Drug-Induced Ototoxicity
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Management of Adult Clostridium Difficile Digestive Contaminations: a Literature Review
European Journal of Clinical Microbiology & Infectious Diseases (2019) 38:209–231 https://doi.org/10.1007/s10096-018-3419-z REVIEW Management of adult Clostridium difficile digestive contaminations: a literature review Fanny Mathias1 & Christophe Curti1,2 & Marc Montana1,3 & Charléric Bornet4 & Patrice Vanelle 1,2 Received: 5 October 2018 /Accepted: 30 October 2018 /Published online: 29 November 2018 # Springer-Verlag GmbH Germany, part of Springer Nature 2018 Abstract Clostridium difficile infections (CDI) dramatically increased during the last decade and cause a major public health problem. Current treatments are limited by the high disease recurrence rate, severity of clinical forms, disruption of the gut microbiota, and colonization by vancomycin-resistant enterococci (VRE). In this review, we resumed current treatment options from official recommendation to promising alternatives available in the management of adult CDI, with regard to severity and recurring or non-recurring character of the infection. Vancomycin remains the first-line antibiotic in the management of mild to severe CDI. The use of metronidazole is discussed following the latest US recommendations that replaced it by fidaxomicin as first-line treatment of an initial episode of non-severe CDI. Fidaxomicin, the most recent antibiotic approved for CDI in adults, has several advantages compared to vancomycin and metronidazole, but its efficacy seems limited in cases of multiple recurrences. Innovative therapies such as fecal microbiota transplantation (FMT) and antitoxin antibodies were developed to limit the occurrence of recurrence of CDI. Research is therefore very active, and new antibiotics are being studied as surotomycin, cadazolid, and rinidazole. Keywords Clostridiumdifficile .Fidaxomicin .Fecalmicrobiotatransplantation .Antitoxinantibodies .Surotomycin .Cadazolid Introduction (Fig. -

Prediction of Premature Termination Codon Suppressing Compounds for Treatment of Duchenne Muscular Dystrophy Using Machine Learning
Prediction of Premature Termination Codon Suppressing Compounds for Treatment of Duchenne Muscular Dystrophy using Machine Learning Kate Wang et al. Supplemental Table S1. Drugs selected by Pharmacophore-based, ML-based and DL- based search in the FDA-approved drugs database Pharmacophore WEKA TF 1-Palmitoyl-2-oleoyl-sn-glycero-3- 5-O-phosphono-alpha-D- (phospho-rac-(1-glycerol)) ribofuranosyl diphosphate Acarbose Amikacin Acetylcarnitine Acetarsol Arbutamine Acetylcholine Adenosine Aldehydo-N-Acetyl-D- Benserazide Acyclovir Glucosamine Bisoprolol Adefovir dipivoxil Alendronic acid Brivudine Alfentanil Alginic acid Cefamandole Alitretinoin alpha-Arbutin Cefdinir Azithromycin Amikacin Cefixime Balsalazide Amiloride Cefonicid Bethanechol Arbutin Ceforanide Bicalutamide Ascorbic acid calcium salt Cefotetan Calcium glubionate Auranofin Ceftibuten Cangrelor Azacitidine Ceftolozane Capecitabine Benserazide Cerivastatin Carbamoylcholine Besifloxacin Chlortetracycline Carisoprodol beta-L-fructofuranose Cilastatin Chlorobutanol Bictegravir Citicoline Cidofovir Bismuth subgallate Cladribine Clodronic acid Bleomycin Clarithromycin Colistimethate Bortezomib Clindamycin Cyclandelate Bromotheophylline Clofarabine Dexpanthenol Calcium threonate Cromoglicic acid Edoxudine Capecitabine Demeclocycline Elbasvir Capreomycin Diaminopropanol tetraacetic acid Erdosteine Carbidopa Diazolidinylurea Ethchlorvynol Carbocisteine Dibekacin Ethinamate Carboplatin Dinoprostone Famotidine Cefotetan Dipyridamole Fidaxomicin Chlormerodrin Doripenem Flavin adenine dinucleotide -

