Diversity and Evolution of the Megaphyll in Euphyllophytes
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Arabidopsis LEAFY COTYLEDON2 Induces Maturation Traits and Auxin Activity: Implications for Somatic Embryogenesis
Arabidopsis LEAFY COTYLEDON2 induces maturation traits and auxin activity: Implications for somatic embryogenesis Sandra L. Stone*, Siobhan A. Braybrook*†, Stephanie L. Paula*‡, Linda W. Kwong*, Jonathan Meuser*§, Julie Pelletier*, Tzung-Fu Hsieh¶, Robert L. Fischer¶, Robert B. Goldbergʈ**, and John J. Harada*†** *Section of Plant Biology, College of Biological Sciences, and †Graduate Program in Plant Biology, University of California, Davis, CA 95616; ¶Department of Plant and Microbial Biology, University of California, Berkeley, CA 94720; and ʈDepartment of Molecular, Cell, and Developmental Biology, University of California, Los Angeles, CA 90024 Contributed by Robert B. Goldberg, December 30, 2007 (sent for review December 16, 2007) LEAFY COTYLEDON2 (LEC2) is a central regulator of embryogenesis INSENSITIVE3 (ABI3) and another LEC protein, FUSCA3 sufficient to induce somatic cells to form embryos when expressed (FUS3), also play critical roles in embryogenesis (11, 12). ectopically. Here, we analyze the cellular processes induced by To gain insight into the mechanisms by which cells change LEC2, a B3 domain transcription factor, that may underlie its ability their fate and become embryogenic, we analyzed postembryonic to promote somatic embryogenesis. We show auxin-responsive 35S:LEC2 plants. We showed previously LEC2 is expressed genes are induced after LEC2 activation in seedlings. Genes en- primarily during seed development and ectopic postembryonic coding enzymes involved in auxin biosynthesis, YUC2 and YUC4, expression of LEC2 induces vegetative cells to undergo somatic are activated within 1 h after induction of LEC2 activity, and YUC4 embryogenesis (8). We hypothesized LEC2 establishes a cellular appears to be a direct transcriptional target of LEC2. We also show environment that promotes embryo development. -

Upregulation of a KN1 Homolog by Transposon Insertion Promotes Leafy Head Development in Lettuce
Upregulation of a KN1 homolog by transposon insertion promotes leafy head development in lettuce Changchun Yua,1, Chenghuan Yana,1, Yuling Liua, Yali Liua, Yue Jiaa, Dean Lavelleb, Guanghui Ana, Weiyi Zhanga, Lei Zhanga, Rongkui Hanb, Robert M. Larkina, Jiongjiong Chena, Richard W. Michelmoreb, and Hanhui Kuanga,2 aKey Laboratory of Horticultural Plant Biology, Ministry of Education, Key Laboratory of Horticultural Crop Biology and Genetic improvement (Central Region), Ministry of Agriculture (MOA), College of Horticulture and Forestry Sciences, Huazhong Agricultural University, 430070 Wuhan, People’s Republic of China; and bGenome Center and Department of Plant Sciences, University of California, Davis, CA 95616 Edited by Martin F. Yanofsky, University of California San Diego, La Jolla, CA, and approved November 4, 2020 (received for review September 19, 2020) Leafy head is a unique type of plant architecture found in some axes. Heading in vegetable crops is considered to be a develop- vegetable crops, with leaves bending inward to form a compact head. mental disorder of leaf dorsoventrality (i.e., incongruous cell di- The genetic and molecular mechanisms underlying leafy head in vision/expansion along the adaxial–abaxial axis). Genes involved in vegetables remain poorly understood. We genetically fine-mapped leaf dorsoventrality include AS1, AS2, miR390, TAS3, KAN, and cloned a major quantitative trait locus controlling heading in YABBY, ARF3, ARF4, PHB,andREV (6–12). Genes associated lettuce. The candidate gene (LsKN1)isahomologofknotted 1 with dorsoventrality are directly or indirectly regulated by key (KN1)fromZea mays. Complementation and CRISPR/Cas9 knockout developmental regulators of leaves, such as members of the experiments confirmed the role of LsKN1 in heading. -

