Seroprevalence and Participatory Epidemiology of Camelpox in Afar Region of Ethiopia
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

An Analysis of the Afar-Somali Conflict in Ethiopia and Djibouti
Regional Dynamics of Inter-ethnic Conflicts in the Horn of Africa: An Analysis of the Afar-Somali Conflict in Ethiopia and Djibouti DISSERTATION ZUR ERLANGUNG DER GRADES DES DOKTORS DER PHILOSOPHIE DER UNIVERSTÄT HAMBURG VORGELEGT VON YASIN MOHAMMED YASIN from Assab, Ethiopia HAMBURG 2010 ii Regional Dynamics of Inter-ethnic Conflicts in the Horn of Africa: An Analysis of the Afar-Somali Conflict in Ethiopia and Djibouti by Yasin Mohammed Yasin Submitted in partial fulfilment of the requirements for the degree PHILOSOPHIAE DOCTOR (POLITICAL SCIENCE) in the FACULITY OF BUSINESS, ECONOMICS AND SOCIAL SCIENCES at the UNIVERSITY OF HAMBURG Supervisors Prof. Dr. Cord Jakobeit Prof. Dr. Rainer Tetzlaff HAMBURG 15 December 2010 iii Acknowledgments First and foremost, I would like to thank my doctoral fathers Prof. Dr. Cord Jakobeit and Prof. Dr. Rainer Tetzlaff for their critical comments and kindly encouragement that made it possible for me to complete this PhD project. Particularly, Prof. Jakobeit’s invaluable assistance whenever I needed and his academic follow-up enabled me to carry out the work successfully. I therefore ask Prof. Dr. Cord Jakobeit to accept my sincere thanks. I am also grateful to Prof. Dr. Klaus Mummenhoff and the association, Verein zur Förderung äthiopischer Schüler und Studenten e. V., Osnabruck , for the enthusiastic morale and financial support offered to me in my stay in Hamburg as well as during routine travels between Addis and Hamburg. I also owe much to Dr. Wolbert Smidt for his friendly and academic guidance throughout the research and writing of this dissertation. Special thanks are reserved to the Department of Social Sciences at the University of Hamburg and the German Institute for Global and Area Studies (GIGA) that provided me comfortable environment during my research work in Hamburg. -
Districts of Ethiopia
Region District or Woredas Zone Remarks Afar Region Argobba Special Woreda -- Independent district/woredas Afar Region Afambo Zone 1 (Awsi Rasu) Afar Region Asayita Zone 1 (Awsi Rasu) Afar Region Chifra Zone 1 (Awsi Rasu) Afar Region Dubti Zone 1 (Awsi Rasu) Afar Region Elidar Zone 1 (Awsi Rasu) Afar Region Kori Zone 1 (Awsi Rasu) Afar Region Mille Zone 1 (Awsi Rasu) Afar Region Abala Zone 2 (Kilbet Rasu) Afar Region Afdera Zone 2 (Kilbet Rasu) Afar Region Berhale Zone 2 (Kilbet Rasu) Afar Region Dallol Zone 2 (Kilbet Rasu) Afar Region Erebti Zone 2 (Kilbet Rasu) Afar Region Koneba Zone 2 (Kilbet Rasu) Afar Region Megale Zone 2 (Kilbet Rasu) Afar Region Amibara Zone 3 (Gabi Rasu) Afar Region Awash Fentale Zone 3 (Gabi Rasu) Afar Region Bure Mudaytu Zone 3 (Gabi Rasu) Afar Region Dulecha Zone 3 (Gabi Rasu) Afar Region Gewane Zone 3 (Gabi Rasu) Afar Region Aura Zone 4 (Fantena Rasu) Afar Region Ewa Zone 4 (Fantena Rasu) Afar Region Gulina Zone 4 (Fantena Rasu) Afar Region Teru Zone 4 (Fantena Rasu) Afar Region Yalo Zone 4 (Fantena Rasu) Afar Region Dalifage (formerly known as Artuma) Zone 5 (Hari Rasu) Afar Region Dewe Zone 5 (Hari Rasu) Afar Region Hadele Ele (formerly known as Fursi) Zone 5 (Hari Rasu) Afar Region Simurobi Gele'alo Zone 5 (Hari Rasu) Afar Region Telalak Zone 5 (Hari Rasu) Amhara Region Achefer -- Defunct district/woredas Amhara Region Angolalla Terana Asagirt -- Defunct district/woredas Amhara Region Artuma Fursina Jile -- Defunct district/woredas Amhara Region Banja -- Defunct district/woredas Amhara Region Belessa -- -

