Alpha Thalassemia
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Non-Commercial Use Only
only use Non-commercial 14th International Conference on Thalassaemia and Other Haemoglobinopathies 16th TIF Conference for Patients and Parents 17-19 November 2017 • Grand Hotel Palace, Thessaloniki, Greece only use For thalassemia patients with chronic transfusional iron overload... Make a lasting impression with EXJADENon-commercial film-coated tablets The efficacy of deferasirox in a convenient once-daily film-coated tablet Please see your local Novartis representative for Full Product Information Reference: EXJADE® film-coated tablets [EU Summary of Product Characteristics]. Novartis; August 2017. Important note: Before prescribing, consult full prescribing information. iron after having achieved a satisfactory body iron level and therefore retreatment cannot be recommended. ♦ Maximum daily dose is 14 mg/kg body weight. ♦ In pediatric patients the Presentation: Dispersible tablets containing 125 mg, 250 mg or 500 mg of deferasirox. dosing should not exceed 7 mg/kg; closer monitoring of LIC and serum ferritin is essential Film-coated tablets containing 90 mg, 180 mg or 360 mg of deferasirox. to avoid overchelation; in addition to monthly serum ferritin assessments, LIC should be Indications: For the treatment of chronic iron overload due to frequent blood transfusions monitored every 3 months when serum ferritin is ≤800 micrograms/l. (≥7 ml/kg/month of packed red blood cells) in patients with beta-thalassemia major aged Dosage: Special population ♦ In moderate hepatic impairment (Child-Pugh B) dose should 6 years and older. ♦ Also indicated for the treatment of chronic iron overload due to blood not exceed 50% of the normal dose. Should not be used in severe hepatic impairment transfusions when deferoxamine therapy is contraindicated or inadequate in the following (Child-Pugh C). -

Kawasaki Disease with Glucose-6-Phosphate Dehydrogenase Deficiency, Case Report
Saudi Pharmaceutical Journal (2014) xxx, xxx–xxx King Saud University Saudi Pharmaceutical Journal www.ksu.edu.sa www.sciencedirect.com CASE REPORT Kawasaki disease with Glucose-6-Phosphate Dehydrogenase deficiency, case report Hesham Radi Obeidat a,*, Sahar Al-Dossary b, Abdulsalam Asseri a a Pharmacy Department, Saad Specialist Hospital, Alkhobar 31952, Saudi Arabia b Pediatric and Neonatology Department, Saad Specialist Hospital, Alkhobar 31952, Saudi Arabia Received 28 August 2014; accepted 11 November 2014 KEYWORDS Abstract Kawasaki disease (KD) is an acute, self-limited vasculitis of unknown etiology that Kawasaki disease; occurs predominantly in infants and children younger than 5 years of age. Coronary artery abnor- G6PD; malities are the most serious complication. Aspirin Based on the literatures infusion of Intravenous Immunoglobulin of 2 g/kg and a high dose of oral aspirin up to 100 mg/kg/day are the standard treatment for Kawasaki disease in the acute stage, and should be followed by antiplatelet dose of aspirin for thrombocytosis. Glucose-6-Phos- phate Dehydrogenase (G6PD) deficiency is an inherited X-linked hereditary disorder, and aspirin can induce hemolysis in patients with G6PD deficiency. We report a case of a 5 year and 8 month old male with KD and G6PD deficiency. ª 2014 The Authors. Production and hosting by Elsevier B.V. on behalf of King Saud University. This is an open access article under the CC BY-NC-ND license (http://creativecommons.org/licenses/by-nc-nd/3.0/). 1. Introduction genetic predisposition or infectious agents are likely to be the cause. Kawasaki disease was first described in 1967 by Tomisaku Recently, guidelines were published by the American Heart Kawasaki and has replaced acute rheumatic fever as the lead- Association (AHA) to aid in the diagnosis and management of ing cause of acquired heart disease among children in devel- Kawasaki disease. -

