Heparanase Overexpression Induces Glucagon Resistance and Protects
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Activation of the Glucagon-Like Peptide-1 (GLP-1) Receptor by Peptide and Non-Peptide Ligands
The Activation of the Glucagon-Like Peptide-1 (GLP-1) Receptor by Peptide and Non-Peptide Ligands Clare Louise Wishart Submitted in accordance with the requirements for the degree of Doctor of Philosophy of Science University of Leeds School of Biomedical Sciences Faculty of Biological Sciences September 2013 I Intellectual Property and Publication Statements The candidate confirms that the work submitted is her own and that appropriate credit has been given where reference has been made to the work of others. This copy has been supplied on the understanding that it is copyright material and that no quotation from the thesis may be published without proper acknowledgement. The right of Clare Louise Wishart to be identified as Author of this work has been asserted by her in accordance with the Copyright, Designs and Patents Act 1988. © 2013 The University of Leeds and Clare Louise Wishart. II Acknowledgments Firstly I would like to offer my sincerest thanks and gratitude to my supervisor, Dr. Dan Donnelly, who has been nothing but encouraging and engaging from day one. I have thoroughly enjoyed every moment of working alongside him and learning from his guidance and wisdom. My thanks go to my academic assessor Professor Paul Milner whom I have known for several years, and during my time at the University of Leeds he has offered me invaluable advice and inspiration. Additionally I would like to thank my academic project advisor Dr. Michael Harrison for his friendship, help and advice. I would like to thank Dr. Rosalind Mann and Dr. Elsayed Nasr for welcoming me into the lab as a new PhD student and sharing their experimental techniques with me, these techniques have helped me no end in my time as a research student. -

Table S1. List of Proteins in the BAHD1 Interactome
Table S1. List of proteins in the BAHD1 interactome BAHD1 nuclear partners found in this work yeast two-hybrid screen Name Description Function Reference (a) Chromatin adapters HP1α (CBX5) chromobox homolog 5 (HP1 alpha) Binds histone H3 methylated on lysine 9 and chromatin-associated proteins (20-23) HP1β (CBX1) chromobox homolog 1 (HP1 beta) Binds histone H3 methylated on lysine 9 and chromatin-associated proteins HP1γ (CBX3) chromobox homolog 3 (HP1 gamma) Binds histone H3 methylated on lysine 9 and chromatin-associated proteins MBD1 methyl-CpG binding domain protein 1 Binds methylated CpG dinucleotide and chromatin-associated proteins (22, 24-26) Chromatin modification enzymes CHD1 chromodomain helicase DNA binding protein 1 ATP-dependent chromatin remodeling activity (27-28) HDAC5 histone deacetylase 5 Histone deacetylase activity (23,29,30) SETDB1 (ESET;KMT1E) SET domain, bifurcated 1 Histone-lysine N-methyltransferase activity (31-34) Transcription factors GTF3C2 general transcription factor IIIC, polypeptide 2, beta 110kDa Required for RNA polymerase III-mediated transcription HEYL (Hey3) hairy/enhancer-of-split related with YRPW motif-like DNA-binding transcription factor with basic helix-loop-helix domain (35) KLF10 (TIEG1) Kruppel-like factor 10 DNA-binding transcription factor with C2H2 zinc finger domain (36) NR2F1 (COUP-TFI) nuclear receptor subfamily 2, group F, member 1 DNA-binding transcription factor with C4 type zinc finger domain (ligand-regulated) (36) PEG3 paternally expressed 3 DNA-binding transcription factor with -
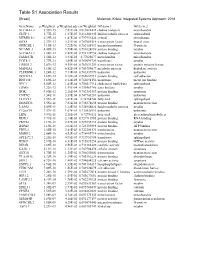
Table S1 Association Results
GeneName p.Weighted p.Weighted.adj cor.Weighted GO.term.1 GO.term.2 SLC44A1.2 3.55E-15 7.81E-08 0.819822422 choline transport mitochondria GLTP.1 5.77E-15 1.27E-07 0.816302445 lipid metabolic process sphingolipid MTMR10.1 6.39E-14 1.41E-06 0.797951424 cytosol phosphatase SOX8 2.37E-13 5.21E-06 0.787085514 transcription factor neural crest GPRC5B.1 4.19E-13 9.22E-06 0.782154937 integral membrane G-protein NCAM1.3 4.45E-13 9.79E-06 0.781624896 protein binding myelin SLC44A1.1 1.28E-12 2.82E-05 0.772132954 choline transport mitochondria FAM107B 1.56E-12 3.43E-05 0.77025677 mitochondria mitochondria UGT8.1 1.77E-12 3.89E-05 0.769099729 transferase myelin ERBB3.2 2.07E-12 4.55E-05 0.767631259 transcription factor protein tyrosine kinase MAN2A1 3.10E-12 6.82E-05 0.763759677 metabolic process hydrolase activity PLEKHH1.1 3.24E-12 7.13E-05 0.763337879 unknown unknown DOCK5.3 3.69E-12 8.12E-05 0.762087913 protein binding cell adhesion RNF130 3.69E-12 8.12E-05 0.762094156 membrane metal ion binding NPC1 6.50E-12 1.43E-04 0.756517114 cholesterol trafficking sphingolipid ERMN 7.22E-12 1.59E-04 0.755469786 actin binding myelin BOK 9.80E-12 2.16E-04 0.752383357 protein binding apoptosis CNTN2 1.54E-11 3.39E-04 0.747743281 unknown unknown ELOVL1 1.55E-11 3.41E-04 0.74764744 fatty acid sphingolipid DBNDD2 3.55E-11 7.81E-04 0.738878658 protein binding neuron projection LASS2 5.09E-11 1.12E-03 0.734954024 lipid metabolic process myelin C12orf34 7.57E-11 1.67E-03 0.730528911 unknown unknown LIPA 9.59E-11 2.11E-03 0.72786111 fatty acid glycerolipid metabolic -