Antibiotic Fidaxomicin Is an Rdrp Inhibitor As a Potential New Therapeutic Agent Against Zika Virus
Yuan et al. BMC Medicine (2020) 18:204 https://doi.org/10.1186/s12916-020-01663-1 RESEARCH ARTICLE Open Access Antibiotic fidaxomicin is an RdRp inhibitor as a potential new therapeutic agent against Zika virus Jie Yuan1,2,3†, Jianchen Yu2,3,4†, Yun Huang3, Zhenjian He2,5, Jia Luo6, Yun Wu2,4,7, Yingchun Zheng8, Jueheng Wu2,4,7, Xun Zhu2,4, Haihe Wang1 and Mengfeng Li2,3,4* Abstract Background: Zika virus (ZIKV) infection is a global health problem, and its complications, including congenital Zika syndrome and Guillain-Barré syndrome, constitute a continued threat to humans. Unfortunately, effective therapeutics against ZIKV infection are not available thus far. Methods: We screened the compounds collection consisting of 1789 FDA-approved drugs by a computational docking method to obtain anti-ZIKV candidate compounds targeting ZIKV RNA-dependent RNA polymerase (RdRp). SPR (BIAcore) assay was employed to demonstrate the candidate compounds’ direct binding to ZIKV RdRp, and polymerase activity assay was used to determine the inhibitory effect on ZIKV RdRp-catalyzed RNA synthesis. The antiviral effects on ZIKV in vitro and in vivo were detected in infected cultured cells and in Ifnar1−/− mice infected by ZIKV virus using plaque assay, western blotting, tissue immunofluorescence, and immunohistochemistry. Results: Here, we report that a first-in-class macrocyclic antibiotic, which has been clinically used to treat Clostridium difficile infection, fidaxomicin, potently inhibits ZIKV replication in vitro and in vivo. Our data showed that fidaxomicin was effective against African and Asian lineage ZIKV in a wide variety of cell lines of various tissue origins, and prominently suppressed ZIKV infection and significantly improved survival of infected mice. -

Objective Measures of Ototoxicity
The following article appeared in the September 2005 issue (Vol. 9, No. 1, pp. 10-16) of the Division 6 publication Perspectives on Hearing and Hearing Disorders: Research and Diagnostics. To learn more about Division 6, contact the ASHA Action Center at 1-800-498-2071 or visit the division’s Web page at www.asha.org/about/membership-certification/divs/div_6.htm. Objective Measures of Ototoxicity Elizabeth Leigh-Paffenroth Veterans Affairs Rehabilitation Research and Development Service, National Center for Rehabilitative Auditory Research, VA Medical Center Portland, OR Department of Otolaryngology, Oregon Health & Science University Portland, OR Kelly M. Reavis, Jane S. Gordon, Kathleen T. Dunckley Veterans Affairs Rehabilitation Research and Development Service, National Center for Rehabilitative Auditory Research, VA Medical Center Portland, OR Stephen A. Fausti and Dawn Konrad-Martin Veterans Affairs Rehabilitation Research and Development Service, National Center for Rehabilitative Auditory Research, VA Medical Center Portland, OR Department of Otolaryngology, Oregon Health & Science University, Portland, OR A leading cause of preventable sensorineural hear- of patients who are unable to provide reliable re- ing loss is therapeutic treatment with medications that sponses; subsequently, many of these patients do not are toxic to inner ear tissues, including certain drugs receive monitoring for ototoxic-induced changes in used to fight cancer and life-threatening infectious dis- their hearing. The development of objective measures eases. Ototoxic-induced hearing loss typically begins that do not require patient cooperation is necessary to in the high frequencies and progresses to lower fre- monitor all patients receiving ototoxic drugs. quencies as drug administration continues (Campbell Two objective measures offer promise in their abil- & Durrant, 1993; Campbell et al., 2003; Macdonald, ity to detect and to monitor hearing changes caused by Harrison, Wake, Bliss, & Macdonald, 1994). -

