Enkephalin Systems in Diencephalon and Brainstem of the Rat
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Interruption of the Connections of the Mammillary Bodies Protects Against Generalized Pentylenetetrazol Seizures in Guinea Pigs
The Journal of Neuroscience, March 1987, 7(3): 662-670 Interruption of the Connections of the Mammillary Bodies Protects Against Generalized Pentylenetetrazol Seizures in Guinea Pigs Marek A. Mirski and James A. Ferrendelli Division of Clinical Neuropharmacology, Department of Pharmacology and Department of Neurology and Neurological Surgery, Washington University School of Medicine, St. Louis, Missouri 63110 Electrolytic lesions in the anterior and mid-diencephalon and Morin, 1953; Gellhorn et al., 1959) fields of Fore1(Jinnai, 1966; ventral midbrain in guinea pigs were produced to examine Jinnai et al., 1969; Jinnai and Mukawa, 1970), substantianigra the effects of interruption of the fornix (FX), mammillothal- (Iadarola and Gale, 1982; Garant and Gale, 1983; Gonzalez and amic tracts (MT), and mammillary peduncles (MP), respec- Hettinger, 1984; McNamara et al., 1983, 1984), and several tively, on the expression of pentylenetetrazol (PTZ) sei- thalamic nuclei (Mullen et al., 1967; Jinnai et al., 1969; Feeney zures. As a group, all mid-diencephalic lesioned animals and Gullotta, 1972; Kusske et al., 1972; Van Straaten, 1975; had some degree of protection from the electroencephalo- Quesney et al., 1977). graphic and behavioral convulsant and lethal effects of the Recently we observed the selective metabolic activation of drug. Through a composite volume analysis of protected the mammillary bodies (MB) and their immediate connections versus unprotected animals, as well as a retrospective com- during a threshold convulsive stimulus induced by the co-in- parison between MT and non-MT lesioned animals, it was fusion of pentylenetetrazol (PTZ) and ethosuximide (ESM) demonstrated that small mid-diencephalic lesions incorpo- (Mirski and Ferrendelli, 1983). -

Distribution of Neurotransmitters in the Sheep Brain
Journal of Reproduction and Fertility Supplement 49, 199-220 Distribution of neurotransmitters in the sheep brain Y. Tillet Laborcttoirede NeuroendocrinologieSexuelle, Station de Physiologiede la Reproductiondes Mammiferes Domestiques, INRA, 37380 Nouzilly, France Although the general organization of the sheep brain is similar to that of other mammals, there are species differences in the fine architecture and neurotransmitter distribution. In sheep, perikarya are generally scattered, unlike the situation in rodents where they are clustered. The same organization is observed in cows and primates. The density of neurones immunoreactive for tyrosine hydroxylase in the dorsorostral diencephalon of sheep is lower than in rodents; A14 and A15 dopaminergic cell groups do not present a dorsal part. Only one adrenergic group, C2, is observed in the dorsomedial medulla oblongata. GnRH-immunoreactive neurones are mainly found in the anterior hypothalamic—preoptic areas, a few being present in the mediobasal hypothalamus. The density of several neurones contain- ing neuropeptides (for example vasoactive intestinal polypeptide, cholecystokinin and somatostatin) in the caudal brain of sheep is lower than in other species and in the forebrain of sheep. These differences contribute to different patterns of innervation of brain areas compared with other species. For example, the supra- chiasmatic nucleus does not present a dense network of fibres immunoreactive for 5-hydroxytryptamine and neuropeptide Y as observed in rats. These morphological studies constitute information necessary for further physiological investigations. Introduction In sheep, as in other species, neurotransmitters in the brain are involved in the control of physiological cues through endocrine and autonomic regulation. Among the species used to study endocrine regulation, sheep present interesting and specific physiological characteristics. -