Modelling the Current Fractional Cover of an Invasive Alien Plant and Drivers of Its Invasion in a Dryland Ecosystem
www.nature.com/scientificreports OPEN Modelling the current fractional cover of an invasive alien plant and drivers of its invasion in a dryland Received: 23 July 2018 Accepted: 23 November 2018 ecosystem Published: xx xx xxxx Hailu Shiferaw1,3, Urs Schafner 2, Woldeamlak Bewket3, Tena Alamirew1, Gete Zeleke1, Demel Teketay4 & Sandra Eckert5 The development of spatially diferentiated management strategies against invasive alien plant species requires a detailed understanding of their current distribution and of the level of invasion across the invaded range. The objectives of this study were to estimate the current fractional cover gradient of invasive trees of the genus Prosopis in the Afar Region, Ethiopia, and to identify drivers of its invasion. We used seventeen explanatory variables describing Landsat 8 image refectance, topography, climate and landscape structures to model the current cover of Prosopis across the invaded range using the random forest (RF) algorithm. Validation of the RF algorithm confrmed high model performance with an accuracy of 92% and a Kappa-coefcient of 0.8. We found that, within 35 years after its introduction, Prosopis has invaded approximately 1.17 million ha at diferent cover levels in the Afar Region (12.3% of the surface). Normalized diference vegetation index (NDVI) and elevation showed the highest explanatory power among the 17 variables, in terms of both the invader’s overall distribution as well as areas with high cover. Villages and linear landscape structures (rivers and roads) were found to be more important drivers of future Prosopis invasion than environmental variables, such as climate and topography, suggesting that Prosopis is likely to continue spreading and increasing in abundance in the case study area if left uncontrolled. -

ETHIOPIA Humanitarian Access Situation Report June – July 2019
ETHIOPIA Humanitarian Access Situation Report June – July 2019 This report is produced by OCHA Ethiopia in collaboration with humanitarian partners. It covers the period June - July 2019. The next report will be issued around September - October 2019. OVERVIEW IUS • In June - July, Ethiopia experienced an at- TIGRAY 276 Access incidents reported tempted government overthrow in Amhara, Western socio-political unrest in Sidama (SNNPR), North Gondar Wag Hamra Central Gondar and a rise in security incidents in Southwest- Zone 4 (Fantana Rasu) AFAR ern Oromia and Gambella. The quality of ac- Zone 1 (Awsi Rasu) cess declined, limiting assistance to people AMHARA No. o incidents by one South Wello Metekel in need, against a backdrop of massive gov- Oromia East Gojam BENISHANGUL Zone 5 (Hari Rasu) 4 13 35 49 AsosaGUMUZ Siti ernment-led returns of IDP to areas of origin. Zone 3 (Gabi Rasu) North Shewa(O) North Shewa(A) Kemashi Dire Dawa urban West Wellega East Wellega DIRE DAWA West Shewa Fafan • Hostilities between Ethiopian Defense Forc- ADDIS ABABA Kelem Wellega East Hararge Finfine Special West Hararge es (EDF) and Unidentified Armed Groups Buno Bedele East Shewa Etang Special Ilu Aba Bora Jarar OROMIA Erer (UAGs) as well as inter-ethnic, remained the GAMBELA Jimma Agnewak main access obstacle, with 197 incidents Doolo Nogob West Arsi SOMALI (out of 276), mostly in Southwestern Oromia SNNP Sidama Bale Korahe (110). The Wellegas, West Guji (Oromia), and Gedeo Shabelle Gambella, were the most insecure areas for Segen Area P. West Guji Guji aid workers. Liban Borena • In June, conflict in the Wellegas scaled up, Daawa with explosive devices attacks causing ci- Source: Access Incidents database vilian casualties in urban centres. -