RI Equisetopsida and Lycopodiopsida.Indd
IIntroductionntroduction byby FFrancisrancis UnderwoodUnderwood Rhode Island Equisetopsida, Lycopodiopsida and Isoetopsida Special Th anks to the following for giving permission for the use their images. Robbin Moran New York Botanical Garden George Yatskievych and Ann Larson Missouri Botanical Garden Jan De Laet, plantsystematics.org Th is pdf is a companion publication to Rhode Island Equisetopsida, Lycopodiopsida & Isoetopsida at among-ri-wildfl owers.org Th e Elfi n Press 2016 Introduction Formerly known as fern allies, Horsetails, Club-mosses, Fir-mosses, Spike-mosses and Quillworts are plants that have an alternate generation life-cycle similar to ferns, having both sporophyte and gametophyte stages. Equisetopsida Horsetails date from the Devonian period (416 to 359 million years ago) in earth’s history where they were trees up to 110 feet in height and helped to form the coal deposits of the Carboniferous period. Only one genus has survived to modern times (Equisetum). Horsetails Horsetails (Equisetum) have jointed stems with whorls of thin narrow leaves. In the sporophyte stage, they have a sterile and fertile form. Th ey produce only one type of spore. While the gametophytes produced from the spores appear to be plentiful, the successful reproduction of the sporophyte form is low with most Horsetails reproducing vegetatively. Lycopodiopsida Lycopodiopsida includes the clubmosses (Dendrolycopodium, Diphasiastrum, Lycopodiella, Lycopodium , Spinulum) and Fir-mosses (Huperzia) Clubmosses Clubmosses are evergreen plants that produce only microspores that develop into a gametophyte capable of producing both sperm and egg cells. Club-mosses can produce the spores either in leaf axils or at the top of their stems. Th e spore capsules form in a cone-like structures (strobili) at the top of the plants. -

New Paleobotanical Data on the Portuguese Pennsylvanian (Douro Carboniferous Basin, NW Portugal)
Versão online: http://www.lneg.pt/iedt/unidades/16/paginas/26/30/185 Comunicações Geológicas (2014) 101, Especial I, 409-414 IX CNG/2º CoGePLiP, Porto 2014 ISSN: 0873-948X; e-ISSN: 1647-581X New paleobotanical data on the Portuguese Pennsylvanian (Douro Carboniferous Basin, NW Portugal) Novos dados paleobotânicos do Pensilvaniano português (Bacia Carbonífera do Douro, NW Portugal) P. Correia1*, Z. Šimůnek2, J. Pšenička3, A. A. Sá4,5, R. Domingos6, A. Carneiro7, D. Flores1,7 Artigo Curto Short Article © 2014 LNEG – Laboratório Nacional de Geologia e Energia IP Abstract: This paper describes nine new macrofloral taxa from 1. Introduction Douro Carboniferous Basin (lower Gzhelian) of Portugal. The plant assemblage is mainly composed by pteridophylls (Sphenopteriss The fossil flora of Carboniferous of Portugal is still little arberi Kidston, Sphenopteris fayoli Zeiller, Sphenopteris tenuis known. The new megafloral occurrences recently found in Schenk, Odontopteris schlotheimii Brongniart), sphenopsids the Upper Pennsylvanian strata of Douro Carboniferous (Annularia spicata Gutbier, Stellotheca robusta (Feistmantel) Basin (DCB) provide new and important data about Surange and Prakash, Calamostachys grandis Zeiller (Jongmans) and Calamostachys calathifera Sterzel) besides the gymnosperm paleobotanical richness and diversity of the Paleozoic Cordaites foliolatus Grand`Eury. The new data provide a better floras of Portugal offering more information to previous understating of the knowledge of Late Carboniferous floras of researches reported from diverse localities and by different Portugal, showing the high plant diversity of Gzhelian floras, when authors (e.g. Wenceslau de Lima, Bernardino António considerable changes in paleogeography and climate dynamics are Gomes, Carlos Ribeiro, Carríngton da Costa, Carlos evidenced in Euramerican floristic assemblages. -

The Origin and Early Evolution of Vascular Plant Shoots and Leaves Rstb.Royalsocietypublishing.Org C
Downloaded from http://rstb.royalsocietypublishing.org/ on January 22, 2018 The origin and early evolution of vascular plant shoots and leaves rstb.royalsocietypublishing.org C. Jill Harrison 1 and Jennifer L. Morris 2 1School of Biological Sciences, and 2School of Earth Sciences, University of Bristol, 24 Tyndall Avenue, Bristol BS8 1TQ, UK Review CJH, 0000-0002-5228-600X; JLM, 0000-0002-7453-3841 Cite this article: Harrison CJ, Morris JL. 2017 The morphology of plant fossils from the Rhynie chert has generated long- standing questions about vascular plant shoot and leaf evolution, for The origin and early evolution of vascular plant instance, which morphologies were ancestral within land plants, when did shoots and leaves. Phil. Trans. R. Soc. B 373 : vascular plants first arise and did leaves have multiple evolutionary origins? 20160496. Recent advances combining insights from molecular phylogeny, palaeobotany http://dx.doi.org/10.1098/rstb.2016.0496 and evo–devo research address these questions and suggest the sequence of morphological innovation during vascular plant shoot and leaf evolution. The evidence pinpoints testable developmental and genetic hypotheses relat- Accepted: 11 August 2017 ing to the origin of branching and indeterminate shoot architectures prior to the evolution of leaves, and demonstrates underestimation of polyphyly in One contribution of 18 to a discussion meeting the evolution of leaves from branching forms in ‘telome theory’ hypotheses issue ‘The Rhynie cherts: our earliest terrestrial of leaf evolution. This review discusses fossil, developmental and genetic ecosystem revisited’. evidence relating to the evolution of vascular plant shoots and leaves in a phylogenetic framework. This article is part of a discussion meeting issue ‘The Rhynie cherts: our Subject Areas: earliest terrestrial ecosystem revisited’. -

