New Advances in Cyclic AMP Signalling”—An Editorial Overview
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Molecular Targets and Biological Functions of Camp Signaling in Arabidopsis
biomolecules Article Molecular Targets and Biological Functions of cAMP Signaling in Arabidopsis Ruqiang Xu 1,2,*, Yanhui Guo 1, Song Peng 1, Jinrui Liu 1, Panyu Li 1, Wenjing Jia 1 and Junheng Zhao 1 1 School of Agricultural Sciences, Zhengzhou University, Zhengzhou 450001, China; [email protected] (Y.G.); [email protected] (S.P.); [email protected] (J.L.); [email protected] (P.L.); [email protected] (W.J.); [email protected] (J.Z.) 2 Zhengzhou Research Base, State Key Laboratory of Cotton Biology, Zhengzhou University, Zhengzhou 450001, China * Correspondence: [email protected]; Tel.: +86-0371-6778-5095 Abstract: Cyclic AMP (cAMP) is a pivotal signaling molecule existing in almost all living organisms. However, the mechanism of cAMP signaling in plants remains very poorly understood. Here, we employ the engineered activity of soluble adenylate cyclase to induce cellular cAMP elevation in Arabidopsis thaliana plants and identify 427 cAMP-responsive genes (CRGs) through RNA-seq analysis. Induction of cellular cAMP elevation inhibits seed germination, disturbs phytohormone contents, promotes leaf senescence, impairs ethylene response, and compromises salt stress tolerance and pathogen resistance. A set of 62 transcription factors are among the CRGs, supporting a prominent role of cAMP in transcriptional regulation. The CRGs are significantly overrepresented in the pathways of plant hormone signal transduction, MAPK signaling, and diterpenoid biosynthesis, but + they are also implicated in lipid, sugar, K , nitrate signaling, and beyond. Our results provide a basic framework of cAMP signaling for the community to explore. The regulatory roles of cAMP signaling Citation: Xu, R.; Guo, Y.; Peng, S.; in plant plasticity are discussed. -

Identi®Cation and Role of Adenylyl Cyclase in Auxin Signalling in Higher
letters to nature + + + P.P. thank the Academy of Finland and the Deutsche Forschungsgemeinschaft, respectively, for ®nancial CO , 53), 77 (C6H5 , 60), 73 (TMSi , 84); 6-methyl-4-hydroxy-2-pyrone: RRt 0.35, 198 (M+, 18), 183 ([M-Me]+, 16), 170 ([M-CO]+, 54), 155 ([M-CO-Me]+, support. + + Correspondence and requests for materials should be addressed to J.S. (e-mail: [email protected] 15), 139 ([M-Me-CO2] , 10), 127 ([M-Me-2CO] , 13), 99 (12), 84 (13), 73 + + freiburg.de). (TMSi , 100), 43 (CH3CO , 55). The numbers show m/z values, and the key fragments and their relative intensities are indicated in parentheses. Received 4 August; accepted 14 October 1998. erratum 1. Helariutta, Y. et al. Chalcone synthase-like genes active during corolla development are differentially expressed and encode enzymes with different catalytic properties in Gerbera hybrida (Asteraceae). Plant Mol. Biol. 28, 47±60 (1995). 2. Helariutta, Y. et al. Duplication and functional divergence in the chalcone synthase gene family of 8 Asteraceae: evolution with substrate change and catalytic simpli®cation. Proc. Natl Acad. Sci. USA 93, Crystal structure of the complex 9033±9038 (1996). 3. Thaisrivongs, S. et al. Structure-based design of HIV protease inhibitors: 5,6-dihydro-4-hydroxy-2- of the cyclin D-dependent pyrones as effective, nonpeptidic inhibitors. J. Med. Chem. 39, 4630±4642 (1996). 4. Hagen, S. E. et al. Synthesis of 5,6-dihydro-4-hydroxy-2-pyrones as HIV-1 protease inhibitors: the kinase Cdk6 bound to the profound effect of polarity on antiviral activity. J. Med. Chem. -

