How Are Endemic and Widely Distributed Bromeliads Responding To
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

General Information Bromeliaceae Family
General Information Bromeliads are a unique and fascinating family of hundreds of extremely diversified and exotic plants, which are amazingly adaptable, tough and relatively easy to grow. People often say that Bromeliads thrive on neglect. The species can tolerate a huge variety of growing conditions including heat, light, air and moisture. No Bromeliads are native to Australia and therefore have all been imported and introduced here. The plants are native to the Southern States of the USA, Central America and deep into South America, with regions like Florida, Mexico, the West Indies, parts of Brazil and as far south as Chile having many and various species. One very primitive species is also found in Africa and has survived since the two continents separated. Bromeliaceae Family The entire bromeliad family called Bromeliaceae, is divided into three subfamilies containing many genera, with the Bromelioideae and Tillandsioideae subfamilies being the most popular bromeliads for enthusiasts and collectors. The subfamily Bromelioideae is distributed from Mexico to Argentina and has the greatest number of genera. They are mostly epiphytic, tank-type plants with spiny leaves and berry-like fruit containing wet seeds. The subfamily Pitcairnioideae are the most primitive bromeliads, descended from the grass family. Nearly all are terrestrial. Most have spiny leaves. The seeds are dry and usually winged. The subfamily Tillandsioideae has few genera, but includes about half of the species of bromeliads. Growing throughout the Americas, they are mostly epiphytes. All have spineless leaves. Seeds are dry, with feathery "parachutes" and are blown and float in the wind. The most notable and commercially developed of the family is the edible pineapple (Ananus comosus). -

Functional Structure of the Bromeliad Tank Microbiome Is Strongly Shaped by Local Geochemical Conditions
Environmental Microbiology (2017) 19(8), 3132–3151 doi:10.1111/1462-2920.13788 Functional structure of the bromeliad tank microbiome is strongly shaped by local geochemical conditions Stilianos Louca,1,2* Saulo M. S. Jacques,3,4 denitrification steps, ammonification, sulfate respira- Aliny P. F. Pires,3 Juliana S. Leal,3,5 tion, methanogenesis, reductive acetogenesis and 6 1,2,7 Angelica L. Gonzalez, Michael Doebeli and anoxygenic phototrophy. Overall, CO2 reducers domi- Vinicius F. Farjalla3 nated in abundance over sulfate reducers, and 1Biodiversity Research Centre, University of British anoxygenic phototrophs largely outnumbered oxy- Columbia, Vancouver, BC, Canada. genic photoautotrophs. Functional community 2Department of Zoology, University of British Columbia, structure correlated strongly with environmental vari- Vancouver, BC, Canada. ables, between and within a single bromeliad species. 3Department of Ecology, Biology Institute, Universidade Methanogens and reductive acetogens correlated Federal do Rio de Janeiro, Rio de Janeiro, Brazil. with detrital volume and canopy coverage, and exhib- 4Programa de Pos-Graduac ¸ao~ em Ecologia e ited higher relative abundances in N. cruenta.A Evoluc¸ao,~ Universidade Estadual do Rio de Janeiro, Rio comparison of bromeliads to freshwater lake sedi- de Janeiro, Brazil. ments and soil from around the world, revealed stark differences in terms of taxonomic as well as func- 5Programa de Pos-Graduac ¸ao~ em Ecologia, tional microbial community structure. Universidade Federal do Rio de Janeiro, Rio de Janeiro, Brazil. 6Biology Department & Center for Computational & Introduction Integrative Biology, Rutgers University, Camden, NJ, USA. Bromeliads (fam. Bromeliaceae) are plants found through- 7Department of Mathematics, University of British out the neotropics, with many species having rosette-like Columbia, Vancouver, BC, Canada. -