The Title of the Dissertation
UNIVERSITY OF CALIFORNIA SAN DIEGO Novel network-based integrated analyses of multi-omics data reveal new insights into CD8+ T cell differentiation and mouse embryogenesis A dissertation submitted in partial satisfaction of the requirements for the degree Doctor of Philosophy in Bioinformatics and Systems Biology by Kai Zhang Committee in charge: Professor Wei Wang, Chair Professor Pavel Arkadjevich Pevzner, Co-Chair Professor Vineet Bafna Professor Cornelis Murre Professor Bing Ren 2018 Copyright Kai Zhang, 2018 All rights reserved. The dissertation of Kai Zhang is approved, and it is accept- able in quality and form for publication on microfilm and electronically: Co-Chair Chair University of California San Diego 2018 iii EPIGRAPH The only true wisdom is in knowing you know nothing. —Socrates iv TABLE OF CONTENTS Signature Page ....................................... iii Epigraph ........................................... iv Table of Contents ...................................... v List of Figures ........................................ viii List of Tables ........................................ ix Acknowledgements ..................................... x Vita ............................................. xi Abstract of the Dissertation ................................. xii Chapter 1 General introduction ............................ 1 1.1 The applications of graph theory in bioinformatics ......... 1 1.2 Leveraging graphs to conduct integrated analyses .......... 4 1.3 References .............................. 6 Chapter 2 Systematic -