Deconstructing Arousal Into Wakeful, Autonomic and Affective Varieties
Neuroscience Letters xxx (xxxx) xxx–xxx Contents lists available at ScienceDirect Neuroscience Letters journal homepage: www.elsevier.com/locate/neulet Review article Deconstructing arousal into wakeful, autonomic and affective varieties ⁎ Ajay B. Satputea,b, , Philip A. Kragelc,d, Lisa Feldman Barrettb,e,f,g, Tor D. Wagerc,d, ⁎⁎ Marta Bianciardie,f, a Departments of Psychology and Neuroscience, Pomona College, Claremont, CA, USA b Department of Psychology, Northeastern University, Boston, MA, USA c Department of Psychology and Neuroscience, University of Colorado Boulder, Boulder, USA d The Institute of Cognitive Science, University of Colorado Boulder, Boulder, USA e Athinoula A. Martinos Center for Biomedical Imaging, Massachusetts General Hospital, Boston, MA, USA f Department of Radiology, Harvard Medical School, Boston, MA, USA g Department of Psychiatry, Massachusetts General Hospital, Boston, MA, USA ARTICLE INFO ABSTRACT Keywords: Arousal plays a central role in a wide variety of phenomena, including wakefulness, autonomic function, affect Brainstem and emotion. Despite its importance, it remains unclear as to how the neural mechanisms for arousal are or- Arousal ganized across them. In this article, we review neuroscience findings for three of the most common origins of Sleep arousal: wakeful arousal, autonomic arousal, and affective arousal. Our review makes two overarching points. Autonomic First, research conducted primarily in non-human animals underscores the importance of several subcortical Affect nuclei that contribute to various sources of arousal, motivating the need for an integrative framework. Thus, we Wakefulness outline an integrative neural reference space as a key first step in developing a more systematic understanding of central nervous system contributions to arousal. -

Cellular Changes in Injured Rat Spinal Cord Following Electrical Brainstem Stimulation
brain sciences Article Cellular Changes in Injured Rat Spinal Cord Following Electrical Brainstem Stimulation Walter J. Jermakowicz 1,* , Stephanie S. Sloley 2, Lia Dan 2, Alberto Vitores 2, Melissa M. Carballosa-Gautam 2 and Ian D. Hentall 2 1 Department of Neurological Surgery, University of Miami, 1095 NW 14th Terr, Miami, FL 33136, USA 2 Miami Project to Cure Paralysis, University of Miami, 1095 NW 14th Terr., Miami, FL 33136, USA; [email protected] (S.S.S.); [email protected] (L.D.); [email protected] (A.V.); [email protected] (M.M.C.-G.); [email protected] (I.D.H.) * Correspondence: [email protected]; Tel.: +1-615-818-3070 Received: 6 May 2019; Accepted: 27 May 2019; Published: 28 May 2019 Abstract: Spinal cord injury (SCI) is a major cause of disability and pain, but little progress has been made in its clinical management. Low-frequency electrical stimulation (LFS) of various anti-nociceptive targets improves outcomes after SCI, including motor recovery and mechanical allodynia. However, the mechanisms of these beneficial effects are incompletely delineated and probably multiple. Our aim was to explore near-term effects of LFS in the hindbrain’s nucleus raphe magnus (NRM) on cellular proliferation in a rat SCI model. Starting 24 h after incomplete contusional SCI at C5, intermittent LFS at 8 Hz was delivered wirelessly to NRM. Controls were given inactive stimulators. At 48 h, 5-bromodeoxyuridine (BrdU) was administered and, at 72 h, spinal cords were extracted and immunostained for various immune and neuroglial progenitor markers and BrdU at the level of the lesion and proximally and distally. -

Memory Loss from a Subcortical White Matter Infarct
J Neurol Neurosurg Psychiatry: first published as 10.1136/jnnp.51.6.866 on 1 June 1988. Downloaded from Journal of Neurology, Neurosurgery, and Psychiatry 1988;51:866-869 Short report Memory loss from a subcortical white matter infarct CAROL A KOOISTRA, KENNETH M HEILMAN From the Department ofNeurology, College ofMedicine, University ofFlorida, and the Research Service, Veterans Administration Medical Center, Gainesville, FL, USA SUMMARY Clinical disorders of memory are believed to occur from the dysfunction of either the mesial temporal lobe, the mesial thalamus, or the basal forebrain. Fibre tract damage at the level of the fornix has only inconsistently produced amnesia. A patient is reported who suffered a cerebro- vascular accident involving the posterior limb of the left internal capsule that resulted in a persistent and severe disorder of verbal memory. The inferior extent of the lesion effectively disconnected the mesial thalamus from the amygdala and the frontal cortex by disrupting the ventral amygdalofugal and thalamic-frontal pathways as they course through the diencephalon. This case demonstrates that an isolated lesion may cause memory loss without involvement of traditional structures associated with memory and may explain memory disturbances in other white matter disease such as multiple sclerosis and lacunar state. Protected by copyright. Memory loss is currently believed to reflect grey day of his illness the patient was transferred to Shands matter damage of either the mesial temporal lobe,' -4 Teaching Hospital at the University of Florida for further the mesial or the basal forebrain.'0 l evaluation. thalamus,5-9 Examination at that time showed the patient to be awake, Cerebrovascular accidents resulting in memory dys- alert, attentive and fully oriented. -