IEHP Medi-Cal Prior Authorization Criteria February 2020
IEHP Medi-Cal Prior Authorization Criteria February 2020 Generic Brand Criteria Covered Uses: Osteoporosis Exclusion Criteria: N/A Required Medical Information: Must meet all of the following requirements: a. Documentation of a T-score less than -2.5 at the lumbar spine, hip (total hip or femoral neck), or radius (one-third radius site). b. Documented inadequate response (e.g. greater than 3 percent decrease in bone mineral density from baseline, fracture from minimal trauma)while receiving the following, or clinically significant adverse effects to all of the following: abaloparatide Tymlos i. An oral bisphosphonate (e.g. alendronate) ii. An intravenous bisphosphonate (e.g. zoledronic acid) iii. Prolia c. Patient is concurrently receiving calcium and vitamin D supplement. d. The combined duration of treatment with any parathyroid hormone analogs has not exceeded a lifetime maximum of 24 months (i.e. abaloparatide and teriparatide) Age Restrictions: N/A Prescriber Restrictions: N/A abobotulinum toxin Dysport Please refer to Botulinum Toxin Drug Class Prior Authorization Criteria A Covered Uses: Herpes labialis or herpes febrilis (cold sore) Exclusion Criteria: N/A acyclovir 5% topical Required Medical Information: Must meet the following requirement: cream a. Failure or clinically significant adverse effects to the alternative: Abreva Age Restrictions: Must be age 12 or older Prescriber Restrictions: N/A Detailed Prior Authorization criteria can be found at: https://www.iehp.org/en/providers/pharmacy-services/rx-pa-drug- treatment-criteria Generic Brand Criteria Covered Uses: Must meet "1" of the following: a. Genital herpes simplex virus infection (HSV) b. Non-life threatening mucocutaneous herpes simplex virus infection, patient immunocompromised acyclovir topical Exclusion Criteria: N/A Required Medical Information: Must meet the following requirement: ointment a. -

DIFICID® (Fidaxomicin) Oral
PHARMACY COVERAGE GUIDELINES ORIGINAL EFFECTIVE DATE: 7/16/2015 SECTION: DRUGS LAST REVIEW DATE: 8/19/2021 LAST CRITERIA REVISION DATE: 8/19/2021 ARCHIVE DATE: DIFICID® (fidaxomicin) oral Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy Coverage Guideline must be read in its entirety to determine coverage eligibility, if any. This Pharmacy Coverage Guideline provides information related to coverage determinations only and does not imply that a service or treatment is clinically appropriate or inappropriate. The provider and the member are responsible for all decisions regarding the appropriateness of care. Providers should provide BCBSAZ complete medical rationale when requesting any exceptions to these guidelines. The section identified as “Description” defines or describes a service, procedure, medical device or drug and is in no way intended as a statement of medical necessity and/or coverage. The section identified as “Criteria” defines criteria to determine whether a service, procedure, medical device or drug is considered medically necessary or experimental or investigational. State or federal mandates, e.g., FEP program, may dictate that any drug, device or biological product approved by the U.S. Food and Drug Administration (FDA) may not be considered experimental or investigational and thus the drug, device or biological product may be assessed only on the basis of medical necessity. Pharmacy Coverage Guidelines are subject to change as new information becomes available. For purposes of this Pharmacy Coverage Guideline, the terms "experimental" and "investigational" are considered to be interchangeable. BLUE CROSS®, BLUE SHIELD® and the Cross and Shield Symbols are registered service marks of the Blue Cross and Blue Shield Association, an association of independent Blue Cross and Blue Shield Plans. -

Preventing Hearing Loss Caused by Chemical (Ototoxicity) and Noise Exposure
Preventing Hearing Loss Caused by Chemical (Ototoxicity) and Noise Exposure Safety and Health Information Bulletin SHIB 03-08-2018 DHHS (NIOSH) Publication No. 2018-124 Introduction Millions of workers are exposed to noise in the workplace every day and when uncontrolled, noise exposure may cause permanent hearing loss. Research demonstrates exposure to certain chemicals, called ototoxicants, may cause hearing loss or balance problems, regardless of noise exposure. Substances including certain pesticides, solvents, and pharmaceuticals that contain ototoxicants can negatively affect how the ear functions, causing hearing loss, and/or affect balance. Source/Copyright: OSHA The risk of hearing loss is increased when workers are exposed to these chemicals while working around elevated noise levels. This combination often results in hearing loss that can be temporary or permanent, depending on the level of noise, the dose of the chemical, and the duration of the exposure. This hearing impairment affects many occupations and industries, from machinists to firefighters. Effects on Hearing Harmful exposure to ototoxicants may occur through inhalation, ingestion, or skin absorption. Health effects caused by ototoxic chemicals vary based on exposure frequency, intensity, duration, workplace exposure to other hazards, and individual factors such as age. Effects may be temporary or permanent, can affect hearing sensitivity and result in a standard threshold shift. Since chemicals can affect central portions of the auditory system (e.g., nerves or nuclei in the central nervous system, the pathways to the brain or in the brain itself), not only do sounds need to be louder to be detected, but also they lose clarity. Specifically, speech discrimination dysfunction, the ability to hear voices separately from background noise, may occur and involve: . -

