Dangerous Marine Species in the Arabian Gulf
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Marine Envenomations
Environmental Marine envenomations Ingrid Berling Geoffrey Isbister Background The majority of marine envenomings are minor and do Marine stings are common but most are minor and do not not require medical intervention. Jellyfish stings are a require medical intervention. Severe and systemic marine frequent reason for presentation to first aid and primary envenoming is uncommon, but includes box jellyfish stings, healthcare providers. A knowledge of the variety of stings Irukandji syndrome, major stingray trauma and blue-ringed and envenoming syndromes that occur in Australia, octopus envenoming. Almost all marine injuries are caused including those that are clinically significant, and available by jellyfish stings, and penetrating injuries from spiny fish, treatments, is necessary for practitioners, particularly those stingrays or sea urchins. working in coastal regions. Objective This article describes the presentation and management Marine envenoming can be considered in two broad categories: of marine envenomations and injuries that may occur in jellyfish stings and penetrating venomous marine injuries. Jellyfish Australia. stings range from the life-threatening major box jellyfish (Chironex Discussion fleckeri) to painful, but generally benign, bluebottle stings common First aid for jellyfish includes tentacle removal, application to most southeastern Australian beaches (Figure 1). Penetrating of vinegar for box jellyfish, and hot water immersion (45°C venomous marine injuries often occur when handling fish, but can for 20 min) for bluebottle jellyfish stings. Basic life support occur to anyone involved in water activities, fresh water or marine. is essential for severe marine envenomings that result in They are typically more painful than just the trauma of the wound, and cardiac collapse or paralysis. -

Traveler Information
Traveler Information QUICK LINKS Marine Hazards—TRAVELER INFORMATION • Introduction • Risk • Hazards of the Beach • Animals that Bite or Wound • Animals that Envenomate • Animals that are Poisonous to Eat • General Prevention Strategies Traveler Information MARINE HAZARDS INTRODUCTION Coastal waters around the world can be dangerous. Swimming, diving, snorkeling, wading, fishing, and beachcombing can pose hazards for the unwary marine visitor. The seas contain animals and plants that can bite, wound, or deliver venom or toxin with fangs, barbs, spines, or stinging cells. Injuries from stony coral and sea urchins and stings from jellyfish, fire coral, and sea anemones are common. Drowning can be caused by tides, strong currents, or rip tides; shark attacks; envenomation (e.g., box jellyfish, cone snails, blue-ringed octopus); or overconsumption of alcohol. Eating some types of potentially toxic fish and seafood may increase risk for seafood poisoning. RISK Risk depends on the type and location of activity, as well as the time of year, winds, currents, water temperature, and the prevalence of dangerous marine animals nearby. In general, tropical seas (especially the western Pacific Ocean) are more dangerous than temperate seas for the risk of injury and envenomation, which are common among seaside vacationers, snorkelers, swimmers, and scuba divers. Jellyfish stings are most common in warm oceans during the warmer months. The reef and the sandy sea bottom conceal many creatures with poisonous spines. The highly dangerous blue-ringed octopus and cone shells are found in rocky pools along the shore. Sea anemones and sea urchins are widely dispersed. Sea snakes are highly venomous but rarely bite. -

