95% uncertainty interval, UI: 3.3–4.5); gonorrhoea 0.9% (95% UI: 0.7–1.1); trichomoniasis 5.3% (95% UI:4.0–7.2); and syphilis 0.5% (95% UI: 0.4–0.6). In men prevalence estimates were: chlamydia 2.7% (95% UI: 1.9–3.7); gonorrhoea 0.7% (95% UI: 0.5–1.1); trichomoniasis 0.6% (95% UI: 0.4–0.9); and syphilis 0.5% (95% UI: 0.4–0.6). Total estimated incident cases were 376.4 million: 127.2 million (95% UI: 95.1–165.9 million) chlamydia cases; 86.9 million (95% UI: 58.6–123.4 million) gonorrhoea cases; 156.0 million (95% UI: 103.4–231.2 million) trichomoniasis cases; and 6.3 million (95% UI: 5.5–7.1 million) syphilis cases. -

Evaluation and Determination of the Sensitivity and Specificity of a Treponema Pallidum Dried Blood Spot Method for Serologic Diagnosis of Syphilis
Georgia State University ScholarWorks @ Georgia State University Public Health Theses School of Public Health Fall 12-20-2012 Evaluation and Determination of the Sensitivity and Specificity of a Treponema Pallidum Dried Blood Spot Method for Serologic Diagnosis of Syphilis David K. Turgeon Follow this and additional works at: https://scholarworks.gsu.edu/iph_theses Recommended Citation Turgeon, David K., "Evaluation and Determination of the Sensitivity and Specificity of a rT eponema Pallidum Dried Blood Spot Method for Serologic Diagnosis of Syphilis." Thesis, Georgia State University, 2012. https://scholarworks.gsu.edu/iph_theses/239 This Thesis is brought to you for free and open access by the School of Public Health at ScholarWorks @ Georgia State University. It has been accepted for inclusion in Public Health Theses by an authorized administrator of ScholarWorks @ Georgia State University. For more information, please contact [email protected]. Institute of Public Health Public Health Thesis Georgia State University Year 2012 EVALUATION AND DETERMINATION OF THE SENSITIVITY AND SPECIFICITY OF A Treponema pallidum DRIED BLOOD SPOT METHOD FOR SEROLOGIC DIAGNOSIS OF SYPHILIS David K. Turgeon Georgia State University, [email protected] ii ABSTRACT EVALUATION AND DETERMINATION OF THE SENSITIVITY AND SPECIFICITY OF A Treponema pallidum DRIED BLOOD SPOT (DBS) METHOD FOR SEROLOGIC DIAGNOSIS OF SYPHILIS Background: Syphilis is a sexually transmitted infection (STI) caused by Treponema pallidum subspecies pallidum. Syphilis is known as the “great imitator" due to the similarity of clinical signs and symptoms to other infectious diseases. The primary diagnosis of syphilis relies on clinical findings, including the examination of treponemal lesions, and/or serologic tests. -

Detection of Kaposi's Sarcoma–Associated Herpesvirus in Oral
1785 CONCISE COMMUNICATION Detection of Kaposi's Sarcoma±Associated Herpesvirus in Oral and Genital Secretions of Zimbabwean Women Thomas M. Lampinen,1,3 Shalini Kulasingam,1 1Departments of Epidemiology and 2Pathobiology and 3Center for AIDS Juno Min,4 Margaret Borok,6 Lovemore Gwanzura,7 and STD, University of Washington, Seattle; 4Department of Medicine 5 Downloaded from https://academic.oup.com/jid/article/181/5/1785/2191825 by guest on 29 September 2021 4 8 9 and Center for AIDS Research, Stanford University, Stanford, Julie Lamb, Kassam Mahomed, Godfrey B. Woelk, 6 7 2 2 California; Departments of Medicine, Medical Laboratory Sciences, Kurt B. Strand, Marnix L. Bosch, 8Obstetrics and Gynecology, and 9Community Medicine, 10 10,11 Daniel C. Edelman, Niel T. Constantine, University of Zimbabwe, Harare; 10Department of Pathology, David Katzenstein,4,5 and Michelle A. Williams1 University of Maryland and 11Institute of Human Virology, Baltimore, Maryland Kaposi's sarcoma±associated herpesvirus (KSHV) in oral and genital secretions of women may be involved in horizontal and vertical transmission in endemic regions. Nested polymerase chain reaction assays were used to detect KSHV DNA sequences in one-third of oral, vaginal, and cervical specimens and in 42% of peripheral blood mononuclear cell (PBMC) specimens collected from 41 women infected with human immunode®ciency virus type 1 who had Ka- posi's sarcoma (KS). KSHV DNA was not detected in specimens from 100 women without KS, 9 of whom were seropositive for KSHV. A positive association was observed between KSHV DNA detection in oral and genital mucosa, neither of which was associated with KSHV DNA detection in PBMC.