SOX4-Mediated Repression of Specific Trnas Inhibits Proliferation of Human Glioblastoma Cells
SOX4-mediated repression of specific tRNAs inhibits proliferation of human glioblastoma cells Jianjing Yanga,b,c, Derek K. Smithc,d, Haoqi Nia,b,c,KeWua,b, Dongdong Huanga,b, Sishi Pana,b,c, Adwait A. Sathee, Yu Tangc,d, Meng-Lu Liuc,d, Chao Xinge,f,g, Chun-Li Zhangc,d,1, and Qichuan Zhugea,b,1 aDepartment of Neurosurgery, The First Affiliated Hospital of Wenzhou Medical University, Wenzhou, Zhejiang, China 325000; bZhejiang Provincial Key Laboratory of Aging and Neurological Disorder Research, The First Affiliated Hospital of Wenzhou Medical University, Wenzhou, Zhejiang, China 325000; cDepartment of Molecular Biology, University of Texas Southwestern Medical Center, Dallas, TX 75390; dHamon Center for Regenerative Science and Medicine, University of Texas Southwestern Medical Center, Dallas, TX 75390; eMcDermott Center of Human Growth and Development, University of Texas Southwestern Medical Center, Dallas, TX 75390; fDepartment of Bioinformatics, University of Texas Southwestern Medical Center, Dallas, TX 75390; and gDepartment of Population and Data Sciences, University of Texas Southwestern Medical Center, Dallas, TX 75390 Edited by S. Altman, Yale University, New Haven, CT, and approved February 5, 2020 (received for review November 15, 2019) Transfer RNAs (tRNAs) are products of RNA polymerase III (Pol III) indicates that tRNA expression may also be under cell state- and essential for mRNA translation and ultimately cell growth and dependent regulations (12–16). proliferation. Whether and how individual tRNA genes are specif- In this study, we performed a systematic analysis on how ically regulated is not clear. Here, we report that SOX4, a well- NGN2/SOX4-mediated cell-fate reprogramming leads to cell known Pol II-dependent transcription factor that is critical for neuro- cycle exit of human glioblastoma cells. -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated. -

Role of Phospholipases in Adrenal Steroidogenesis
229 1 W B BOLLAG Phospholipases in adrenal 229:1 R29–R41 Review steroidogenesis Role of phospholipases in adrenal steroidogenesis Wendy B Bollag Correspondence should be addressed Charlie Norwood VA Medical Center, One Freedom Way, Augusta, GA, USA to W B Bollag Department of Physiology, Medical College of Georgia, Augusta University (formerly Georgia Regents Email University), Augusta, GA, USA [email protected] Abstract Phospholipases are lipid-metabolizing enzymes that hydrolyze phospholipids. In some Key Words cases, their activity results in remodeling of lipids and/or allows the synthesis of other f adrenal cortex lipids. In other cases, however, and of interest to the topic of adrenal steroidogenesis, f angiotensin phospholipases produce second messengers that modify the function of a cell. In this f intracellular signaling review, the enzymatic reactions, products, and effectors of three phospholipases, f phospholipids phospholipase C, phospholipase D, and phospholipase A2, are discussed. Although f signal transduction much data have been obtained concerning the role of phospholipases C and D in regulating adrenal steroid hormone production, there are still many gaps in our knowledge. Furthermore, little is known about the involvement of phospholipase A2, Endocrinology perhaps, in part, because this enzyme comprises a large family of related enzymes of that are differentially regulated and with different functions. This review presents the evidence supporting the role of each of these phospholipases in steroidogenesis in the Journal Journal of Endocrinology adrenal cortex. (2016) 229, R1–R13 Introduction associated GTP-binding protein exchanges a bound GDP for a GTP. The G protein with GTP bound can then Phospholipids serve a structural function in the cell in that activate the enzyme, phospholipase C (PLC), that cleaves they form the lipid bilayer that maintains cell integrity. -

A Rapid Transcriptome Response Is Associated with Desiccation Resistance in Aerially-Exposed Killifish Embryos
A Rapid Transcriptome Response Is Associated with Desiccation Resistance in Aerially-Exposed Killifish Embryos Ange`le Tingaud-Sequeira1¤a, Juan-Jose´ Lozano2, Cinta Zapater1, David Otero1, Michael Kube3¤b, Richard Reinhardt3¤c, Joan Cerda` 1* 1 Institut de Recerca i Tecnologia Agroalimenta`ries (IRTA)-Institut de Cie`ncies del Mar, CSIC, Barcelona, Spain, 2 Bioinformatics Platform, CIBERHED, Barcelona, Spain, 3 Max Planck Institute for Molecular Genetics, Berlin-Dahlem, Germany Abstract Delayed hatching is a form of dormancy evolved in some amphibian and fish embryos to cope with environmental conditions transiently hostile to the survival of hatchlings or larvae. While diapause and cryptobiosis have been extensively studied in several animals, very little is known concerning the molecular mechanisms involved in the sensing and response of fish embryos to environmental cues. Embryos of the euryhaline killifish Fundulus heteroclitus advance dvelopment when exposed to air but hatching is suspended until flooding with seawater. Here, we investigated how transcriptome regulation underpins this adaptive response by examining changes in gene expression profiles of aerially incubated killifish embryos at ,100% relative humidity, compared to embryos continuously flooded in water. The results confirm that mid-gastrula embryos are able to stimulate development in response to aerial incubation, which is accompanied by the differential expression of at least 806 distinct genes during a 24 h period. Most of these genes (,70%) appear to be differentially expressed within 3 h of aerial exposure, suggesting a broad and rapid transcriptomic response. This response seems to include an early sensing phase, which overlaps with a tissue remodeling and activation of embryonic development phase involving many regulatory and metabolic pathways. -