The Polyp and the Medusa Life on the Move
The Polyp and the Medusa Life on the Move Millions of years ago, unlikely pioneers sparked a revolution. Cnidarians set animal life in motion. So much of what we take for granted today began with Cnidarians. FROM SHAPE OF LIFE The Polyp and the Medusa Life on the Move Take a moment to follow these instructions: Raise your right hand in front of your eyes. Make a fist. Make the peace sign with your first and second fingers. Make a fist again. Open your hand. Read the next paragraph. What you just did was exhibit a trait we associate with all animals, a trait called, quite simply, movement. And not only did you just move your hand, but you moved it after passing the idea of movement through your brain and nerve cells to command the muscles in your hand to obey. To do this, your body needs muscles to move and nerves to transmit and coordinate movement, whether voluntary or involuntary. The bit of business involved in making fists and peace signs is pretty complex behavior, but it pales by comparison with the suites of thought and movement associated with throwing a curve ball, walking, swimming, dancing, breathing, landing an airplane, running down prey, or fleeing a predator. But whether by thought or instinct, you and all animals except sponges have the ability to move and to carry out complex sequences of movement called behavior. In fact, movement is such a basic part of being an animal that we tend to define animalness as having the ability to move and behave. -
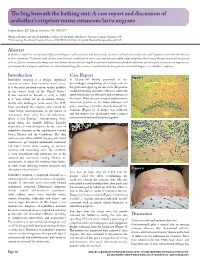
The Bug Beneath the Bathing Suit: a Case Report and Discussion of Seabather’S Eruption Versus Cutaneous Larva Migrans
The bug beneath the bathing suit: A case report and discussion of seabather’s eruption versus cutaneous larva migrans Andrew Jensen, BS,* Marcus Goodman, DO, FAOCD** *Medical Student, 4th year, Philadelphia College of Osteopathic Medicine - Georgia Campus, Suwanee, GA **Dermatology Residency Program Director, PCOM/North Fulton Hospital Medical Campus, Roswell, GA Abstract Seabather’s eruption is an important differential diagnosis when a patient who has recently swum in a subtropical ocean presents with a pruritic rash in the distribution of their swimwear. Treatment with systemic corticosteroids is indicated in severe cases and can successfully reduce symptoms. Oral steroid therapy in general has proven to be an effective treatment for many acute and chronic diseases but has long been associated with increased risk for infections. In this report, we present an atypical case of cutaneous larva migrans and discuss its clinical unmasking after systemic steroid treatment was given for an initial diagnosis of seabather’s eruption. Introduction Case Report Figure 2 Seabather’s eruption is a benign, superficial A 52-year-old female presented to her reaction to toxins from marine-animal larvae. dermatologist complaining of an itchy rash on It is the most common marine-related problem her groin and upper leg for one week. The patient in the waters south of the United States.1 stated she recently traveled to Mexico, where she It was reported in Florida as early as 1903 spent several days on the beach and swimming in as a “rash which set up an intense itching” the ocean. Physical exam revealed erythematous, shortly after bathing in ocean water.2 In 1949, edematous papules on her lower abdomen and Sams postulated the eruption was caused by groin, assuming a location directly beneath her “some living, microorganism, in the nature of swimsuit (Figure 1). -

Marine Mammals and Sea Turtles of the Mediterranean and Black Seas
Marine mammals and sea turtles of the Mediterranean and Black Seas MEDITERRANEAN AND BLACK SEA BASINS Main seas, straits and gulfs in the Mediterranean and Black Sea basins, together with locations mentioned in the text for the distribution of marine mammals and sea turtles Ukraine Russia SEA OF AZOV Kerch Strait Crimea Romania Georgia Slovenia France Croatia BLACK SEA Bosnia & Herzegovina Bulgaria Monaco Bosphorus LIGURIAN SEA Montenegro Strait Pelagos Sanctuary Gulf of Italy Lion ADRIATIC SEA Albania Corsica Drini Bay Spain Dardanelles Strait Greece BALEARIC SEA Turkey Sardinia Algerian- TYRRHENIAN SEA AEGEAN SEA Balearic Islands Provençal IONIAN SEA Syria Basin Strait of Sicily Cyprus Strait of Sicily Gibraltar ALBORAN SEA Hellenic Trench Lebanon Tunisia Malta LEVANTINE SEA Israel Algeria West Morocco Bank Tunisian Plateau/Gulf of SirteMEDITERRANEAN SEA Gaza Strip Jordan Suez Canal Egypt Gulf of Sirte Libya RED SEA Marine mammals and sea turtles of the Mediterranean and Black Seas Compiled by María del Mar Otero and Michela Conigliaro The designation of geographical entities in this book, and the presentation of the material, do not imply the expression of any opinion whatsoever on the part of IUCN concerning the legal status of any country, territory, or area, or of its authorities, or concerning the delimitation of its frontiers or boundaries. The views expressed in this publication do not necessarily reflect those of IUCN. Published by Compiled by María del Mar Otero IUCN Centre for Mediterranean Cooperation, Spain © IUCN, Gland, Switzerland, and Malaga, Spain Michela Conigliaro IUCN Centre for Mediterranean Cooperation, Spain Copyright © 2012 International Union for Conservation of Nature and Natural Resources With the support of Catherine Numa IUCN Centre for Mediterranean Cooperation, Spain Annabelle Cuttelod IUCN Species Programme, United Kingdom Reproduction of this publication for educational or other non-commercial purposes is authorized without prior written permission from the copyright holder provided the sources are fully acknowledged. -