Newborn Screening Result for Bart's Hemoglobin
NEWBORN SCREENING RESULT FOR BART’S HEMOGLOBIN Physician’s information sheet developed by the Nebraska Newborn Screening Program with review by James Harper, MD, Pediatric Hematologist with UNMC Follow-up program, and member of the Nebraska Newborn Screening Advisory Committee. BACKGROUND The alpha thalassemias result from the loss of alpha globin genes. There are normally four genes for alpha globin production so that the loss of one to four genes is possible. The lack of one gene causes alpha thalassemia 2 (silent carrier) with no clinically detectable problems but may cause small amounts of hemoglobin Barts to be present in newborn blood samples. Alpha thalassemia trait (Alpha thalassemia 1) results from loss of two genes and causes a mild microcytic anemia which may resemble iron deficiency anemia. The loss of three genes causes hemoglobin H diseases which is a moderately severe form of thalassemia. The lack of all four genes causes hydrops fetalis and is usually fatal in utero. In general, only the loss of one or two genes is seen in African Americans. Individuals from Southeast Asia and the Mediterranean may have all four types of alpha thalassemia. The percentage of hemoglobin Barts in the blood sample may indicate the number of alpha genes that have been lost. However, the percentage of hemoglobin Barts is not directly measurable with the current methodology used by the newborn screening laboratory. Only the presence of Barts hemoglobin in relation to fetal and adult hemoglobin, and variants S, C, D and E can be detected. RECOMMENDED WORK UP In addition to the standard newborn hemoglobinopathy confirmation (hemoglobin electrophoresis), to separate those patients with alpha thalassemia silent carrier from the patients with alpha thalassemia trait, we recommend that these babies have the following labs drawn at their 6 month well baby check: CBC with retic count, ferritin, and a hemoglobin electropheresis. -

Hemolytic Anemia Caused by Hereditary Pyruvate Kinase Deficiency in a West Highland White Terrier Dog
Arch Med Vet 44, 195-200 (2012) COMMUNICATION Hemolytic anemia caused by hereditary pyruvate kinase deficiency in a West Highland White Terrier dog Anemia hemolítica causada por la deficiencia de piruvato quinasa hereditaria en un perro West Highland White Terrier NRC Hlavaca, LA Lacerdaa*, FO Conradob, PS Hünninga, M Seibertc, FHD Gonzálezd, U Gigere aPostgraduate Program in Veterinary Sciences, Universidade Federal Rio Grande do Sul, Porto Alegre, RS, Brasil. bVeterinary Clinic Pathology, Universidade Federal Rio Grande do Sul, Porto Alegre, RS, Brasil. cClinic Pathology, PetLab Ltda, Porto Alegre, Brasil. dDepartment of Veterinary Clinic Pathology, Faculty of Veterinary, Universidade Federal Rio Grande do Sul, Porto Alegre, RS, Brasil. eLaboratory of Genetic Diseases, University of Pennsilvania, Philadelphia, United States. RESUMEN La deficiencia de piruvato quinasa (PK) es un desorden hemolítico autosómico recesivo descrito en perros y gatos. La piruvato quinasa es una de las enzimas regulatorias esenciales de la glicólisis anaeróbica, la deficiencia de esta enzima causa una destrucción prematura de los eritrocitos. El presente es un estudio de caso y relata los hallazgos clínicos y paraclínicos en un perro brasileño de la raza West Highland White Terrier (WHWT) con historia de debilidad e intolerancia al ejercicio. El paciente presentaba mucosas pálidas, anemia hemolítica bastante regenerativa y osteoclerosis. La deficiencia de PK fue confirmada a través de una prueba de ADN raza específica para la inserción 6bp en el extremo 3’ del exón 10 de la secuencia del gen de la piruvato quinasa eritrocitaria (R-PK) como fue descrito. Al perro se le practicó eutanasia a los 20 meses de edad debido al deterioro de su estado clínico, el cual incluyó anemia e incompatibilidad sanguínea. -