Mechanisms Whereby Extracellular Adenosine 3',5'- Monophosphate Inhibits Phosphate Transport in Cultured Opossum Kidney Cells and in Rat Kidney
Mechanisms whereby extracellular adenosine 3',5'- monophosphate inhibits phosphate transport in cultured opossum kidney cells and in rat kidney. Physiological implication. G Friedlander, … , C Coureau, C Amiel J Clin Invest. 1992;90(3):848-858. https://doi.org/10.1172/JCI115960. Research Article The mechanism of phosphaturia induced by cAMP infusion and the physiological role of extracellular cAMP in modulation of renal phosphate transport were examined. In cultured opossum kidney cells, extracellular cAMP (10-1,000 microM) inhibited Na-dependent phosphate uptake in a time- and concentration-dependent manner. The effect of cAMP was reproduced by ATP, AMP, and adenosine, and was blunted by phosphodiesterase inhibitors or by dipyridamole which inhibits adenosine uptake. [3H]cAMP was degraded extracellularly into AMP and adenosine, and radioactivity accumulated in the cells as labeled adenosine and, subsequently, as adenine nucleotides including cAMP. Radioactivity accumulation was decreased by dipyridamole and by inhibitors of phosphodiesterases and ecto-5'-nucleotidase, assessing the existence of stepwise hydrolysis of extracellular cAMP and intracellular processing of taken up adenosine. In vivo, dipyridamole abolished the phosphaturia induced by exogenous cAMP infusion in acutely parathyroidectomized (APTX) rats, decreased phosphate excretion in intact rats, and blunted phosphaturia induced by PTH infusion in APTX rats. These results indicate that luminal degradation of cAMP into adenosine, followed by cellular uptake of the nucleoside by tubular cells, is a key event which accounts for the phosphaturic effect of exogenous cAMP and for the part of the phosphaturic effect of PTH which is mediated by cAMP added to the tubular lumen under the influence of the hormone. -

G Protein-Coupled Receptors
G PROTEIN-COUPLED RECEPTORS Overview:- The completion of the Human Genome Project allowed the identification of a large family of proteins with a common motif of seven groups of 20-24 hydrophobic amino acids arranged as α-helices. Approximately 800 of these seven transmembrane (7TM) receptors have been identified of which over 300 are non-olfactory receptors (see Frederikson et al., 2003; Lagerstrom and Schioth, 2008). Subdivision on the basis of sequence homology allows the definition of rhodopsin, secretin, adhesion, glutamate and Frizzled receptor families. NC-IUPHAR recognizes Classes A, B, and C, which equate to the rhodopsin, secretin, and glutamate receptor families. The nomenclature of 7TM receptors is commonly used interchangeably with G protein-coupled receptors (GPCR), although the former nomenclature recognises signalling of 7TM receptors through pathways not involving G proteins. For example, adiponectin and membrane progestin receptors have some sequence homology to 7TM receptors but signal independently of G-proteins and appear to reside in membranes in an inverted fashion compared to conventional GPCR. Additionally, the NPR-C natriuretic peptide receptor has a single transmembrane domain structure, but appears to couple to G proteins to generate cellular responses. The 300+ non-olfactory GPCR are the targets for the majority of drugs in clinical usage (Overington et al., 2006), although only a minority of these receptors are exploited therapeutically. Signalling through GPCR is enacted by the activation of heterotrimeric GTP-binding proteins (G proteins), made up of α, β and γ subunits, where the α and βγ subunits are responsible for signalling. The α subunit (tabulated below) allows definition of one series of signalling cascades and allows grouping of GPCRs to suggest common cellular, tissue and behavioural responses. -

Challenges on Cyclic Nucleotide Phosphodiesterases Imaging with Positron Emission Tomography: Novel Radioligands and (Pre-)Clinical Insights Since 2016
International Journal of Molecular Sciences Review Challenges on Cyclic Nucleotide Phosphodiesterases Imaging with Positron Emission Tomography: Novel Radioligands and (Pre-)Clinical Insights since 2016 Susann Schröder 1,2,* , Matthias Scheunemann 2, Barbara Wenzel 2 and Peter Brust 2 1 Department of Research and Development, ROTOP Pharmaka Ltd., 01328 Dresden, Germany 2 Department of Neuroradiopharmaceuticals, Institute of Radiopharmaceutical Cancer Research, Research Site Leipzig, Helmholtz-Zentrum Dresden-Rossendorf (HZDR), 04318 Leipzig, Germany; [email protected] (M.S.); [email protected] (B.W.); [email protected] (P.B.) * Correspondence: [email protected]; Tel.: +49-341-234-179-4631 Abstract: Cyclic nucleotide phosphodiesterases (PDEs) represent one of the key targets in the research field of intracellular signaling related to the second messenger molecules cyclic adenosine monophosphate (cAMP) and/or cyclic guanosine monophosphate (cGMP). Hence, non-invasive imaging of this enzyme class by positron emission tomography (PET) using appropriate isoform- selective PDE radioligands is gaining importance. This methodology enables the in vivo diagnosis and staging of numerous diseases associated with altered PDE density or activity in the periphery and the central nervous system as well as the translational evaluation of novel PDE inhibitors as therapeutics. In this follow-up review, we summarize the efforts in the development of novel PDE radioligands and highlight (pre-)clinical insights from PET studies using already known PDE Citation: Schröder, S.; Scheunemann, radioligands since 2016. M.; Wenzel, B.; Brust, P. Challenges on Cyclic Nucleotide Keywords: positron emission tomography; cyclic nucleotide phosphodiesterases; PDE inhibitors; Phosphodiesterases Imaging with PDE radioligands; radiochemistry; imaging; recent (pre-)clinical insights Positron Emission Tomography: Novel Radioligands and (Pre-)Clinical Insights since 2016. -