A Guide to Harmful and Toxic Creatures in the Goa of Jordan
Published by the Royal Marine Conservation Society of Jordan. P. O. Box 831051, Abdel Aziz El Thaalbi St., Shmesani 11183. Amman Copyright: © The Royal Marine Conservation Society of Jordan Reproduction of this publication for educational and other non- commercial purposes is authorized without prior written approval from the copyright holder provided the source is fully acknowledged. ISBN: 978-9957-8740-1-8 Deposit Number at the National Library: 2619/6/2016 Citation: Eid, E and Al Tawaha, M. (2016). A Guide to Harmful and Toxic Creature in the Gulf of Aqaba of Jordan. The Royal Marine Conservation Society of Jordan. ISBN: 978-9957-8740-1-8. Pp 84. Material was reviewed by Dr Nidal Al Oran, International Research Center for Water, Environment and Energy\ Al Balqa’ Applied University,and Dr. Omar Attum from Indiana University Southeast at the United State of America. Cover page: Vlad61; Shutterstock Library All photographs used in this publication remain the property of the original copyright holder, and it should not be reproduced or used in other contexts without permission. 1 Content Index of Creatures Described in this Guide ......................................................... 5 Preface ................................................................................................................ 6 Part One: Introduction ......................................................................................... 8 1.1 The Gulf of Aqaba; Jordan ......................................................................... 8 1.2 Aqaba; -

Report on the Status of Mediterranean Chondrichthyan Species
United Nations Environment Programme Mediterranean Action Plan Regional Activity Centre For Specially Protected Areas REPORT ON THE STATUS OF MEDITERRANEAN CHONDRICHTHYAN SPECIES D. CEBRIAN © L. MASTRAGOSTINO © R. DUPUY DE LA GRANDRIVE © Note : The designations employed and the presentation of the material in this document do not imply the expression of any opinion whatsoever on the part of UNEP concerning the legal status of any State, Territory, city or area, or of its authorities, or concerning the delimitation of their frontiers or boundaries. © 2007 United Nations Environment Programme Mediterranean Action Plan Regional Activity Centre for Specially Protected Areas (RAC/SPA) Boulevard du leader Yasser Arafat B.P.337 –1080 Tunis CEDEX E-mail : [email protected] Citation: UNEP-MAP RAC/SPA, 2007. Report on the status of Mediterranean chondrichthyan species. By Melendez, M.J. & D. Macias, IEO. Ed. RAC/SPA, Tunis. 241pp The original version (English) of this document has been prepared for the Regional Activity Centre for Specially Protected Areas (RAC/SPA) by : Mª José Melendez (Degree in Marine Sciences) & A. David Macías (PhD. in Biological Sciences). IEO. (Instituto Español de Oceanografía). Sede Central Spanish Ministry of Education and Science Avda. de Brasil, 31 Madrid Spain [email protected] 2 INDEX 1. INTRODUCTION 3 2. CONSERVATION AND PROTECTION 3 3. HUMAN IMPACTS ON SHARKS 8 3.1 Over-fishing 8 3.2 Shark Finning 8 3.3 By-catch 8 3.4 Pollution 8 3.5 Habitat Loss and Degradation 9 4. CONSERVATION PRIORITIES FOR MEDITERRANEAN SHARKS 9 REFERENCES 10 ANNEX I. LIST OF CHONDRICHTHYAN OF THE MEDITERRANEAN SEA 11 1 1. -