Getting to the Roots: a Developmental Genetic View of Root Anatomy and Function from Arabidopsis to Lycophytes
fpls-09-01410 September 21, 2018 Time: 17:3 # 1 REVIEW published: 25 September 2018 doi: 10.3389/fpls.2018.01410 Getting to the Roots: A Developmental Genetic View of Root Anatomy and Function From Arabidopsis to Lycophytes Frauke Augstein and Annelie Carlsbecker* Department of Organismal Biology, Physiological Botany and Linnean Centre for Plant Biology in Uppsala, Uppsala University, Uppsala, Sweden Roots attach plants to the ground and ensure efficient and selective uptake of water and nutrients. These functions are facilitated by the morphological and anatomical structures of the root, formed by the activity of the root apical meristem (RAM) and consecutive patterning and differentiation of specific tissues with distinct functions. Despite the importance of this plant organ, its evolutionary history is not clear, but fossils suggest that roots evolved at least twice, in the lycophyte (clubmosses and their allies) and in the euphyllophyte (ferns and seed plants) lineages. Both lycophyte and euphyllophyte roots grow indeterminately by the action of an apical meristem, which is protected by a root cap. They produce root hairs, and in most species the vascular stele is Edited by: guarded by a specialized endodermal cell layer. Hence, most of these traits must have Annette Becker, evolved independently in these lineages. This raises the question if the development Justus Liebig Universität Gießen, Germany of these apparently analogous tissues is regulated by distinct or homologous genes, Reviewed by: independently recruited from a common ancestor of lycophytes and euphyllophytes. Hongchang Cui, Currently, there are few studies of the genetic and molecular regulation of lycophyte Florida State University, United States and fern roots. -
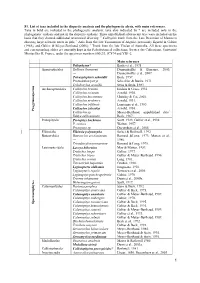
S1. List of Taxa Included in the Disparity Analysis and the Phylogenetic Alysis, with Main References
S1. List of taxa included in the disparity analysis and the phylogenetic alysis, with main references. Taxa in bold are included in the phylogenetic analysis; taxa also indicated by * are included only in the phylogenetic analysis and not in the disparity analysis. Three unpublished arborescent taxa were included on the basis that they showed additional anatomical diversity. 1 Callixylon trunk from the Late Devonian of Marrocco showing large sclerotic nests in pith; 2 Axis from the late Tournaisian of Algeria, previously figured in Galtier (1988), and Galtier & Meyer-Berthaud (2006); 3 Trunk from the late Viséan of Australia. All these specimens and corresponding slides are currently kept in the Paleobotanical collections, Service des Collections, Université Montpellier II, France, under the specimen numbers 600/2/3, JC874 and YB1-2. Main reference Psilophyton* Banks et al., 1975 Aneurophytales Rellimia thomsonii Dannenhoffer & Bonamo, 2003; --- Dannenhoffer et al., 2007. Tetraxylopteris schmidtii Beck, 1957. Proteokalon petryi Scheckler & Banks, 1971. Triloboxylon arnoldii Stein & Beck, 1983. s m Archaeopteridales Callixylon brownii Hoskin & Cross, 1951. r e Callixylon erianum Arnold, 1930. p s o Callixylon huronensis Chitaley & Cai, 2001. n Callixylon newberry Arnold, 1931. m y g Callixylon trifilievii Lemoigne et al., 1983. o r Callixylon zalesskyi Arnold, 1930. P Callixylon sp. Meyer-Berthaud, unpublished data1. Eddya sullivanensis Beck, 1967. Protopityales Protopitys buchiana Scott, 1923; Galtier et al., 1998. P. scotica Walton, 1957. Protopitys sp. Decombeix et al., 2005. Elkinsiales Elkinsia polymorpha Serbet & Rothwell, 1992. Buteoxylales Buteoxylon gordonianum Barnard &Long, 1973; Matten et al., --- 1980. Triradioxylon primaevum Barnard & Long, 1975. Lyginopteridales Laceya hibernica May & Matten, 1983. Tristichia longii Galtier, 1977. -