Supporting Information
Supporting Information Biagioli et al. 10.1073/pnas.0813216106 SI Text and the procedure was repeated twice. Single cells were then RNA Isolation, Reverse Transcription, qPCR, Cloning, and Sequencing. resuspended in panning buffer and incubated on lectin-coated RNA was extracted from cell lines and blood by using TRIzol panning plates for 15 min at room temperature. Nonadherent reagent (Invitrogen) and following vendor instructions. cells were transferred to the next panning plate (four pannings, RNA was extracted from LCM- or FACS-purified cells with an 15 min each). Then, nonadherent cells were collected, centri- Absolutely RNA Nanoprep Kit (Stratagene). Single-strand fuged, and resuspended in serum-free neuronal medium or PBS. cDNA was obtained from purified RNA by using the iSCRIPT A similar procedure was also followed for the dissociation of cDNA Synhesis Kit (Bio-Rad) according to the manufacturer’s cortical and hippocampal astrocytes and oligodendrocytes. instructions. Quantitative RT-PCR was performed by using A cell strainer with 70-m nylon mesh was used to obtain a SYBER-Green PCR Master Mix and an iQ5 Real-Time PCR single-cell suspension (BD Falcon) before sorting. 7-AAD Detection System (Bio-Rad). (Beckman–Coulter) was added to the cell suspension to exclude Quantitative RT-PCR was performed with an iCycler IQ dead cells. A high-speed cell sorter (MoFlo) was used to sort (Bio-Rad); -actin was used as an endogenous control to nor- subpopulation of cells expressing GFP. Sorting parameters used malize the expression level of target genes. Primers were chosen for the three different populations are visualized in Fig. -
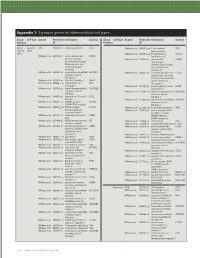
Technical Note, Appendix: an Analysis of Blood Processing Methods to Prepare Samples for Genechip® Expression Profiling (Pdf, 1
Appendix 1: Signature genes for different blood cell types. Blood Cell Type Source Probe Set Description Symbol Blood Cell Type Source Probe Set Description Symbol Fraction ID Fraction ID Mono- Lympho- GSK 203547_at CD4 antigen (p55) CD4 Whitney et al. 209813_x_at T cell receptor TRG nuclear cytes gamma locus cells Whitney et al. 209995_s_at T-cell leukemia/ TCL1A Whitney et al. 203104_at colony stimulating CSF1R lymphoma 1A factor 1 receptor, Whitney et al. 210164_at granzyme B GZMB formerly McDonough (granzyme 2, feline sarcoma viral cytotoxic T-lymphocyte- (v-fms) oncogene associated serine homolog esterase 1) Whitney et al. 203290_at major histocompatibility HLA-DQA1 Whitney et al. 210321_at similar to granzyme B CTLA1 complex, class II, (granzyme 2, cytotoxic DQ alpha 1 T-lymphocyte-associated Whitney et al. 203413_at NEL-like 2 (chicken) NELL2 serine esterase 1) Whitney et al. 203828_s_at natural killer cell NK4 (H. sapiens) transcript 4 Whitney et al. 212827_at immunoglobulin heavy IGHM Whitney et al. 203932_at major histocompatibility HLA-DMB constant mu complex, class II, Whitney et al. 212998_x_at major histocompatibility HLA-DQB1 DM beta complex, class II, Whitney et al. 204655_at chemokine (C-C motif) CCL5 DQ beta 1 ligand 5 Whitney et al. 212999_x_at major histocompatibility HLA-DQB Whitney et al. 204661_at CDW52 antigen CDW52 complex, class II, (CAMPATH-1 antigen) DQ beta 1 Whitney et al. 205049_s_at CD79A antigen CD79A Whitney et al. 213193_x_at T cell receptor beta locus TRB (immunoglobulin- Whitney et al. 213425_at Homo sapiens cDNA associated alpha) FLJ11441 fis, clone Whitney et al. 205291_at interleukin 2 receptor, IL2RB HEMBA1001323, beta mRNA sequence Whitney et al. -

Recombinant Human Hemoglobin Subunit Theta-1/HBQ1 (N-6His)
9853 Pacific Heights Blvd. Suite D. San Diego, CA 92121, USA Tel: 858-263-4982 Email: [email protected] 32-8592: Recombinant Human Hemoglobin Subunit theta-1/HBQ1 (N-6His) Gene : HBQ1 Gene ID : 3049 Uniprot ID : P09105 Description Source: E.coli. MW :17.7kD. Recombinant Human Hemoglobin subunit theta-1 is produced by our E.coli expression system and the target gene encoding Met1-Arg142 is expressed with a 6His tag at the N-terminus. Hemoglobin subunit theta-1 is a protein that in humans is encoded by the HBQ1 gene. Theta-globin mRNA is originally found in human fetal erythroid tissue but not in adult erythroid or other nonerythroid tissue. Theta-1 is a member of the human alpha-globin gene cluster that includes five functional genes and two pseudogenes. Research supports a transcriptionally active role for the gene and a functional role for the peptide in specific cells, possibly those of early erythroid tissue. Hemoglobin has a quaternary structure characteristically composed of many multi- subunit globular proteins. Most of the amino acids in hemoglobin form alpha helices, connected by short non-helical segments. Hydrogen bonds stabilize the helical sections inside this protein, causing attractions within the molecule, folding each polypeptide chain into a specific shape. Hemoglobin's quaternary structure comes from its four subunits in roughly a tetrahedral arrangement. Product Info Amount : 10 µg / 50 µg Content : Lyophilized from a 0.2 µm filtered solution of 20mM PB, 150mM NaCl, pH 7.0. Lyophilized protein should be stored at -20°C, though stable at room temperature for 3 weeks. -