Geochemical Influences on Nonenzymatic Oligomerization Of
bioRxiv preprint doi: https://doi.org/10.1101/872234; this version posted December 11, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Geochemical influences on nonenzymatic oligomerization of prebiotically relevant cyclic nucleotides Authors: Shikha Dagar‡, Susovan Sarkar‡, Sudha Rajamani‡* ‡ Department of Biology, Indian Institute of Science Education and Research, Pune 411008, India Correspondence: [email protected]; Tel.: +91-20-2590-8061 Running title: Cyclic nucleotides and emergence of an RNA World Key words: Dehydration-rehydration cycles, lipid-assisted oligomerization, cyclic nucleotides, analogue environments Dagar, S. 1 bioRxiv preprint doi: https://doi.org/10.1101/872234; this version posted December 11, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Abstract The spontaneous emergence of RNA on the early Earth continues to remain an enigma in the field of origins of life. Few studies have looked at the nonenzymatic oligomerization of cyclic nucleotides under neutral to alkaline conditions, in fully dehydrated state. Herein, we systematically investigated the oligomerization of cyclic nucleotides under prebiotically relevant conditions, where starting reactants were subjected to repeated dehydration-rehydration (DH- RH) regimes, like they would have been on an early Earth. DH-RH conditions, a recurring geological theme, are driven by naturally occurring processes including diurnal cycles and tidal pool activity. These conditions have been shown to facilitate uphill oligomerization reactions in terrestrial geothermal niches, which are hypothesized to be pertinent sites for the emergence of life. -

Oxidative Stress and the Guanosine Nucleotide Triphosphate Pool: Implications for a Biomarker and Mechanism of Impaired Cell Function
University of Montana ScholarWorks at University of Montana Graduate Student Theses, Dissertations, & Professional Papers Graduate School 2008 OXIDATIVE STRESS AND THE GUANOSINE NUCLEOTIDE TRIPHOSPHATE POOL: IMPLICATIONS FOR A BIOMARKER AND MECHANISM OF IMPAIRED CELL FUNCTION Celeste Maree Bolin The University of Montana Follow this and additional works at: https://scholarworks.umt.edu/etd Let us know how access to this document benefits ou.y Recommended Citation Bolin, Celeste Maree, "OXIDATIVE STRESS AND THE GUANOSINE NUCLEOTIDE TRIPHOSPHATE POOL: IMPLICATIONS FOR A BIOMARKER AND MECHANISM OF IMPAIRED CELL FUNCTION" (2008). Graduate Student Theses, Dissertations, & Professional Papers. 728. https://scholarworks.umt.edu/etd/728 This Dissertation is brought to you for free and open access by the Graduate School at ScholarWorks at University of Montana. It has been accepted for inclusion in Graduate Student Theses, Dissertations, & Professional Papers by an authorized administrator of ScholarWorks at University of Montana. For more information, please contact [email protected]. OXIDATIVE STRESS AND THE GUANOSINE NUCLEOTIDE TRIPHOSPHATE POOL: IMPLICATIONS FOR A BIOMARKER AND MECHANISM OF IMPAIRED CELL FUNCTION By Celeste Maree Bolin B.A. Chemistry, Whitman College, Walla Walla, WA 2001 Dissertation presented in partial fulfillment of the requirements for the degree of Doctor of Philosophy in Toxicology The University of Montana Missoula, Montana Spring 2008 Approved by: Dr. David A. Strobel, Dean Graduate School Dr. Fernando Cardozo-Pelaez, -