An Automated Pipeline for Inferring Variant-Driven Gene
www.nature.com/scientificreports OPEN MAGPEL: an autoMated pipeline for inferring vAriant‑driven Gene PanEls from the full‑length biomedical literature Nafseh Saberian1, Adib Shaf 1, Azam Peyvandipour1 & Sorin Draghici 1,2* In spite of the eforts in developing and maintaining accurate variant databases, a large number of disease‑associated variants are still hidden in the biomedical literature. Curation of the biomedical literature in an efort to extract this information is a challenging task due to: (i) the complexity of natural language processing, (ii) inconsistent use of standard recommendations for variant description, and (iii) the lack of clarity and consistency in describing the variant-genotype-phenotype associations in the biomedical literature. In this article, we employ text mining and word cloud analysis techniques to address these challenges. The proposed framework extracts the variant- gene‑disease associations from the full‑length biomedical literature and designs an evidence‑based variant-driven gene panel for a given condition. We validate the identifed genes by showing their diagnostic abilities to predict the patients’ clinical outcome on several independent validation cohorts. As representative examples, we present our results for acute myeloid leukemia (AML), breast cancer and prostate cancer. We compare these panels with other variant‑driven gene panels obtained from Clinvar, Mastermind and others from literature, as well as with a panel identifed with a classical diferentially expressed genes (DEGs) approach. The results show that the panels obtained by the proposed framework yield better results than the other gene panels currently available in the literature. One crucial step in understanding the biological mechanism underlying a disease condition is to capture the relationship between the variants and the disease risk1. -

9. Atypical Dusps: 19 Phosphatases in Search of a Role
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Digital.CSIC Transworld Research Network 37/661 (2), Fort P.O. Trivandrum-695 023 Kerala, India Emerging Signaling Pathways in Tumor Biology, 2010: 185-208 ISBN: 978-81-7895-477-6 Editor: Pedro A. Lazo 9. Atypical DUSPs: 19 phosphatases in search of a role Yolanda Bayón and Andrés Alonso Instituto de Biología y Genética Molecular, CSIC-Universidad de Valladolid c/ Sanz y Forés s/n, 47003 Valladolid, Spain Abstract. Atypical Dual Specificity Phosphatases (A-DUSPs) are a group of 19 phosphatases poorly characterized. They are included among the Class I Cys-based PTPs and contain the active site motif HCXXGXXR conserved in the Class I PTPs. These enzymes present a phosphatase domain similar to MKPs, but lack any substrate targeting domain similar to the CH2 present in this group. Although most of these phosphatases have no more than 250 amino acids, their size ranges from the 150 residues of the smallest A-DUSP, VHZ/DUSP23, to the 1158 residues of the putative PTP DUSP27. The substrates of this family include MAPK, but, in general terms, it does not look that MAPK are the general substrates for the whole group. In fact, other substrates have been described for some of these phosphatases, like the 5’CAP structure of mRNA, glycogen, or STATs and still the substrates of many A-DUSPs have not been identified. In addition to the PTP domain, most of these enzymes present no additional recognizable domains in their sequence, with the exception of CBM-20 in laforin, GTase in HCE1 and a Zn binding domain in DUSP12. -