The Role of the Parabrachial/Kolliker Fuse Respiratory Complex in the Control of Respiration
THE ROLE OF THE PARABRACHIAL/KOLLIKER FUSE RESPIRATORY COMPLEX IN THE CONTROL OF RESPIRATION by JOYCE A. BOON B.Sc. Honors The University of Alberta, 1967 M.Sc. The University of British Columbia, 1970 A THESIS SUBMITTED IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF DOCTOR OF PHILOSOPHY in THE FACULTY OF GRADUATE STUDIES ZOOLOGY THE UNIVERSITY OF BRITISH COLUMBIA DECEMBER 2004 ©Joyce A. Boon, 2004 Abstract: My goal was to explore the role of the parabrachial/Kolliker Fuse region (PBrKF) of the pons in the production of "state-related" changes in breathing in rats. I hypothesized that the effects of changes in cortical activation state on breathing and respiratory sensitivity are relayed from the pontine reticular formation to the respiratory centres of the medulla via the PBrKF. I found that urethane anaesthetized Sprague Dawley rats spontaneously cycled between a cortically desynchronized state (State I) and a cortically synchronized state (State III), which were very similar to awake and slow wave sleep (SWS) states in unanaesthetized animals, based on EEG criteria. Urethane produced no significant respiratory depression or reduction in sensitivity to hypoxia or hypercapnia. However, breathing frequency (TR), tidal volume (VT) and total ventilation (V TOT) all increased on cortical activation, and changes in the relative sensitivity to hypoxia and hypercapnia with changes in state were similar to those seen in unanaesthetized rats. This indicated that the urethane model of sleep and wakefulness could be used to investigate the effects of cortical activation state on respiration. Since NMDA-type glutamate receptor mediated processes in the PBrKF are known to be important in respiratory control, I examined the role of the PBrKF as a relay site for state effects on respiration by blocking neurons with NMDA-type glutamate receptors with MK-801. -

Opioid-Sensitive Brainstem Neurons Separately Modulate Pain and Respiration
OPIOID-SENSITIVE BRAINSTEM NEURONS SEPARATELY MODULATE PAIN AND RESPIRATION By Daniel R. Cleary A DISSERTATION Presented to the Neuroscience Graduate Program and the Oregon Health & Science University School of Medicine in partial fulfillment of the requirements for the degree of Doctor of Philosophy June, 2012 School of Medicine Oregon Health & Science University CERTIFICATE OF APPROVAL _______________________________ This is to certify that the PhD dissertation of Daniel R. Cleary has been approved ____________________________________ Mentor : Mary M. Heinricher, PhD ____________________________________ Committee Chair: Michael C. Andresen, PhD ____________________________________ Member: Nabil J. Alkayed, MD, PhD ____________________________________ Member: Michael M. Morgan, PhD ____________________________________ Member: Shaun F. Morrison, PhD ____________________________________ Member: Susan L. Ingram, PhD TABLE OF CONTENTS Table of contents i List of figures and tables vii List of abbreviations ix Acknowledgements xi Abstract xiii Chapter 1. Introduction 1 1.1 Overview 2 1.2 Rostral ventromedial medulla and the maintenance of homeostasis 3 1.2.1 Convergence of pain modulation and homeostasis 3 1.2.2 Anatomy of brainstem modulation 4 1.2.2.1 RVM in the modulation of nociception 5 1.2.2.2 Respiratory modulation by raphe nuclei 5 1.2.2.3 Thermoregulation via raphe nuclei 7 1.2.2.4 Cardiovascular regulation 8 1.2.3 Specificity of function of RVM neurons 9 1.3 Modulation of pain in the maintenance of homeostasis 9 1.3.1 Anatomy of -
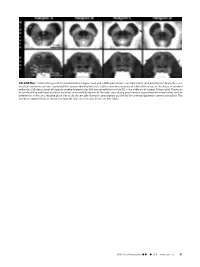
ON-LINE FIG 1. Selected Images of the Caudal Midbrain (Upper Row
ON-LINE FIG 1. Selected images of the caudal midbrain (upper row) and middle pons (lower row) from 4 of 13 total postmortem brains illustrate excellent anatomic contrast reproducibility across individual datasets. Subtle variations are present. Note differences in the shape of cerebral peduncles (24), decussation of superior cerebellar peduncles (25), and spinothalamic tract (12) in the midbrain of subject D (top right). These can be attributed to individual anatomic variation, some mild distortion of the brain stem during procurement at postmortem examination, and/or differences in the axial imaging plane not easily discernable during its prescription parallel to the anterior/posterior commissure plane. The numbers in parentheses in the on-line legends refer to structures in the On-line Table. AJNR Am J Neuroradiol ●:●●2019 www.ajnr.org E1 ON-LINE FIG 3. Demonstration of the dentatorubrothalamic tract within the superior cerebellar peduncle (asterisk) and rostral brain stem. A, Axial caudal midbrain image angled 10° anterosuperior to posteroinferior relative to the ACPC plane demonstrates the tract traveling the midbrain to reach the decussation (25). B, Coronal oblique image that is perpendicular to the long axis of the hippocam- pus (structure not shown) at the level of the ventral superior cerebel- lar decussation shows a component of the dentatorubrothalamic tract arising from the cerebellar dentate nucleus (63), ascending via the superior cerebellar peduncle to the decussation (25), and then enveloping the contralateral red nucleus (3). C, Parasagittal image shows the relatively long anteroposterior dimension of this tract, which becomes less compact and distinct as it ascends toward the thalamus. ON-LINE FIG 2. -