Simultaneous Detection of Target Cnvs and Snvs of Thalassemia by Multiplex PCR and Next‑Generation Sequencing
MOLECULAR MEDICINE REPORTS 19: 2837-2848, 2019 Simultaneous detection of target CNVs and SNVs of thalassemia by multiplex PCR and next‑generation sequencing DONG-MEI FAN1*, XU YANG2*, LI-MIN HUANG1, GUO-JUN OUYANG3, XUE-XI YANG1 and MING LI1 1Institute of Antibody Engineering, School of Laboratory Medicine and Biotechnology, Southern Medical University, Guangzhou, Guangdong 510515; 2Clinical Innovation and Research Center, Shenzhen Hospital of Southern Medical University, Shenzhen, Guangdong 518110; 3Guangzhou Darui Biotechnology Co., Ltd., Guangzhou, Guangdong 510663, P.R. China Received May 16, 2018; Accepted December 3, 2018 DOI: 10.3892/mmr.2019.9896 Abstract. Thalassemia is caused by complex mechanisms, Introduction including copy number variants (CNVs) and single nucleotide variants (SNVs). The CNV types of α-thalassemia are typi- Thalassemia is caused by copy number variants (CNVs) and cally detected by gap-polymerase chain reaction (PCR). The single nucleotide variants (SNVs) in the α-globin (HBA) or SNV types are detected by Sanger sequencing. In the present β-globin (HBB) genes that result in the absence or lack of α- or study, a novel method was developed that simultaneously β-globin chains, and ultimately hemolytic anemia. It is esti- detects CNVs and SNVs by multiplex PCR and next-genera- mated that ~7% of the world population carries the gene for the tion sequencing (NGS). To detect CNVs, 33 normal samples disease (1), and the birth rate of children with hemoglobin (Hb) were used as a cluster of control values to build a baseline, disorders is ≥2.4% per year (2). Thalassemia occurs most in the and the A, B, C, and D ratios were developed to evaluate-SEA, Mediterranean region, East South Asia, and the subcontinents -α4.2, -α3.7, and compound or homozygous CNVs, respectively. -

Acoi Board Review 2019 Text
CHERYL KOVALSKI, DO FACOI NO DISCLOSURES ACOI BOARD REVIEW 2019 TEXT ANEMIA ‣ Hemoglobin <13 grams or ‣ Hematocrit<39% TEXT ANEMIA MCV RETICULOCYTE COUNT Corrected retic ct = hematocrit/45 x retic % (45 considered normal hematocrit) >2%: blood loss or hemolysis <2%: hypoproliferative process TEXT ANEMIA ‣ MICROCYTIC ‣ Obtain and interpret iron studies ‣ Serum iron ‣ Total iron binding capacity (TIBC) ‣ Transferrin saturation ‣ Ferritin-correlates with total iron stores ‣ can be normal or increased if co-existent inflammation TEXT IRON DEFICIENCY ‣ Most common nutritional problem in the world ‣ Absorbed in small bowel, enhanced by gastric acid ‣ Absorption inhibited by inflammation, phytates (bran) & tannins (tea) TEXT CAUSES OF IRON DEFICIENCY ‣ Blood loss – most common etiology ‣ Decreased intake ‣ Increased utilization-EPO therapy, chronic hemolysis ‣ Malabsorption – gastrectomy, sprue ‣ ‣ ‣ TEXT CLINICAL MANIFESTATIONS OF IRON DEFICIENCY ‣ Impaired psychomotor development ‣ Fatigue, Irritability ‣ PICA ‣ Koilonychiae, Glossitis, Angular stomatitis ‣ Dysphagia TEXT IRON DEFICIENCY LAB FINDINGS ‣ Low serum iron, increased TIBC ‣ % sat <20 TEXT MANAGEMENT OF IRON DEFICIENCY ‣ MUST LOOK FOR SOURCE OF BLEED: ie: GI, GU, Regular blood donor ‣ Replacement: 1. Oral: Ferrous sulfate 325 mg TID until serum iron, % sat, and ferritin mid-range normal, 6-12 months 2. IV TEXT SIDEROBLASTIC ANEMIAS Diverse group of disorders of RBC production characterized by: 1. Defect involving incorporation of iron into heme molecule 2. Ringed sideroblasts in -