Fidaxomicin Related Metabolic Acidosis
ABSTRACT NUMBER FIDAXOMICIN RELATED METABOLIC 5PSQ-121 ACIDOSIS: A CASE REPORT B. Serna Serrano1, A. Serrano Martínez 2, A. Valladolid Walsh 1, I. Pérez Alpuente 1, V. Lerma Gaude 1, M. Clemente Andújar 1, R. Aldaz Francés 1 Section 5: Patient Safety 1Complejo Hospitalario Universitario de Albacete, Pharmacy, Albacete, Spain. and Quality Assurance 2Complejo Hospitalario Universitario de Albacete, Hematology, Albacete, Spain. ATC code: A07 - Antidiarrheals, intestinal antiinflammatory/antiinfective agents BACKGROUND AND IMPORTANCE Fidaxomicin is a macrolide antibiotic used to treat intestinal Clostridium difficile (CD) infection in case of absence to metronidazole or vancomycin treatments. On the other hand, pharmacovigilance collects information, analyzes and notifies cases of suspected Adverse Drug Reactions (ADRs) in order to prevent them in the future. AIM AND To describe a case of metabolic acidosis OBJECTIVES in a patient treated with fidaxomicin and establish it is possible association. MATERIALS AND METHODS We describe the case of an 82-year-old male, diagnosed with multiple myeloma and treated with two full cycles of bortezomib-dexamethasone. He was referred to the Emergency department after presenting melenic diarrhea for one week. As a result, he was hospitalized and diagnosed with upper gastrointestinal bleeding, acute prerenal renal failure, mild thrombopenia, hypokalemia and hyponatremia. After fluid and electrolyte stabilization, it was decided to start with fidaxomicin 200 mg/12h due to fever, confusional syndrome, persistence of diarrhea and positive CD toxin test. The following constants were measured to confirm metabolic - acidosis: gas level of bicarbonate (HCO3 ), partial pressure of carbon dioxide (pCO2), hydrogen ion potential (pH) and anion GAP. The degree of drug/adverse reaction causality was evaluated using the Naranjo algorithm. -

Patent Application Publication ( 10 ) Pub . No . : US 2019 / 0192440 A1
US 20190192440A1 (19 ) United States (12 ) Patent Application Publication ( 10) Pub . No. : US 2019 /0192440 A1 LI (43 ) Pub . Date : Jun . 27 , 2019 ( 54 ) ORAL DRUG DOSAGE FORM COMPRISING Publication Classification DRUG IN THE FORM OF NANOPARTICLES (51 ) Int . CI. A61K 9 / 20 (2006 .01 ) ( 71 ) Applicant: Triastek , Inc. , Nanjing ( CN ) A61K 9 /00 ( 2006 . 01) A61K 31/ 192 ( 2006 .01 ) (72 ) Inventor : Xiaoling LI , Dublin , CA (US ) A61K 9 / 24 ( 2006 .01 ) ( 52 ) U . S . CI. ( 21 ) Appl. No. : 16 /289 ,499 CPC . .. .. A61K 9 /2031 (2013 . 01 ) ; A61K 9 /0065 ( 22 ) Filed : Feb . 28 , 2019 (2013 .01 ) ; A61K 9 / 209 ( 2013 .01 ) ; A61K 9 /2027 ( 2013 .01 ) ; A61K 31/ 192 ( 2013. 01 ) ; Related U . S . Application Data A61K 9 /2072 ( 2013 .01 ) (63 ) Continuation of application No. 16 /028 ,305 , filed on Jul. 5 , 2018 , now Pat . No . 10 , 258 ,575 , which is a (57 ) ABSTRACT continuation of application No . 15 / 173 ,596 , filed on The present disclosure provides a stable solid pharmaceuti Jun . 3 , 2016 . cal dosage form for oral administration . The dosage form (60 ) Provisional application No . 62 /313 ,092 , filed on Mar. includes a substrate that forms at least one compartment and 24 , 2016 , provisional application No . 62 / 296 , 087 , a drug content loaded into the compartment. The dosage filed on Feb . 17 , 2016 , provisional application No . form is so designed that the active pharmaceutical ingredient 62 / 170, 645 , filed on Jun . 3 , 2015 . of the drug content is released in a controlled manner. Patent Application Publication Jun . 27 , 2019 Sheet 1 of 20 US 2019 /0192440 A1 FIG . -