Deep Brain Stimulation in Epilepsy: a Role for Modulation of the Mammillothalamic Tract in Frédéric L
RESEARCH—HUMAN—CLINICAL STUDIES Deep Brain Stimulation in Epilepsy: A Role for Modulation of the Mammillothalamic Tract in Frédéric L. W. V. J. Schaper, MD ∗ ‡§ Birgit R. Plantinga, PhD‡§ Seizure Control? Albert J. Colon, MD, PhD¶|| G. Louis Wagner, MD¶|| Paul Boon, MD, PhD¶||# BACKGROUND: Deep brain stimulation of the anterior nucleus of the thalamus (ANT-DBS) Nadia Blom, MSc‡§ ∗∗ can improve seizure control for patients with drug-resistant epilepsy (DRE). Yet, one cannot Erik D. Gommer, PhD Govert Hoogland, PhD‡§|| overlook the high discrepancy in efficacy among patients, possibly resulting from differ- Linda Ackermans, MD, PhD‡ encesinstimulationsite. RobP.W.Rouhl,MD,PhD∗ §|| Yasin Temel, MD, PhD‡§ OBJECTIVE: To test the hypothesis that stimulation at the junction of the ANT and mammillothalamic tract (ANT-MTT junction) increases seizure control. ∗Department of Neurology, Maast- richt University Medical Center METHODS: The relationship between seizure control and the location of the active (MUMC+), Maastricht, The Nether- contacts to the ANT-MTT junction was investigated in 20 patients treated with ANT-DBS lands; ‡Department of Neurosurgery, Maastricht University Medical Cen- for DRE. Coordinates and Euclidean distance of the active contacts relative to the ANT-MTT ter (MUMC+), Maastricht, The Nether- junction were calculated and related to seizure control. Stimulation sites were mapped by lands; §School for Mental Health and Neuroscience (MHeNS), Maast- modelling the volume of tissue activation (VTA) and generating stimulation heat maps. richt University, Maastricht, The RESULTS: After 1 yr of stimulation, patients had a median 46% reduction in total seizure Netherlands; ¶Academic Center for Epileptology Kempenhaeghe/Maast- frequency, 50% were responders, and 20% of patients were seizure-free. -