Approach to Anemia
APPROACH TO ANEMIA Mahsa Mohebtash, MD Medstar Union Memorial Hospital Definition of Anemia • Reduced red blood mass • RBC measurements: RBC mass, Hgb, Hct or RBC count • Hgb, Hct and RBC count typically decrease in parallel except in severe microcytosis (like thalassemia) Normal Range of Hgb/Hct • NL range: many different values: • 2 SD below mean: < Hgb13.5 or Hct 41 in men and Hgb 12 or Hct of 36 in women • WHO: Hgb: <13 in men, <12 in women • Revised WHO/NCI: Hgb <14 in men, <12 in women • Scrpps-Kaiser based on race and age: based on 5th percentiles of the population in question • African-Americans: Hgb 0.5-1 lower than Caucasians Approach to Anemia • Setting: • Acute vs chronic • Isolated vs combined with leukopenia/thrombocytopenia • Pathophysiologic approach • Morphologic approach Reticulocytes • Reticulocytes life span: 3 days in bone marrow and 1 day in peripheral blood • Mature RBC life span: 110-120 days • 1% of RBCs are removed from circulation each day • Reticulocyte production index (RPI): Reticulocytes (percent) x (HCT ÷ 45) x (1 ÷ RMT): • <2 low Pathophysiologic approach • Decreased RBC production • Reduced effective production of red cells: low retic production index • Destruction of red cell precursors in marrow (ineffective erythropoiesis) • Increased RBC destruction • Blood loss Reduced RBC precursors • Low retic production index • Lack of nutrients (B12, Fe) • Bone marrow disorder => reduced RBC precursors (aplastic anemia, pure RBC aplasia, marrow infiltration) • Bone marrow suppression (drugs, chemotherapy, radiation) -

Laboratory Approach to Anemia Laboratory Approach to Anemia
DOI: 10.5772/intechopen.70359 Provisional chapter Chapter 12 Laboratory Approach to Anemia Laboratory Approach to Anemia Ebru Dündar Yenilmez and Abdullah Tuli Ebru Dündar Yenilmez and Abdullah Tuli Additional information is available at the end of the chapter Additional information is available at the end of the chapter http://dx.doi.org/10.5772/intechopen.70359 Abstract Anemia is a major cause of morbidity and mortality worldwide and can be defined as a decreased quantity of circulating red blood cells (RBCs). The epidemiological studies suggested that one-third of the world’s population is affected with anemia. Anemia is not a disease, but it is instead the sign of an underlying basic pathological process. However, the sign may function as a compass in the search for the cause. Therefore, the prediag- nosis revealed by thorough investigation of this sign should be supported by laboratory parameters according to the underlying pathological process. We expect that this review will provide guidance to clinicians with findings and laboratory tests that can be followed from the initial stage in the anemia search. Keywords: anemia, complete blood count, red blood cell indices, reticulocyte 1. Introduction Anemia, the meaning of which in Greek is “without blood,” is a relatively common sign and symptom of various medical conditions. Anemia is defined as a significant decrease in the count of total erythrocyte [red blood cell (RBC)] mass, although this definition is rarely used in clinical settings. According to the World Health Organization, anemia is a condition in which the number of red blood cells (RBCs, and consequently their oxygen-carrying capacity) is insufficient to meet the body’s physiologic needs [1, 2]. -

Clinical Evaluation of Different Types of Anemia 1Richa Saxena, 2Sudha Chamoli, 3Monisha Batra
WJOA Richa Saxena et al 10.5005/jp-journals-10065-0024 REVIEW ARTICLE Clinical Evaluation of Different Types of Anemia 1Richa Saxena, 2Sudha Chamoli, 3Monisha Batra World Health Organization criteria for anemia ABSTRACT Table 1: Venous blood (gm/dL) MCHC Anemia, defined as a hemoglobin level two standard devia- Adult males 13 34 tions below the mean for age, is prevalent among infants and children as well as adults worldwide. The evaluation Adult females, nonpregnant 12 34 of an individual with anemia should begin with a thorough Adult females, pregnant 11 34 history and risk assessment. Characterizing the anemia as Children (6 months–6 years) 11 34 microcytic, normocytic, or macrocytic based on the mean Children (6–14 years) 12 34 corpuscular volume (MCV) will aid in the work-up and man- agement. Microcytic anemia due to iron deficiency is the most common type of anemia in children. Iron deficiency CLASSIFICATION OF ANEMIA anemia, which can be associated with cognitive issues, is prevented and treated with iron supplements or increased The classification of anemia based on two factors (Table 2): intake of dietary iron. This review article discusses the clinical 1. Red cell morphology evaluation of different types of anemias based on the find- 2. Etiology of anemia ings of clinical examination (i.e., pallor, pedal edema, nail changes, and epithelial changes) as well as the results of Anemia Classification Based on Morphology various investigations such as routine blood investigations (hemoglobin, mean cell hemoglobin concentration [MCHC], Anemia can be classified based on morphology as: packed cell volume, etc.), peripheral smear examination, • Normocytic normochromic (MCV 76–96 fL, MCHC bone marrow examination, etc.