Our Summer Newsletter Has Gone to the Dogs!
Summer 2013 Newsletter Our summer newsletter has gone to the dogs! In this issue… Pet-Friendly Rentals | Tips for taking your dog to the beach | Beach essentials equipment list | Area vets and boarding | OIB doggy photos | What’s new? and more. There are huge benefits when you travel with your pet. No boarding fees is just one, but there's also no anxiety about how your pet is coping while you're away, less separation stress for your pet, more spacious accommodations that will be just like they're used to at home, and a fun-filled vacation that includes your pet being included with the family. Whether you're a family taking a summer vacation or snowbirds coming south for the winter, you'll be comfortable knowing your pet is welcome here too and that we have a variety of pet friendly rentals to choose from. OCEANFRONT HOUSES 358 East First Street…4 bedroom…$2195 434 East Second St….3 bedroom…$2195 SECOND ROW HOUSES 93 East First Street…..3 bedroom….$965 119 East First Street….4 bedroom…$915 345 East First Street…6 bedroom…$4015 THIRD ROW HOUSES 78 East Second Street..4 bedroom…$995 MID-ISLAND HOUSES 2 Driftwood Street…...2 bedroom…$995 CANAL HOUSES 1 Concord Street……4 bedroom…$1145 41 Concord Street…...4 bedroom…$1315 69 Fairmont Street……4 bedroom…$995 44 Leland Street….…5 bedroom…$1460 26 Wilmington St……6 bedroom…$1095 CONDOS Ocean Cove 105……..1 bedroom…$995 Ocean Cove 219……...1 bedroom…$995 Oceanside West F-1…..3 bedroom…$975 VIOLET, photo courtesy of Julie 1. -

Ivermectin Binding Sites in Human and Invertebrate Cys-Loop Receptors
1 Ivermectin binding sites in human and invertebrate Cys-loop receptors 2 Timothy Lynagh1, Joseph W. Lynch2 3 4 1 Neurosensory Systems Group, Technical University of Darmstadt, Schnittspahnstrasse 3, 64287 5 Darmstadt, Germany 6 2 Queensland Brain Institute and School of Biomedical Sciences, The University of Queensland, 7 Brisbane, QLD 4072, Australia 8 9 Corresponding author: Lynch, J.W. ([email protected]). Phone: +617 33466375 10 11 1 12 Abstract 13 Ivermectin is a gold-standard antiparasitic drug that has been used successfully to treat billions of 14 humans, livestock and pets. Until recently, the binding site on its Cys-loop receptor target had 15 been a mystery. Recent protein crystal structures, site-directed mutagenesis data and molecular 16 modelling now explain how ivermectin binds to these receptors and reveal why it is selective for 17 invertebrate members of the Cys-loop receptor family. Combining this with emerging genomic 18 information, we are now in a position to predict species sensitivity to ivermectin and better 19 understand the molecular basis of ivermectin resistance. An understanding of the molecular 20 structure of the ivermectin binding site, which is formed at the interface of two adjacent subunits 21 in the transmembrane domain of the receptor, should also aid the development of new lead 22 compounds as both anthelmintics and as therapies for a wide variety of human neurological 23 disorders. 24 25 New insight into a wonder drug 26 Orally- or topically-administered ivermectin is used to eliminate parasitic nematodes and 27 arthropods from animals and humans[1]. It is effective yet well-tolerated due to the potency with 28 which it activates a uniquely invertebrate member of the Cys-loop receptor family of ligand-gated 29 ion channels[2]. -