Water Relations of Bromeliaceae in Their Evolutionary Context
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Apollo Botanical Journal of the Linnean Society, 2016, 181, 415–440. With 2 figures Think tank: water relations of Bromeliaceae in their evolutionary context JAMIE MALES* Department of Plant Sciences, University of Cambridge, Downing Street, Cambridge CB2 3EA, UK Received 31 July 2015; revised 28 February 2016; accepted for publication 1 March 2016 Water relations represent a pivotal nexus in plant biology due to the multiplicity of functions affected by water status. Hydraulic properties of plant parts are therefore likely to be relevant to evolutionary trends in many taxa. Bromeliaceae encompass a wealth of morphological, physiological and ecological variations and the geographical and bioclimatic range of the family is also extensive. The diversification of bromeliad lineages is known to be correlated with the origins of a suite of key innovations, many of which relate directly or indirectly to water relations. However, little information is known regarding the role of change in morphoanatomical and hydraulic traits in the evolutionary origins of the classical ecophysiological functional types in Bromeliaceae or how this role relates to the diversification of specific lineages. In this paper, I present a synthesis of the current knowledge on bromeliad water relations and a qualitative model of the evolution of relevant traits in the context of the functional types. I use this model to introduce a manifesto for a new research programme on the integrative biology and evolution of bromeliad water-use strategies. The need for a wide-ranging survey of morphoanatomical and hydraulic traits across Bromeliaceae is stressed, as this would provide extensive insight into structure– function relationships of relevance to the evolutionary history of bromeliads and, more generally, to the evolutionary physiology of flowering plants. -
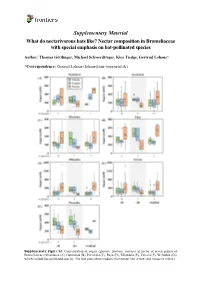
Supplementary Material What Do Nectarivorous Bats Like? Nectar Composition in Bromeliaceae with Special Emphasis on Bat-Pollinated Species
Supplementary Material What do nectarivorous bats like? Nectar composition in Bromeliaceae with special emphasis on bat-pollinated species Author: Thomas Göttlinger, Michael Schwerdtfeger, Kira Tiedge, Gertrud Lohaus* *Correspondence: Gertrud Lohaus ([email protected]) Supplementary Figure S1: Concentration of sugars (glucose, fructose, sucrose) in nectar of seven genera of Bromeliaceae (Alcantarea (A), Guzmania (B), Pitcairnia (C), Puya (D), Tillandsia (E), Vriesea (F), Werauhia (G)) which include bat-pollinated species. The box plots show medians (horizontal line in box) and means (x in box). Supplementary Material What do nectarivorous bats like? Nectar composition in Bromeliaceae with special emphasis on bat-pollinated species Author: Thomas Göttlinger, Michael Schwerdtfeger, Kira Tiedge, Gertrud Lohaus* *Correspondence: Gertrud Lohaus ([email protected]) Supplementary Figure S2: Concentration of amino acids (ala, arg, asn, asp, gaba, gln, glu, gly, his, iso, leu, lys, met, phe, pro, ser, thr, trp, tyr, val) in nectar of seven genera of Bromeliaceae (Alcantarea (A), Guzmania (B), Pitcairnia (C), Puya (D), Tillandsia (E), Vriesea (F), Werauhia (G)), which include bat-pollinated species. The box plots show medians (horizontal line in box) and means (x in box). Supplementary Material What do nectarivorous bats like? Nectar composition in Bromeliaceae with special emphasis on bat-pollinated species Author: Thomas Göttlinger, Michael Schwerdtfeger, Kira Tiedge, Gertrud Lohaus* *Correspondence: Gertrud Lohaus ([email protected]) Supplementary Figure S3: Cation concentrations (Ca2+, K+, Na+, Mg2+) in nectar of seven genera of Bromeliaceae (Alcantarea (A), Guzmania (B), Pitcairnia (C), Puya (D), Tillandsia (E), Vriesea (F), Werauhia (G)), which include bat-pollinated species. The box plots show medians (horizontal line in box) and means (x in box). -