<I>Equisetum Giganteum</I>
Florida International University FIU Digital Commons FIU Electronic Theses and Dissertations University Graduate School 3-24-2009 Ecophysiology and Biomechanics of Equisetum Giganteum in South America Chad Eric Husby Florida International University, [email protected] DOI: 10.25148/etd.FI10022522 Follow this and additional works at: https://digitalcommons.fiu.edu/etd Recommended Citation Husby, Chad Eric, "Ecophysiology and Biomechanics of Equisetum Giganteum in South America" (2009). FIU Electronic Theses and Dissertations. 200. https://digitalcommons.fiu.edu/etd/200 This work is brought to you for free and open access by the University Graduate School at FIU Digital Commons. It has been accepted for inclusion in FIU Electronic Theses and Dissertations by an authorized administrator of FIU Digital Commons. For more information, please contact [email protected]. FLORIDA INTERNATIONAL UNIVERSITY Miami, Florida ECOPHYSIOLOGY AND BIOMECHANICS OF EQUISETUM GIGANTEUM IN SOUTH AMERICA A dissertation submitted in partial fulfillment of the requirements for the degree of DOCTOR OF PHILOSOPHY in BIOLOGY by Chad Eric Husby 2009 To: Dean Kenneth Furton choose the name of dean of your college/school College of Arts and Sciences choose the name of your college/school This dissertation, written by Chad Eric Husby, and entitled Ecophysiology and Biomechanics of Equisetum Giganteum in South America, having been approved in respect to style and intellectual content, is referred to you for judgment. We have read this dissertation and recommend that it be approved. _______________________________________ Bradley C. Bennett _______________________________________ Jack B. Fisher _______________________________________ David W. Lee _______________________________________ Leonel Da Silveira Lobo O'Reilly Sternberg _______________________________________ Steven F. Oberbauer, Major Professor Date of Defense: March 24, 2009 The dissertation of Chad Eric Husby is approved. -

Earliest Record of Megaphylls and Leafy Structures, and Their Initial Diversification
Review Geology August 2013 Vol.58 No.23: 27842793 doi: 10.1007/s11434-013-5799-x Earliest record of megaphylls and leafy structures, and their initial diversification HAO ShouGang* & XUE JinZhuang Key Laboratory of Orogenic Belts and Crustal Evolution, School of Earth and Space Sciences, Peking University, Beijing 100871, China Received January 14, 2013; accepted February 26, 2013; published online April 10, 2013 Evolutionary changes in the structure of leaves have had far-reaching effects on the anatomy and physiology of vascular plants, resulting in morphological diversity and species expansion. People have long been interested in the question of the nature of the morphology of early leaves and how they were attained. At least five lineages of euphyllophytes can be recognized among the Early Devonian fossil plants (Pragian age, ca. 410 Ma ago) of South China. Their different leaf precursors or “branch-leaf com- plexes” are believed to foreshadow true megaphylls with different venation patterns and configurations, indicating that multiple origins of megaphylls had occurred by the Early Devonian, much earlier than has previously been recognized. In addition to megaphylls in euphyllophytes, the laminate leaf-like appendages (sporophylls or bracts) occurred independently in several dis- tantly related Early Devonian plant lineages, probably as a response to ecological factors such as high atmospheric CO2 concen- trations. This is a typical example of convergent evolution in early plants. Early Devonian, euphyllophyte, megaphyll, leaf-like appendage, branch-leaf complex Citation: Hao S G, Xue J Z. Earliest record of megaphylls and leafy structures, and their initial diversification. Chin Sci Bull, 2013, 58: 27842793, doi: 10.1007/s11434- 013-5799-x The origin and evolution of leaves in vascular plants was phology and evolutionary diversification of early leaves of one of the most important evolutionary events affecting the basal euphyllophytes remain enigmatic. -
Ferns of the National Forests in Alaska
Ferns of the National Forests in Alaska United States Forest Service R10-RG-182 Department of Alaska Region June 2010 Agriculture Ferns abound in Alaska’s two national forests, the Chugach and the Tongass, which are situated on the southcentral and southeastern coast respectively. These forests contain myriad habitats where ferns thrive. Most showy are the ferns occupying the forest floor of temperate rainforest habitats. However, ferns grow in nearly all non-forested habitats such as beach meadows, wet meadows, alpine meadows, high alpine, and talus slopes. The cool, wet climate highly influenced by the Pacific Ocean creates ideal growing conditions for ferns. In the past, ferns had been loosely grouped with other spore-bearing vascular plants, often called “fern allies.” Recent genetic studies reveal surprises about the relationships among ferns and fern allies. First, ferns appear to be closely related to horsetails; in fact these plants are now grouped as ferns. Second, plants commonly called fern allies (club-mosses, spike-mosses and quillworts) are not at all related to the ferns. General relationships among members of the plant kingdom are shown in the diagram below. Ferns & Horsetails Flowering Plants Conifers Club-mosses, Spike-mosses & Quillworts Mosses & Liverworts Thirty of the fifty-four ferns and horsetails known to grow in Alaska’s national forests are described and pictured in this brochure. They are arranged in the same order as listed in the fern checklist presented on pages 26 and 27. 2 Midrib Blade Pinnule(s) Frond (leaf) Pinna Petiole (leaf stalk) Parts of a fern frond, northern wood fern (p. -