Addis Ababa University, College of Health Sciences
Addis Ababa University, College of Health Sciences, School of Public Health Ethiopia Field Epidemiology Training Program (EFETP) Compiled Body of Works in Field Epidemiology By Elsabetee Megrssa Submitted to the School of Graduate Studies of Addis Ababa University in partial fulfillment for the degree of Master of Public Health in Field Epidemiology May 2014 Addis Ababa Addis Ababa University College of Health Sciences School of Public Health Ethiopia Field Epidemiology Training Program (EFETP) Compiled Body of Works in Field Epidemiology By Elsabetee Megrssa Submitted to the School of Graduate Studies of Addis Ababa University in partial fulfillment for the degree of Master of Public Health in Field Epidemiology Advisors Dr.Ayele Belachew Dr. John Fogarty May 2014 Addis Ababa ii Addis Ababa University School of Graduate Studies Compiled Body of Works in Field Epidemiology By Elsabetee Megrssa Ethiopia Field Epidemiology Training Program (EFETP) School of Public Health, College of Health Sciences Addis Ababa University Approval by Examining Board _________________________ ___________________ Chairman, School Graduate Committee _________________________ ___________________ Advisor _________________________ ___________________ Examiner _________________________ ___________________ Examiner iii Acknowledgments First of all I would like to thank my mentors Dr Ayele Belachew and Dr.John Fogarty for their guidance, support and inputs in my work. Secondly, I would like to thank Dr. Lucy Boulanger, Dr.Getahun Aynalem, Dr.Desalegn Dalecha, Dr. Adamu Addis, Dr. Zegeye Hailemariam, Ms. Abigail Greenleaf, Mr. Alemayehu Bekele and Mr. Abyot Bekele for their constructive comments and contribution during field base supervisions. Third, I would like to thank Dr. Daddi Jimma; he is a deputy director general of Ethiopian Public health Institute and all PHEM staff for their cooperation and contribution during my study period. -

Ethiopia: Administrative Map (August 2017)
Ethiopia: Administrative map (August 2017) ERITREA National capital P Erob Tahtay Adiyabo Regional capital Gulomekeda Laelay Adiyabo Mereb Leke Ahferom Red Sea Humera Adigrat ! ! Dalul ! Adwa Ganta Afeshum Aksum Saesie Tsaedaemba Shire Indasilase ! Zonal Capital ! North West TigrayTahtay KoraroTahtay Maychew Eastern Tigray Kafta Humera Laelay Maychew Werei Leke TIGRAY Asgede Tsimbila Central Tigray Hawzen Medebay Zana Koneba Naeder Adet Berahile Region boundary Atsbi Wenberta Western Tigray Kelete Awelallo Welkait Kola Temben Tselemti Degua Temben Mekele Zone boundary Tanqua Abergele P Zone 2 (Kilbet Rasu) Tsegede Tselemt Mekele Town Special Enderta Afdera Addi Arekay South East Ab Ala Tsegede Mirab Armacho Beyeda Woreda boundary Debark Erebti SUDAN Hintalo Wejirat Saharti Samre Tach Armacho Abergele Sanja ! Dabat Janamora Megale Bidu Alaje Sahla Addis Ababa Ziquala Maychew ! Wegera Metema Lay Armacho Wag Himra Endamehoni Raya Azebo North Gondar Gonder ! Sekota Teru Afar Chilga Southern Tigray Gonder City Adm. Yalo East Belesa Ofla West Belesa Kurri Dehana Dembia Gonder Zuria Alamata Gaz Gibla Zone 4 (Fantana Rasu ) Elidar Amhara Gelegu Quara ! Takusa Ebenat Gulina Bugna Awra Libo Kemkem Kobo Gidan Lasta Benishangul Gumuz North Wello AFAR Alfa Zone 1(Awsi Rasu) Debre Tabor Ewa ! Fogera Farta Lay Gayint Semera Meket Guba Lafto DPubti DJIBOUTI Jawi South Gondar Dire Dawa Semen Achefer East Esite Chifra Bahir Dar Wadla Delanta Habru Asayita P Tach Gayint ! Bahir Dar City Adm. Aysaita Guba AMHARA Dera Ambasel Debub Achefer Bahirdar Zuria Dawunt Worebabu Gambela Dangura West Esite Gulf of Aden Mecha Adaa'r Mile Pawe Special Simada Thehulederie Kutaber Dangila Yilmana Densa Afambo Mekdela Tenta Awi Dessie Bati Hulet Ej Enese ! Hareri Sayint Dessie City Adm. -