Neuroimaging Advances in Deep Brain Stimulation: Review of Indications, Anatomy, and Brain Connectomics
Published August 13, 2020 as 10.3174/ajnr.A6693 REVIEW ARTICLE Neuroimaging Advances in Deep Brain Stimulation: Review of Indications, Anatomy, and Brain Connectomics E.H. Middlebrooks, R.A. Domingo, T. Vivas-Buitrago, L. Okromelidze, T. Tsuboi, J.K. Wong, R.S. Eisinger, L. Almeida, M.R. Burns, A. Horn, R.J. Uitti, R.E. Wharen Jr, V.M. Holanda, and S.S. Grewal ABSTRACT SUMMARY: Deep brain stimulation is an established therapy for multiple brain disorders, with rapidly expanding potential indi- cations. Neuroimaging has advanced the field of deep brain stimulation through improvements in delineation of anatomy, and, more recently, application of brain connectomics. Older lesion-derived, localizationist theories of these conditions have evolved to newer, network-based “circuitopathies,” aided by the ability to directly assess these brain circuits in vivo through the use of advanced neuroimaging techniques, such as diffusion tractography and fMRI. In this review, we use a combination of ultra-high-field MR imaging and diffusion tractography to highlight relevant anatomy for the currently approved indications for deep brain stimulation in the United States: essential tremor, Parkinson disease, drug-resistant epilepsy, dystonia, and obsessive-compulsive disorder. We also review the literature regarding the use of fMRI and diffusion tractography in under- standing the role of deep brain stimulation in these disorders, as well as their potential use in both surgical targeting and de- vice programming. ABBREVIATIONS: AL ¼ ansa lenticularis; ALIC -

Qt59x2b1ds.Pdf
UCLA UCLA Previously Published Works Title Efferent projections of excitatory and inhibitory preBötzinger Complex neurons. Permalink https://escholarship.org/uc/item/59x2b1ds Journal The Journal of comparative neurology, 526(8) ISSN 0021-9967 Authors Yang, Cindy F Feldman, Jack L Publication Date 2018-06-01 DOI 10.1002/cne.24415 Peer reviewed eScholarship.org Powered by the California Digital Library University of California Received: 28 September 2017 | Revised: 4 February 2018 | Accepted: 9 February 2018 DOI: 10.1002/cne.24415 RESEARCH ARTICLE Efferent projections of excitatory and inhibitory preBotzinger€ Complex neurons Cindy F. Yang | Jack L. Feldman Department of Neurobiology, David Geffen School of Medicine, UCLA, Los Angeles, Abstract California 90095-1763 The preBotzinger€ Complex (preBotC),€ a compact medullary region essential for generating normal breathing rhythm and pattern, is the kernel of the breathing central pattern generator (CPG). Exci- Correspondence tatory preBotC€ neurons in rats project to major breathing-related brainstem regions. Here, we Jack L. Feldman, Box 951763, Department € of Neurobiology, David Geffen School of provide a brainstem connectivity map in mice for both excitatory and inhibitory preBotC neurons. Medicine, UCLA, Los Angeles, Using a genetic strategy to label preBotC€ neurons, we confirmed extensive projections of preBotC€ CA 90095-1763. excitatory neurons within the brainstem breathing CPG including the contralateral preBotC,€ Email: [email protected] Botzinger€ Complex (BotC),€ ventral respiratory group, nucleus of the solitary tract, parahypoglossal € € Funding information nucleus, parafacial region (RTN/pFRG or alternatively, pFL/pFV), parabrachial and Kolliker-Fuse A.P. Giannini Foundation and the National nuclei, as well as major projections to the midbrain periaqueductal gray. -

The Three Amnesias
The Three Amnesias Russell M. Bauer, Ph.D. Department of Clinical and Health Psychology College of Public Health and Health Professions Evelyn F. and William L. McKnight Brain Institute University of Florida PO Box 100165 HSC Gainesville, FL 32610-0165 USA Bauer, R.M. (in press). The Three Amnesias. In J. Morgan and J.E. Ricker (Eds.), Textbook of Clinical Neuropsychology. Philadelphia: Taylor & Francis/Psychology Press. The Three Amnesias - 2 During the past five decades, our understanding of memory and its disorders has increased dramatically. In 1950, very little was known about the localization of brain lesions causing amnesia. Despite a few clues in earlier literature, it came as a complete surprise in the early 1950’s that bilateral medial temporal resection caused amnesia. The importance of the thalamus in memory was hardly suspected until the 1970’s and the basal forebrain was an area virtually unknown to clinicians before the 1980’s. An animal model of the amnesic syndrome was not developed until the 1970’s. The famous case of Henry M. (H.M.), published by Scoville and Milner (1957), marked the beginning of what has been called the “golden age of memory”. Since that time, experimental analyses of amnesic patients, coupled with meticulous clinical description, pathological analysis, and, more recently, structural and functional imaging, has led to a clearer understanding of the nature and characteristics of the human amnesic syndrome. The amnesic syndrome does not affect all kinds of memory, and, conversely, memory disordered patients without full-blown amnesia (e.g., patients with frontal lesions) may have impairment in those cognitive processes that normally support remembering.