Telephone Request Service EAP Criteria
Telephone Request Service Reimbursement Criteria Exceptional Access Program, Ministry of Health and Long-Term Care Disclaimer: The information in this document is updated on a regular basis. Although we strive to ensure that all information is accurate at the time of posting, please be aware that some items may be subject to change from time-to-time. The information provided in this document and website is intended for information purposes only and does not provide any medical diagnosis, symptom assessment, health counseling or medical opinion for individual users. This information also does not constitute medical advice for physicians or patients. For more detailed information on prescription drugs, please consult a qualified healthcare professional. Last updated August, 2015. The EAP response letter will list the specific drug, strength and dosage form that will be approved. Refer to the formulary for a list of interchangeable drug products that may be dispensed. 2 Table of contents TELEPHONE REQUEST SERVICE REIMBURSEMENT CRITERIA ............................. 1 Exceptional Access Program, Ministry of Health and Long-Term Care ................... 1 TABLE OF CONTENTS .................................................................................................. 3 INTRODUCTION ............................................................................................................. 6 ANTIBIOTICS ................................................................................................................. 7 Cefazolin ....................................................................................................................... -

Why Is Drug-Induced Ototoxicity Still Not Preventable Nor Is It Treatable in 2019? - a Literature Review of Aminoglycoside and Cisplatin Ototoxicity Cherrabi Kaoutar*
Review Article iMedPub Journals Archives of Medicine 2020 www.imedpub.com Vol.12 No.2:3 ISSN 1989-5216 DOI: 10.36648/1989-5216.12.2.304 Why is Drug-Induced Ototoxicity Still Not Preventable nor is it Treatable in 2019? - A Literature Review of Aminoglycoside and Cisplatin Ototoxicity Cherrabi Kaoutar* Department of Oto-Rhino-Laryngology and Cervico-Facial Studies, Fez, Morocco *Corresponding author: Cherrabi Kaoutar, Resident, Department of Oto-Rhino-Laryngology and Cervico-Facial Studies, Fez, Morocco, Tel: 0679382957; E-mail: [email protected] Received date: October 17, 2019; Accepted date: March 02, 2019; Published date: March 09, 2020 Citation: Cherrabi K (2020) Why is Drug-Induced Ototoxicity Still Not Preventable nor is it Treatable in 2019? - A Literature Review of Aminoglycoside and Cisplatin Ototoxicity. Arch Med Vol: 12 Iss: 2:3 Copyright: ©2020 Cherrabi K. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. The thorough exploration of inner ear proteome, transcriptome, and genome in physiological and Abstract pathological contexts as potential biomarkers of early stages of cell lesions: The clinical application would not Oto-toxicity is defined by cochlear and or vestibular only allow a more specific understanding of different cellular lesions due to the use of drugs. The difference in mechanisms of iatrogenic oto-toxicity, but also allow new specific mechanisms, the great semi logical variability, the insights into targeted protective and post-lesional heterogeneity of incriminated drugs, the inaccessibility to therapies. -
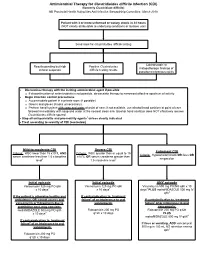
Clostridium Difficile Infection
Antimicrobial Therapy for Clostridioides difficile Infection (CDI) (formerly Clostridium difficile) NB Provincial Health Authorities Anti-Infective Stewardship Committee, March 2019 Patient with 3 or more unformed or watery stools in 24 hours (NOT clearly attributable to underlying conditions or laxative use) Send stool for Clostridioides difficile testing Colonoscopic or Results pending but high Positive Clostridioides histopathologic findings of clinical suspicion difficile testing results pseudomembranous colitis Discontinue therapy with the inciting antimicrobial agent if possible o If discontinuation of antimicrobials is not possible, de-escalate therapy to narrowest effective spectrum of activity Begin infection control precautions o Accommodate patient in a private room (if possible) o Gowns and gloves (masks unnecessary) o Perform hand hygiene with soap and water at point of care; if not available, use alcohol hand sanitizer at point of care followed immediately with soap and water at the nearest clean sink (alcohol hand sanitizer does NOT effectively remove Clostridioides difficile spores) Stop all anti-peristaltic and pro-motility agents1 unless clearly indicated Treat according to severity of CDI (see below) Mild-to-moderate CDI Severe CDI Fulminant CDI Criteria: WBC lower than 15 x109/L AND Criteria: WBC greater than or equal to 15 Criteria: Hypotension/shock OR ileus OR serum creatinine less than 1.5 x baseline x109/L OR serum creatinine greater than megacolon level2 1.5 x baseline level2 Initial episode Initial episode