School of Social Sciences Indira Gandhi National Open University
IGNOU UNIVERSITY DEPARTMENT OF PUBLIC ADMINISTRATION School of Social Sciences Indira Gandhi National Open University IMPACT AND ASSOSIATED FACTORS OF CONFLICT OVER RESOURCE AND LIVELIHOOD IN PASTORALIST COMMUNITY: THE CASE OF AFAR - ISSA CONFLICT IN AMIBARA WOREDA, AFAR REGIONAL STATE By: Shimles Abebaw(BA) ID No. ID1322083 A THESISDIRAFT REPORT SUBMITTED TO IGNOU UNIVERSITY, DEPARTMENT OF PUBLIC ADMINISTRATION IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF MASTER OF ART Advisor: Dr. Elias Berhanu Debela April, 2016 Addis Ababa Oct, 2015 AddisAbeba, DECLARATION I hereby Declare that the DISSERTATION ENTITLED IMPACT AND ASSOCIATED FACTORS OF CONFLICT OVER RESOURCE AND LIVELIHOOD IN PASTORALIST COMMUNITY: THE CASE OF AFAR - ISSA CONFLICT IN AMIBARA WOREDA, AFAR REGIONAL STATE Submitted by me for the partial fulfillment the MPA to Indira Gandhi National Open University ( IGNOU) New Delhi is my own original work and has not been summated earlier, either to IGNOUS or to any other institution for the fulfillment of the requirement for any other programmer of study. I also declare that no chapter of this manuscript in whole or in part is lifted and incorporated in this report from any earlier work done by me or others. Place:- Addis Ababa, Ethiopia Signature --------------------------- Date:- April,2016 Enrolment No: ID1322083 Name: Shimelis Abebaw Shiferaw Address :- Addis Ababa, Ethiopia i CERTIFICATE I certified that the Dissertation entitled IMPACT AND ASSOSIATED FACTORS OF CONFLICT OVER RESOURCE AND LIVELIHOOD IN PASTORALIST COMMUNITY: THE CASE OF AFAR - ISSA CONFLICT IN AMIBARA WOREDA, AFAR REGIONAL STATE submitted by Shimelis Ababaw is his own work and has been done under my supervision It is recommended that this dissertation be placed before the examiner of evaluation. -

Enabling Successful Migration for Youth in Addis Ababa Quantitative Survey
Enabling Successful Migration for Youth in Addis Ababa Quantitative Survey Z1 Respondent Code Sequential number according to order of the respondent (101, 102, 103, etc.) Z2 Interviewer Code Z3 Date of Interview (DD/MM/YY) / / 1 8 Z4 Location Code Based on site Z5 Start time of Interview (according to Western clock) : CONSENT: Did you complete the entire verbal script for consent, answer all questions from the respondent, and and receive a fully informed consent to participate? Investigator name: Investigator Signature: Are you willing to go ahead? Yes → continue to A1 No → stop the survey, thank the respondent, and approach another potential respondent 1 I. DEMOGRAPHIC CHARACTERISTICS A1 How old are you? Enter age A2 Sex 0 = male 1 = female 8 = don’t know 9 = refused to answer A3 Where in Ethiopia are you from? Enter code for region Enter code for zone Enter name of woreda Enter name of kebele A4 Where in Addis Ababa do you live? Enter code for subcity Enter name of district A5 Which year did you first arrive in Enter year (according to Western Addis? calendar, not Ethiopian) A6 How much time (cumulative) have Enter total number of months you spent in Addis Ababa? A7 What is your marital status? 1 = married 2 = engaged, 3 = in a partnership 4 = single 5 = widowed 6 = divorced/separated 8 = don’t know 9 = refused to answer A8 Do you have any children? 0 = no 1 = yes 8 = don’t know 9 = refused to answer A9 What is your religion? 1 = Ethiopian Orthodox 2 = Muslim 3 = Protestant 4 = Traditional/local 5 = Catholic 6 = Other 8 = Don’t know -