Training Manual Series No.15/2018
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by CMFRI Digital Repository DBTR-H D Indian Council of Agricultural Research Ministry of Science and Technology Central Marine Fisheries Research Institute Department of Biotechnology CMFRI Training Manual Series No.15/2018 Training Manual In the frame work of the project: DBT sponsored Three Months National Training in Molecular Biology and Biotechnology for Fisheries Professionals 2015-18 Training Manual In the frame work of the project: DBT sponsored Three Months National Training in Molecular Biology and Biotechnology for Fisheries Professionals 2015-18 Training Manual This is a limited edition of the CMFRI Training Manual provided to participants of the “DBT sponsored Three Months National Training in Molecular Biology and Biotechnology for Fisheries Professionals” organized by the Marine Biotechnology Division of Central Marine Fisheries Research Institute (CMFRI), from 2nd February 2015 - 31st March 2018. Principal Investigator Dr. P. Vijayagopal Compiled & Edited by Dr. P. Vijayagopal Dr. Reynold Peter Assisted by Aditya Prabhakar Swetha Dhamodharan P V ISBN 978-93-82263-24-1 CMFRI Training Manual Series No.15/2018 Published by Dr A Gopalakrishnan Director, Central Marine Fisheries Research Institute (ICAR-CMFRI) Central Marine Fisheries Research Institute PB.No:1603, Ernakulam North P.O, Kochi-682018, India. 2 Foreword Central Marine Fisheries Research Institute (CMFRI), Kochi along with CIFE, Mumbai and CIFA, Bhubaneswar within the Indian Council of Agricultural Research (ICAR) and Department of Biotechnology of Government of India organized a series of training programs entitled “DBT sponsored Three Months National Training in Molecular Biology and Biotechnology for Fisheries Professionals”. -

You're a Jellyfish!
You’re a Jellyfish! LEVELED BOOK • O A Reading A–Z Level O Leveled Book Word Count: 718 Written by Kira Freed • Illustrated by Cende Hill Visit www.readinga-z.com www.readinga-z.com for thousands of books and materials. Photo Credits: Front cover: © Randy Olson/National Geographic Image Collection; back cover: © Ingo Arndt/Minden Pictures/National Geographic Stock; title page: © iStock.com/ Robert Weber; page 3: © Jupiterimages Corporation; page 4: © Kyodo/Newscom; page 5: © Mariusz Jurgielewicz/123RF; page 7 (main): © Stuart Westmorland/Corbis Documentary/Getty Images; page 7 (inset): © Ken Lucas/Visuals Unlimited/Getty Images; page 9: © iStock.com/Richard McMillin; page 11 (left): © Paul Sutherland/ National Geographic Stock; page 11 (top right, bottom right): © David Doubilet/National Geographic Stock; page 12: © ullstein bild/Getty Images; page 13: © Brian Cassey/ AP Images; page 14 (background): © iStock.com/10four design group; page 14 (main): Caroline Hall/Alamy; page 15: © NOAA/Monterey Bay Aquarium Research Institute You’re a Jellyfish! Level O Leveled Book © Learning A–Z Correlation Written by Kira Freed LEVEL O Written by Kira Freed Illustrated by Cende Hill Illustrated by Cende Hill Fountas & Pinnell M All rights reserved. Reading Recovery 20 www.readinga-z.com www.readinga-z.com DRA 28 Table of Contents This is a lion’s mane jellyfish. Floating at Sea . 4 Floating at Sea Jelly Bodies . 6 Imagine that your body is shaped like Getting Around . 8 a bell, and you have long tentacles Life Cycle . 10 streaming down under you . You are Hunter and Hunted . 11 mostly made of water and cannot survive away from it . -

2014 What-To-Do-In-A-Medical-Emerg
What To Do in a Medical Emergency MEDICAL EMERGENCIES WHAT YOU NEED TO KNOW FROM AMERICA’S EMEGENCY PHYSICIANS www.EmergencyCareForYou.org What To Do in a Medical Emergency At what point does a fever or stomach ache become a med- ical emergency? If you slice your finger with a knife, or you are having the worst headache you’ve ever had, should you seek emergency care? How do you know? The following information from the nation's emergency physicians will help you identify the signs and symptoms of emergency conditions. However, emergency physicians will tell you — if you think you are having a medical emer- gency, seek emergency care right away. Better safe, than sorry. Too many people, such as men with chest pain, wait too long to seek care. The advice in this publication is about how to handle com- mon emergency medical conditions. It does not contain all the signs or symptoms of medical emergencies, and the advice is not intended to be a substitute for consulting with a medical professional. If you think you are experiencing a medical emergency, seek immediate medical attention. www.EmergencyCareForYou.org 2 TABLE OF CONTENTS Contents Abdominal Pain ..............................................................................................4 Asthma and Allergies.....................................................................................4 Anaphylaxis.....................................................................................................8 Adverse Drug Reactions ................................................................................9