Bromeletter the Official Journal of the Bromeliad Society of Australia Inc
1 BROMELETTER THE OFFICIAL JOURNAL OF THE BROMELIAD SOCIETY OF AUSTRALIA INC. MEETING DATE *3rd Saturday in bromeliad.org.au NOVEMBER ONLY ISSN 2208-0465 (Online) 18th NOVEMBER Vol 55 No 6 - November / December 2017. CONTENTS Management Details 2,3, 15,18,19 September - Plant of the Month, Margaret Draddy Artistic Competition 4,5 September Show, Tell, Ask 6 Aechmea ‘Pie in the Sky’ 6,7 BSA Spring Show - Results and Photos 8,9,10,11, 12,13 How to Ease Sunburn in Bromeliads - Paul Turvey 14 New Members 15 October - Plant of the Month, Margaret Draddy Artistic Competition 16,17 Seed Bank 18 Time to RENEW your MEMBERSHIP Membership renewal is due JANUARY 1st each year. Please consider whether or not you wish to receive your Bromeletter by email (as a PDF) Please return the enclosed (attached) renewal form to renew and update any details which have changed (or email the form found on our website). BROMELETTER is published bi-monthly at Sydney by The Bromeliad Society of Australia Incorporated. Deadlines for articles:15th of February, April, June, Au- gust, October and December, To allow for publishing in the first week of March, May, July, September, November and January. 2 COMMITTEE President Ian Hook 408 202 269 (president @bromeliad.org.au) Vice President(1), Kerry McNicol 0439 998 049 & Editor ([email protected]) Vice President (2) Meryl Thomas 0401 040 762 Secretary Carolyn Bunnell 02 9649 5762 Treasurer Alan Mathew 02 9516 5976 Member Helga Nitschke 0447 955 562 Member Patricia Sharpley 0439 672 826 Member Bob Sharpley 0409 361 778 Member Joy Clark 02 4572 3534 Member John Noonan 02 9627 5704 Member Ron Farrugia 02 9533 2546 BROMELIAD SOCIETIES AFFILIATED WITH THE BROMELIAD SOCIETY OF AUSTRALIA INC. -

Plant-Hummingbird Interactions and Temporal Nectar Availability in a Restinga from Brazil
Anais da Academia Brasileira de Ciências (2015) 87(4): 2163-2175 (Annals of the Brazilian Academy of Sciences) Printed version ISSN 0001-3765 / Online version ISSN 1678-2690 http://dx.doi.org/10.1590/0001-3765201520140349 www.scielo.br/aabc Plant-hummingbird interactions and temporal nectar availability in a restinga from Brazil LORENA C.N. FONSECA1,5, JEFERSON VIZENTIN-BUGONI2,6, ANDRÉ R. RECH3 and MARIA ALICE S. ALVES4 1Programa de Pós-Graduação em Ecologia, Universidade Federal do Rio de Janeiro/ UFRJ, CCS, IB, Caixa Postal 68020, 21941-590 Rio de Janeiro, RJ, Brasil 2Programa de Pós-Graduação em Ecologia, Instituto de Biologia, Universidade Estadual de Campinas/ UNICAMP, Rua Monteiro Lobato, 970, Barão Geraldo, 13083-970 Campinas, SP, Brasil 3Instituto de Biologia, Universidade Federal de Uberlândia/UFU, Av. Pará, 1720, Campus Umuarama, 38405-320 Uberlândia, MG, Brasil 4Departamento de Ecologia, Instituto de Biologia Roberto Alcantara Gomes, Universidade do Estado do Rio de Janeiro/UERJ, Rua São Francisco Xavier, 524, Maracanã, 20550-011 Rio de Janeiro, RJ, Brasil 5Companhia Ambiental do Estado de São Paulo/CETESB, Av. Professor Frederico Hermann Jr, 345, Pinheiros, 05459-900 São Paulo, SP, Brasil 6Center for Macroecology, Evolution and Climate, Natural History Museum of Denmark, University of Copenhagen, Universitetsparken 15, DK-2100 Copenhagen Ø, Denmark Manuscript received on July 10, 2014; accepted for publication on April 27, 2015 ABSTRACT Hummingbirds are the most important and specialized group of pollinating birds in the Neotropics and their interactions with plants are key components to many communities. In the present study we identified the assemblage of plants visited by hummingbirds and investigated the temporal availability of floral resources in an area of restinga, sandy plain coastal vegetation associated with the Atlantic forest, in Southeastern Brazil. -