Supplementary Table S4. FGA Co-Expressed Gene List in LUAD
Supplementary Table S4. FGA co-expressed gene list in LUAD tumors Symbol R Locus Description FGG 0.919 4q28 fibrinogen gamma chain FGL1 0.635 8p22 fibrinogen-like 1 SLC7A2 0.536 8p22 solute carrier family 7 (cationic amino acid transporter, y+ system), member 2 DUSP4 0.521 8p12-p11 dual specificity phosphatase 4 HAL 0.51 12q22-q24.1histidine ammonia-lyase PDE4D 0.499 5q12 phosphodiesterase 4D, cAMP-specific FURIN 0.497 15q26.1 furin (paired basic amino acid cleaving enzyme) CPS1 0.49 2q35 carbamoyl-phosphate synthase 1, mitochondrial TESC 0.478 12q24.22 tescalcin INHA 0.465 2q35 inhibin, alpha S100P 0.461 4p16 S100 calcium binding protein P VPS37A 0.447 8p22 vacuolar protein sorting 37 homolog A (S. cerevisiae) SLC16A14 0.447 2q36.3 solute carrier family 16, member 14 PPARGC1A 0.443 4p15.1 peroxisome proliferator-activated receptor gamma, coactivator 1 alpha SIK1 0.435 21q22.3 salt-inducible kinase 1 IRS2 0.434 13q34 insulin receptor substrate 2 RND1 0.433 12q12 Rho family GTPase 1 HGD 0.433 3q13.33 homogentisate 1,2-dioxygenase PTP4A1 0.432 6q12 protein tyrosine phosphatase type IVA, member 1 C8orf4 0.428 8p11.2 chromosome 8 open reading frame 4 DDC 0.427 7p12.2 dopa decarboxylase (aromatic L-amino acid decarboxylase) TACC2 0.427 10q26 transforming, acidic coiled-coil containing protein 2 MUC13 0.422 3q21.2 mucin 13, cell surface associated C5 0.412 9q33-q34 complement component 5 NR4A2 0.412 2q22-q23 nuclear receptor subfamily 4, group A, member 2 EYS 0.411 6q12 eyes shut homolog (Drosophila) GPX2 0.406 14q24.1 glutathione peroxidase -

Supplemental Information For
Supplemental Information for: Gene Expression Profiling of Pediatric Acute Myelogenous Leukemia Mary E. Ross, Rami Mahfouz, Mihaela Onciu, Hsi-Che Liu, Xiaodong Zhou, Guangchun Song, Sheila A. Shurtleff, Stanley Pounds, Cheng Cheng, Jing Ma, Raul C. Ribeiro, Jeffrey E. Rubnitz, Kevin Girtman, W. Kent Williams, Susana C. Raimondi, Der-Cherng Liang, Lee-Yung Shih, Ching-Hon Pui & James R. Downing Table of Contents Section I. Patient Datasets Table S1. Diagnostic AML characteristics Table S2. Cytogenetics Summary Table S3. Adult diagnostic AML characteristics Table S4. Additional T-ALL characteristics Section II. Methods Table S5. Summary of filtered probe sets Table S6. MLL-PTD primers Additional Statistical Methods Section III. Genetic Subtype Discriminating Genes Figure S1. Unsupervised Heirarchical clustering Figure S2. Heirarchical clustering with class discriminating genes Table S7. Top 100 probe sets selected by SAM for t(8;21)[AML1-ETO] Table S8. Top 100 probe sets selected by SAM for t(15;17) [PML-RARα] Table S9. Top 63 probe sets selected by SAM for inv(16) [CBFβ-MYH11] Table S10. Top 100 probe sets selected by SAM for MLL chimeric fusion genes Table S11. Top 100 probe sets selected by SAM for FAB-M7 Table S12. Top 100 probe sets selected by SAM for CBF leukemias (whole dataset) Section IV. MLL in combined ALL and AML dataset Table S13. Top 100 probe sets selected by SAM for MLL chimeric fusions irrespective of blast lineage (whole dataset) Table S14. Class discriminating genes for cases with an MLL chimeric fusion gene that show uniform high expression, irrespective of blast lineage Section V.