Environmental Associated Emotional Distress and the Dangers of Climate
Global Environmental Change 59 (2019) 101994 Contents lists available at ScienceDirect Global Environmental Change journal homepage: www.elsevier.com/locate/gloenvcha Environmental associated emotional distress and the dangers of climate change for pastoralist mental health T ⁎ Sarah Coopera, Paul Hutchingsa, , John Butterworthb, Solome Josepha,b,c,d,e, Abinet Kebedeb, Alison Parkera, Bethel Terefea,b,c,d,e, Barbara Van Koppena,b,c,d,e a Cranfield Water Science Institute, Cranfield University, Vincent Building, Cranfield, Bedfordshire MK43 0AL, UK b IRC Ethiopia, Golagul Towers Building, Bole sub city, Woreda 4 H.No. 275/276, Addis Ababa, Ethiopia c Friendship Support Association, Nefas Selek Lafto Sub-city, Woreda 02, Mekanisa S. Sara building, P.O.Box 22068, 1000 Addis Ababa, Ethiopia d Oxfam International – Ethiopia, Bole Megenagna ring road, Addis Ababa, Ethiopia e International Water Management Institute (IWMI), 127, Sunil Mawatha, Pelawatte, Battaramulla, Sri Lanka ARTICLE INFO ABSTRACT Keywords: Pastoralists in the Horn of Africa are among the most vulnerable populations to climate change yet little is Environmental change known about how environmental change shapes their wellbeing and mental health. This paper presents a for- Wellbeing mative study into the relations between emotion, wellbeing and water security among pastoralist communities in Mental health Afar, Ethiopia. It uses focus group and interview data to demonstrate the close relationship between environ- Pastoralists mental conditions and emotional wellbeing, and shows how current water insecurity leads to extreme worry and Ethiopia fatigue among the studied population, especially in the dry season. In the context of difficulties of translating mental health clinical classifications and diagnostic tools in cross-cultural settings, the paper argues the in- ductive study of emotion may be a useful approach for studying environmental determined wellbeing outcomes among marginal populations in the light of understanding climate change impacts. -

Ethiopia: Humanitarian Access Situation Report
ETHIOPIA Humanitarian Access Situation Report January - March 2020 This report is produced by OCHA Ethiopia in collaboration with humanitarian partners. It covers the period January to March 2020. The next report will be issued in June 2020. OVERVIEW • The operational environment to relief operations North Number of incidents by woreda Western Central remained permissive through the reporting period. Western TIGRAY Eastern 1 - 2 3 - 4 5 - 6 South Kilbet Most access impediments continue related to hos- North Rasu Gondar Eastern Wag tilities, intra-community conflicts or social unrest, Central Southern Gondar Hamra West Fantana hindering the quality of the humanitarian response, Gondar AMHARA North Wello Rasu AFAR and to COVID-19. South Awsi Gondar Rasu Metekel Hari Awi West East South Wello Gojam Rasu • Humanitarian partners are committed to support BENISHANGUL Gojam Oromia GUMUZ Siti the government response to COVID-19 and ensure North North Gabi Kemashi Horo Shewa DIRE DAWA West Shewa Rasu that critical activities are sustained. Partners are Gudru West Mao Komo Wellega Wellega Shewa Fafan Special East Addis Ababa actively implementing precautionary measures to Wellega HARARI Kelem Wellega East South West West ensure the safety of aid personnel and the popula- Buno Bedele East Hararge Hararge Ilu Aba Shewa Shewa Guraghe GAMBELA Bora Jarar tion. Nuwer Arsi Erer Agnewak Jimma Hadiya Siltie Sheka Yem Sp.Halaba Sp. OROMIA Kembata Mejenger Kefa Doolo Dawuro Tibaro Nogob SOMALI • The humanitarian community is working with gov- Bench Maji West Arsi Konta Sp. Wolayita Bale Gofa Sidama ernment counterparts to ensure that partners can Gamo Korahe Mirab Basketo Gedeo continue movements and operations throughout Omo Amaro SNNP Derashe Alle Guji Shabelle the country, bearing in mind restrictions to contain South Omo BurjiWest Guji Konso Afder the spread of COVID-19. -