Bromeletter the Official Journal of the Bromeliad Society of Australia Inc
1 BROMELETTER THE OFFICIAL JOURNAL OF THE BROMELIAD SOCIETY OF AUSTRALIA INC. bromeliad.org.au ISSN 2208-0465 (Online) Vol 56 No 6 - November / December 2018. Have a Merry Christmas & a safe and Happy New Year Photo by Ross Little Reminder: Annual subscriptions fall due 1.01.2019 BROMELETTER is published bi-monthly at Sydney by The Bromeliad Society of Australia Incorporated. Deadlines for articles:15th of February, April, June, August, October and December, To allow for publishing in the first week of March, May, July, September, November and January. 2 CONTENTS Management Details 2,3,15,18,19 Plant Of The Month, Discussion: September 4,5 Spring Show 2018 Results and photographs 6,7,8,9,10 Vale - Ivan Hope 10 Frost then Sun!!! Presentation of Trophies, Spring Show 11 Plant Of The Month, Discussion: October 12,13,14 Removing pups from plants with short stolons, eg Vriesea 16 Nidularium angustifolium - from FNCBSG - April2018 17 COMMITTEE President Ian Hook 0408 202 269 (president @bromeliad.org.au) Vice President(1), Kerry McNicol 0439 998 049 & Editor ([email protected]) Vice President (2) Meryl Thomas 0401 040 762 Secretary Carolyn Bunnell 02 9649 5762 Treasurer Alan Mathew 02 9516 5976 Member Helga Nitschke 0447 955 562 Member Patricia Sharpley 0439 672 826 Member Bob Sharpley 0409 361 778 Member Joy Clark 02 4572 3534 Member John Noonan 02 9627 5704 BROMELIAD SOCIETIES AFFILIATED WITH THE BROMELIAD SOCIETY OF AUSTRALIA INC. Bromeliad Society of Victoria. The Secretary, P.O. Box 101, Darling. Vic. 3145 Cover: Caboolture & Districts Brom. Society Inc., Nidularium angustifolium The Secretary, P.O. -

Ecosystem Services Provided by Bromeliad Plants: A
1 ECOSYSTEM SERVICES PROVIDED BY BROMELIAD PLANTS: A 2 SYSTEMATIC REVIEW 3 4 Geraldine Ladino Vásquez a,*, Fabiola Ospina-Bautista a, Jaime Vicente Estévez Varón 5 a, Lucie Jerabkovab, Pavel Kratina c 6 7 a Departamento de Ciencias Biológicas, Universidad de Caldas, Street 65 No. 26 – 10, 8 Manizales, Caldas, 170004, Colombia. 9 b Department of Geography, King’s College London, 30 Aldwych, London WC2B 10 4GB, UK. 11 c School of Biological and Chemical Sciences, Queen Mary University of London, Mile 12 End Rd, London E1 4NS, UK. 13 14 15 16 *Corresponding author 17 18 E-mail addresses: [email protected] (G. Ladino-V), 19 [email protected] (F. Ospina-B), [email protected] (J.V. 20 Estévez Varón), [email protected] (L. Jerabkova), [email protected] (P. 21 Kratina). 22 23 24 Keywords: Biodiversity; bromeliad plants; climate regulation; disease; ecosystem 25 services; microecosystems; Neotropics; pharmaceutical potential; water storage. 1 26 ABSTRACT 27 The unprecedented loss of biological diversity has negative impacts on ecosystems and 28 the associated benefits which they provide to humans. Bromeliads have high diversity 29 throughout the Neotropics, but they have been negatively affected by habitat loss and 30 fragmentation, climate change, herbivorous species invasions, and they are also being 31 commercialized for ornamental use. These plants provide direct benefits to the human 32 society and they also form micro ecosystems in which accumulated water and nutrients 33 support the communities of aquatic and terrestrial species, thus maintaining local 34 diversity. We performed a systematic review of the contribution of bromeliads to 35 ecosystem services across their native geographical distribution. -