AMP-Activated Protein Kinase: the Current Landscape for Drug Development
REVIEWS AMP-activated protein kinase: the current landscape for drug development Gregory R. Steinberg 1* and David Carling2 Abstract | Since the discovery of AMP-activated protein kinase (AMPK) as a central regulator of energy homeostasis, many exciting insights into its structure, regulation and physiological roles have been revealed. While exercise, caloric restriction, metformin and many natural products increase AMPK activity and exert a multitude of health benefits, developing direct activators of AMPK to elicit beneficial effects has been challenging. However, in recent years, direct AMPK activators have been identified and tested in preclinical models, and a small number have entered clinical trials. Despite these advances, which disease(s) represent the best indications for therapeutic AMPK activation and the long-term safety of such approaches remain to be established. Cardiovascular disease Dramatic improvements in health care coupled with identifying a unifying mechanism that could link these (CVD). A term encompassing an increased standard of living, including better nutri- changes to multiple branches of metabolism followed diseases affecting the heart tion and education, have led to a remarkable increase in the discovery that the AMP-activated protein kinase or circulatory system. human lifespan1. Importantly, the number of years spent (AMPK) provided a common regulatory mechanism in good health is also increasing2. Despite these positive for inhibiting both cholesterol (through phosphoryla- Non-alcoholic fatty liver disease developments, there are substantial risks that challenge tion of HMG-CoA reductase (HMGR)) and fatty acid (NAFLD). A very common continued improvements in human health. Perhaps the (through phosphorylation of acetyl-CoA carboxylase disease in humans in which greatest threat to future health is a chronic energy imbal- (ACC)) synthesis8 (BOx 1). -
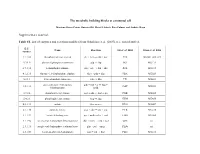
The Metabolic Building Blocks of a Minimal Cell Supplementary
The metabolic building blocks of a minimal cell Mariana Reyes-Prieto, Rosario Gil, Mercè Llabrés, Pere Palmer and Andrés Moya Supplementary material. Table S1. List of enzymes and reactions modified from Gabaldon et. al. (2007). n.i.: non identified. E.C. Name Reaction Gil et. al. 2004 Glass et. al. 2006 number 2.7.1.69 phosphotransferase system glc + pep → g6p + pyr PTS MG041, 069, 429 5.3.1.9 glucose-6-phosphate isomerase g6p ↔ f6p PGI MG111 2.7.1.11 6-phosphofructokinase f6p + atp → fbp + adp PFK MG215 4.1.2.13 fructose-1,6-bisphosphate aldolase fbp ↔ gdp + dhp FBA MG023 5.3.1.1 triose-phosphate isomerase gdp ↔ dhp TPI MG431 glyceraldehyde-3-phosphate gdp + nad + p ↔ bpg + 1.2.1.12 GAP MG301 dehydrogenase nadh 2.7.2.3 phosphoglycerate kinase bpg + adp ↔ 3pg + atp PGK MG300 5.4.2.1 phosphoglycerate mutase 3pg ↔ 2pg GPM MG430 4.2.1.11 enolase 2pg ↔ pep ENO MG407 2.7.1.40 pyruvate kinase pep + adp → pyr + atp PYK MG216 1.1.1.27 lactate dehydrogenase pyr + nadh ↔ lac + nad LDH MG460 1.1.1.94 sn-glycerol-3-phosphate dehydrogenase dhp + nadh → g3p + nad GPS n.i. 2.3.1.15 sn-glycerol-3-phosphate acyltransferase g3p + pal → mag PLSb n.i. 2.3.1.51 1-acyl-sn-glycerol-3-phosphate mag + pal → dag PLSc MG212 acyltransferase 2.7.7.41 phosphatidate cytidyltransferase dag + ctp → cdp-dag + pp CDS MG437 cdp-dag + ser → pser + 2.7.8.8 phosphatidylserine synthase PSS n.i. cmp 4.1.1.65 phosphatidylserine decarboxylase pser → peta PSD n.i. -

Targeting the Sac-Dependent Camp Pool to Prevent SARS-Cov-2 Infection
cells Review Targeting the sAC-Dependent cAMP Pool to Prevent SARS-Cov-2 Infection Muhammad Aslam 1,2 and Yury Ladilov 3,* 1 Experimental Cardiology, Department of Internal Medicine, Justus Liebig University, 35392 Giessen, Germany; [email protected] 2 DZHK (German Centre for Cardiovascular Research), Department of Cardiology, Kerckhoff Clinic GmbH partner site Rhein-Main, 61231 Bad Nauheim, Germany 3 Independent Researcher, 42929 Wermelskirchen, Germany * Correspondence: [email protected] Received: 27 July 2020; Accepted: 24 August 2020; Published: 25 August 2020 Abstract: An outbreak of the novel coronavirus (CoV) SARS-CoV-2, the causative agent of COVID-19 respiratory disease, infected millions of people since the end of 2019, led to high-level morbidity and mortality and caused worldwide social and economic disruption. There are currently no antiviral drugs available with proven efficacy or vaccines for its prevention. An understanding of the underlying cellular mechanisms involved in virus replication is essential for repurposing the existing drugs and/or the discovery of new ones. Endocytosis is the important mechanism of entry of CoVs into host cells. Endosomal maturation followed by the fusion with lysosomes are crucial events in endocytosis. Late endosomes and lysosomes are characterized by their acidic pH, which is generated by a proton transporter V-ATPase and required for virus entry via endocytic pathway. The cytoplasmic cAMP pool produced by soluble adenylyl cyclase (sAC) promotes V-ATPase recruitment to endosomes/lysosomes and thus their acidification. In this review, we discuss targeting the sAC-specific cAMP pool as a potential strategy to impair the endocytic entry of the SARS-CoV-2 into the host cell. -