Ethiopia Access Snapshot - Afar Region and Siti Zone, Somali Region As of 31 January 2020
Ethiopia Access Snapshot - Afar region and Siti zone, Somali region As of 31 January 2020 Afar region is highly prone to natural disasters Afdera The operating environment is highly compromised, with a high such as droughts and seasonal flooding. Long-- risk for humanitarian operations of becoming politicized. In ErebtiDalol Zone 2 term historical grievances coupled with Bidu March 2019, four aid workers were detained by Afar authorities TIGRAY resource-based tensions between ethnic Afar for having allegedly entered the region illegally. They were KunnebaBerahile and its neighbors i.e. Issa (Somali), and Oromo Megale conducting a humanitarian activity in Sitti zone, and decided to Teru Ittu (Amibara woreda) and Karayu (Awash Fentale woreda) in ERITREA overnight in a village of Undufo kebele. In a separate incident, in Yalo AFAR Kurri Red Sea October 2019, an attack by unidentified armed men in Afambo zone 3, and in areas adjacent to Oromia special zone and Amhara- Afdera Robe Town Aso s a Ethnic Somali IDPZone 2016/2018 4 Zone 2 (Kilbet Rasu) Elidar region, continue to cause casualties and forced displacement, Aba 'Ala woreda, Zone 1, near Djibouti, killed a number of civilians spark- Gulina Goba Town limiting partners’ movements and operations. Overall, Ethnican Oromia IDP 2016/2018 L. Afrera Ye'ch'ew ing outrage across the region and prompting peaceful demon- Awra estimated 50,000 people remain displaced, the majority of whom Erebti strations and temporarily road blockages of the Awash highway SNNP Zone 1 Bidu rely almost entirely on assistance provided by host communities. Semera TIGRAYEwa On the other hand, in 2019, the overflow of Awash River and DJIBOUTI Clashes involving Afar and Somali Issa clan continue along Megale Afele Kola flash floods displaced some 3,300 households across six Dubti boundary areas between Afar’s zone 1 and 3 and Sitti zone. -

ETHIOPIA: AGRICULTURE SECTOR PARTNERS PRESENCE (November 2018) Agriculture Partners Operational Presence ERITREA
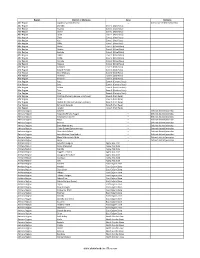
ETHIOPIA: AGRICULTURE SECTOR PARTNERS PRESENCE (November 2018) Agriculture Partners Operational Presence ERITREA Asgede Ts mbila Tigray Tselemt Tach Armacho Sahla SUDAN Dehana Gaz Afar Bugna Gibla Amhara Meket DJIBOUTI Simada Beneshangul Gewane Bure Homosha Gumu Mudaytu Dire Argoba Chinaksen Amibara Special Goro Dawa Gutu SOMALIA Awash Mieso HareGrurisum Addis Fentale Bedeno Fedis Midega Boset SOUTH SUDAN Ababa Daro Tola Aware Sire Lebu Dodota Hawi Gashamo Adami Tulu Gudina Jido Gambela Kombolcha Shashogo Shashemene Kacha Zuria Bira Siraro Oromia Damot Goro Somali Boricha Kebridehar Sore SNNPR Gode Debeweyin Shilabo Amaro Adadle Mustahil Ferfer Bena Dugda Tsemay Chereti/Weyib Gnangatom Dawa Legend Hamer Dasenech International Boundary (Kuraz) Moyale Regional Boundary Dolo Odo Dire Dehas Moyale_Oro No.of Partners by Woreda Miyo 1 - 2 3 - 4 KENYA Creation date: 20 November 2018 Feedback: Espico Iga (Denis), Information Management Officer: [email protected] / Farshad Tami, Sector Coordinator: [email protected] / https://www.humanitarianresponse.info/en/operations/ethiopia/agriculture-livestock Sources: 1. Revised as of November 2018. | 2. Response target figures are as of 31 October 2018 and are compiled on the basis of information submitted by Agriculture Sector partners on monthly basis. 3. Funding data is collected from partners. Region Zone Woreda Organiza�on Donor Afar Zone 3 (Gabi Rasu ) Argoba Special CARE USAID Afar Zone 3 (Gabi Rasu ) Argoba Special CARE USAID Afar Zone 3 (Gabi Rasu ) Amibara CARE USAID Afar Zone 3 (Gabi Rasu