Published Version
Received: 12 October 2017 | Revised: 4 March 2018 | Accepted: 9 March 2018 DOI: 10.1002/ece3.4111 ORIGINAL RESEARCH Long- term regeneration of a tropical plant community after sand mining Mário L. Garbin1 | Flora Misaki1 | Poliana F. Ferreira1 | Karlo G. Guidoni-Martins2 | Rayara B. Soares1 | Pierre Mariotte3,4 | Jerônimo B. B. Sansevero5 | Patryck Gouvea Rocha1 | Ary G. Silva1 1Programa de Pós-Graduação em Ecologia de Ecossistemas, Universidade Vila Velha, Abstract Vila Velha, Brazil Sandy coastal plant communities in tropical regions have been historically under 2 Programa de Pós-Graduação em Ecologia strong anthropic pressure. In Brazil, these systems shelter communities with highly e Evolução, Universidade Federal de Goiás, Goiânia, Brazil plastic plant species. However, the potential of these systems to regenerate without 3School of Architecture, Civil human assistance after disturbances has hardly been examined. We determined the and Environmental Engineering natural regeneration of a coastal sandy plain vegetation (restinga) in Brazil, 16 years (ENAC), Laboratory of Ecological Systems (ECOS), Ecole Polytechnique Fédérale de after the end of sand removal. We inventoried 38 plots: 20 within a sand- mined site Lausanne EPFL, Lausanne, Switzerland and 18 in an adjacent undisturbed site. We expected lower diversity values in the 4Snow and Landscape Research (WSL), Swiss Federal Institute for Forest, Lausanne, sand- mined site compared to the undisturbed site, but similar species composition Switzerland between the two sites due to the spatial proximity of the two sites and the high plas- 5 Instituto de Florestas (IF), Departamento ticity of restinga species. Species were ranked using abundance and importance de Ciências Ambientais (DCA), Universidade Federal Rural do Rio de Janeiro (UFRRJ), Rio value index in both sites, and comparisons were performed using Rényi entropy pro- de Janeiro, Brazil files, rarefaction curves, principal component analysis, and redundancy analysis. -

S.F.V.B.S. San Fernando Valley Bromeliad Society May 2018 P.O
S.F.V.B.S. SAN FERNANDO VALLEY BROMELIAD SOCIETY MAY 2018 P.O. BOX 16561, ENCINO, CA 91416-6561 sfvbromeliad.homestead.com [email protected] Elected OFFICERS & Volunteers Pres: Bryan Chan & Carole Scott V.P.: John Martinez Sec: Leni Koska Treas: Mary Chan Membership: Joyce Schumann Advisors/Directors: Steve Ball, Richard Kaz –fp, Mike Wisnev –fp, Mary K., Sunshine Chair: Georgia Roiz Refreshments: vacant Web: Mike Wisnev, Editors: Mike Wisnev & Mary K., Snail Mail: Nancy P-Hapke next meeting: Saturday May 5, 2018 @ 10:00 am Sepulveda Garden Center 16633 Magnolia Blvd. Encino, California 91436 AGENDA 9:30 – SET UP & SOCIALIZE 11:15 - Refreshment Break and Show and Tell: 10:00 - Door Prize drawing – one member Will the following members please provide who arrives before 10:00 gets a Bromeliad refreshments this month: Barbara Wynn, Colleen 10:05 -Welcome Visitors and New Members. Baida, Steve Ball, Wesley Bartera, David Bassani, Make announcements and Introduce Speaker Kaz Benadom, Helen Berger, Jeanette Bond and anyone else who has a snack they would like to 10:15 –Speaker : Wendell S. (Woody) Minnich share. If you can’t contribute this month don’t stay Program Topic: “Bromeliads, Cacti and away…. just bring a snack next time you come. Succulents in Oaxaca” Feed The Kitty This is a new program presentation, no one has seen it before. If you don’t contribute to the refreshment table, Woody, as he is commonly please make a small donation to (feed the kitty jar) called, has been in the on the table; this helps fund the coffee breaks. -