Feedback Regulation of G Protein-Coupled Receptor Signaling
G Model YSCDB-1903; No. of Pages 10 ARTICLE IN PRESS Seminars in Cell & Developmental Biology xxx (2016) xxx–xxx Contents lists available at ScienceDirect Seminars in Cell & Developmental Biology j ournal homepage: www.elsevier.com/locate/semcdb Review Feedback regulation of G protein-coupled receptor signaling by GRKs and arrestins a b a,∗ Joseph B. Black , Richard T. Premont , Yehia Daaka a Department of Anatomy and Cell Biology, University of Florida College of Medicine, Gainesville, FL 32610, United States b Department of Medicine, Duke University Medical Center, Durham, NC 27710, United States a r a t b i c s t l e i n f o r a c t Article history: GPCRs are ubiquitous in mammalian cells and present intricate mechanisms for cellular signaling Received 23 October 2015 and communication. Mechanistically, GPCR signaling was identified to occur vectorially through het- Accepted 19 December 2015 erotrimeric G proteins that are negatively regulated by GRK and arrestin effectors. Emerging evidence Available online xxx highlights additional roles for GRK and Arrestin partners, and establishes the existence of interconnected feedback pathways that collectively define GPCR signaling. GPCRs influence cellular dynamics and can Keywords: mediate pathologic development, such as cancer and cardiovascular remolding. Hence, a better under- GPCR standing of their overall signal regulation is of great translational interest and research continues to G protein Arrestin exploit the pharmacologic potential for modulating their activity. GRK © 2016 Elsevier Ltd. All rights reserved. Signal transduction Biased signaling Contents 1. Introduction . 00 2. 7TMR/GPCR signaling overview . 00 2.1. Diversity . 00 2.2. Structure and classification . -

Cyclic Nucleotide-Independent Forms of a Protein Kinase from Beef Heart
Proc. Nat. Acad. Sci. USA Vol. 68, No. 4, pp. 731-735, April 1971 Interconversion of Cyclic Nucleotide-Activated and Cyclic Nucleotide-Independent Forms of a Protein Kinase from Beef Heart JACK ERLICHMAN, ALLEN H. HIRSCH, AND ORA M. ROSEN* Departments of Medicine and Molecular Biology, Division of Biological Sciences, Albert Einstein College of Medicine, Bronx, New York 10461 Communicated by B. L. Horecker, January 14, 1971 ABSTRACT A protein kinase activated by cyclic nucle- presence. This observation suggests that cyclic AMP activates otides was purified from beef heart. Upon exposure to the protein kinase by relieving an inhibition brought about adenosine 3': 5'-cyclic monophosphate (cyclic AMP) during kinase with a AMP- gel filtration on Sephadex G-200, the protein kinase dis- by the association of the protein cyclic sociated into a cyclic nucleotide-independent protein binding protein. A similar mechanism for the regulation of kinase and a cyclic nucleotide-binding protein. A similar protein kinases from skeletal muscle and parotid gland has or identical cyclic nucleotide-independent protein kinase also been suggested (Z. Sellinger and M. Schramm, personal could be obtained in highly purified form by elution from communication). Tao et al. (15) reported that a cyclic AMP- a DEAE-cellulose column with 10-6 M cyclic AMP; the cyclic AMP-binding protein was apparently retained by dependent protein kinase from rabbit reticulocytes was dis- the resin. The addition of cyclic nucleotide-binding pro- sociated into two subunits in the presence of cyclic AMP. tein to cyclic nucleotide-independent protein kinas e One subunit bound cyclic AMP and lacked catalytic activity, resulted in the reappearance of cyclic nucleotide-depen- whereas the other subunit possessed kinase activity and dent protein kinase, which could be isolated by filtration on nucleotides.