Embryophytic Sporophytes in the Rhynie and Windyfield Cherts
Transactions of the Royal Society of Edinburgh: Earth Sciences http://journals.cambridge.org/TRE Additional services for Transactions of the Royal Society of Edinburgh: Earth Sciences: Email alerts: Click here Subscriptions: Click here Commercial reprints: Click here Terms of use : Click here Embryophytic sporophytes in the Rhynie and Windyeld cherts Dianne Edwards Transactions of the Royal Society of Edinburgh: Earth Sciences / Volume 94 / Issue 04 / December 2003, pp 397 - 410 DOI: 10.1017/S0263593300000778, Published online: 26 July 2007 Link to this article: http://journals.cambridge.org/abstract_S0263593300000778 How to cite this article: Dianne Edwards (2003). Embryophytic sporophytes in the Rhynie and Windyeld cherts. Transactions of the Royal Society of Edinburgh: Earth Sciences, 94, pp 397-410 doi:10.1017/S0263593300000778 Request Permissions : Click here Downloaded from http://journals.cambridge.org/TRE, IP address: 131.251.254.13 on 25 Feb 2014 Transactions of the Royal Society of Edinburgh: Earth Sciences, 94, 397–410, 2004 (for 2003) Embryophytic sporophytes in the Rhynie and Windyfield cherts Dianne Edwards ABSTRACT: Brief descriptions and comments on relationships are given for the seven embryo- phytic sporophytes in the cherts at Rhynie, Aberdeenshire, Scotland. They are Rhynia gwynne- vaughanii Kidston & Lang, Aglaophyton major D. S. Edwards, Horneophyton lignieri Barghoorn & Darrah, Asteroxylon mackiei Kidston & Lang, Nothia aphylla Lyon ex Høeg, Trichopherophyton teuchansii Lyon & Edwards and Ventarura lyonii Powell, Edwards & Trewin. The superb preserva- tion of the silica permineralisations produced in the hot spring environment provides remarkable insights into the anatomy of early land plants which are not available from compression fossils and other modes of permineralisation. -

Scouring-Rush Horsetail Scientific Name: Equisetum Hyemale Order
Common Name: Scouring-rush Horsetail Scientific Name: Equisetum hyemale Order: Equisetales Family: Equisetaceae Wetland Plant Status: Facultative Ecology & Description Scouring-rush horsetail is an evergreen, perennial plant that completes a growing season in two years. At maturity, scouring-rush horsetail usually averages 3 feet in height but can be range anywhere from 2 to 5 feet. It can survive in a variety of environments. One single plant can spread 6 feet in diameter. It has cylindrical stems that averages a third of an inch in diameter. Noticeably spotted are the jointed unions that are located down the plant. The stems are hollow and don’t branch off into additional stems. Also, scouring- rush horsetail has rough ridges that run longitudinal along the stem. Although not covered in leaves, tiny leaves are joined together around the stem which then forms a black or green band, or sheath at each individual joint on the stem. This plant has an enormous root system that can reach 6 feet deep and propagates in two ways: rhizomes and spores. Incredibly, due to the fact that this plant is not full of leaves, it is forced to photosynthesize through the stem rather than leaves. Habitat Scouring-rush horsetail is highly tolerant of tough conditions. It can survive and thrive in full sun or part shade and can successfully grow in a variety of soil types. It can also grow in moderate to wet soils, and can survive in up to 4 inches of water. Distribution Scouring-rush horsetail can be found throughout the United States, Eurasia, and Canada.