Bat Pollination in Bromeliaceae Pedro A
PLANT ECOLOGY & DIVERSITY https://doi.org/10.1080/17550874.2019.1566409 ARTICLE Bat pollination in Bromeliaceae Pedro A. Aguilar-Rodrígueza, Thorsten Krömer a, Marco Tschapka b,c, José G. García-Francod, Jeanett Escobedo-Sartie and M.Cristina MacSwiney G. a aCentro de Investigaciones Tropicales, Universidad Veracruzana, Xalapa, Veracruz, Mexico; bInstitute of Evolutionary Ecology and Conservation Genomics, University of Ulm, Ulm, Germany; cSmithsonian Tropical Research Institute, Balboa Ancón, Panamá, República de Panamá; dRed de Ecología Funcional, Instituto de Ecología, Xalapa, Veracruz, México; eFacultad de Ciencias Biológicas y Agropecuarias, Universidad de Colima, Crucero de Tecomán, Tecomán, Colima, México ABSTRACT ARTICLE HISTORY Background: Chiropterophily encompasses the floral traits by which bats are attracted as the Received 2 May 2017 main pollinators. Among the chiropterophilous flowering plants of the New World, Accepted 3 January 2019 Bromeliaceae is one of the most ecologically important families; however, information KEYWORDS about the chiropterophilous interaction in this family is still scarce. Anoura; bromeliads; Aims: We present a comprehensive review of bat pollination in bromeliads, covering floral chiropterophily; floral scent; traits, rewards offered to pollinators, floral attractants and the identity of visiting bat species. nectar; pollination; Werauhia Methods: We discuss traits shared among chiropterophilous bromeliads and present general trends in an evolutionary context. We constructed a phylogenetic tree to elucidate the ancestral pollination syndromes of the 42 extant bromeliad species (ca. 1% of total) known to be bat-pollinated. Results: Most of the species within the ten genera reported belong to the Tillandsioideae subfamily, with three genera appearing to be exclusively bat-pollinated. Floral visitors include 19 bat species of 11 genera from the Phyllostomidae. -

Nuclear Genes, Matk and the Phylogeny of the Poales
Zurich Open Repository and Archive University of Zurich Main Library Strickhofstrasse 39 CH-8057 Zurich www.zora.uzh.ch Year: 2018 Nuclear genes, matK and the phylogeny of the Poales Hochbach, Anne ; Linder, H Peter ; Röser, Martin Abstract: Phylogenetic relationships within the monocot order Poales have been well studied, but sev- eral unrelated questions remain. These include the relationships among the basal families in the order, family delimitations within the restiid clade, and the search for nuclear single-copy gene loci to test the relationships based on chloroplast loci. To this end two nuclear loci (PhyB, Topo6) were explored both at the ordinal level, and within the Bromeliaceae and the restiid clade. First, a plastid reference tree was inferred based on matK, using 140 taxa covering all APG IV families of Poales, and analyzed using parsimony, maximum likelihood and Bayesian methods. The trees inferred from matK closely approach the published phylogeny based on whole-plastome sequencing. Of the two nuclear loci, Topo6 supported a congruent, but much less resolved phylogeny. By contrast, PhyB indicated different phylo- genetic relationships, with, inter alia, Mayacaceae and Typhaceae sister to Poaceae, and Flagellariaceae in a basally branching position within the Poales. Within the restiid clade the differences between the three markers appear less serious. The Anarthria clade is first diverging in all analyses, followed by Restionoideae, Sporadanthoideae, Centrolepidoideae and Leptocarpoideae in the matK and Topo6 data, but in the PhyB data Centrolepidoideae diverges next, followed by a paraphyletic Restionoideae with a clade consisting of the monophyletic Sporadanthoideae and Leptocarpoideae nested within them. The Bromeliaceae phylogeny obtained from Topo6 is insufficiently sampled to make reliable statements, but indicates a good starting point for further investigations.