Dai Et Al-MS-Recovery-Accepted.Pdf
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Fenhe (Fen He)
China ―10 Fenhe (Fen He) Map of River Table of Basic Data Name(s): Fenhe (in Huanghe River) Serial No. : China-10 Location: Shanxi Province, Northern China N 35° 34' ~ 38° 53' E 110° 34' ~ 111° 58' Area: 39,471 km2 Length of the main stream: 694 km Origin: Mt. Guancen (2,147 m) Highest point: Mt.Woyangchang (2,603 m) Outlet: Huanghe Lowest point: 365 (m) Main geological features: Hard layered clastic rocks, Group of hard massive metamorphic rocks Main tributaries: Lanhe (1,146 km2), Xiaohe (3,894 km2), Changyuanhe (2,274 km2), Wenyuhe (3,979 km2), Honganjiandong (1,123 km2), Huihe (2,060 km2) Main lakes: ------------ 6 3 6 3 Main reservoirs: Fenhe (723×10 m , 1961), Wenyuhe (105×10 m , 1970), Fenhe II (under construction) Mean annual precipitation: 493.2 mm (1971 ~ 1990) (basin average) Mean annual runoff: 48.7 m3/s at Hejin (38,728 km2) (1971 ~ 1990) Population: 3,410,700 (1998) Main cities: Taiyuan, Linfen, Yuci, Houma Land use: Forest (24%), Rice paddy (2%), Other agriculture (29%), Water surface (2%),Urban (6%), Uncultivated land (20%), Qthers (17%) 3 China ―10 1. General Description The Fenhe is a main tributary of The Yellow River. It is located in the middle of Shanxi province. The main river originates from northwest of Mt. Guanqing and flows from north to south before joining the Yellow River at Wanrong county. It flows through 18 counties and cities, including Ningwu, Jinle, Loufan, Gujiao, and Taiyuan. The catchment area is 39,472 km2 and the main channel length is 693 km. -

Impact of Coal Mining on Karst Water System in North China
Available online at www.sciencedirect.com Procedia Earth and Planetary Science 3 ( 2011 ) 293 – 302 2011 Xican International Conference on Fine Geological Exploration and Groundwater & Gas Hazards Control in Coal Mines Impact of Coal Mining on Karst Water System in North China Xiangqing Fang*, Yaojun Fu Hydrogeology Bureau of China Nat ion al Administration of Coal Geology, Handan 056004, China Abstract Based on a large number of data, the paper analysed the influence factors of coal mining for the karst water system in north China, used analytic hierarchy process (AHP) for evaluation of the effect of coal mining on the karst water system, and divided influence degree subareas of State-planed 21 coal mining areas. We also come up with some suggestions on prevention and control measures of principle according to the different influence degrees. ©© 20112011 Published Published by by Elsevier Elsevier Ltd. Ltd. Selection Selection and and/or peer-review peer-review under under responsibility responsibility of China of Xi’an Coal Research Society Institute of China Coal Technology & Engineering Group Corp Keywords: karst-water system in north China, influence degree, prevention and control measures; North China type coalfield and the karst water system in the north are inseparable, there exists three superposition relationships [1]: monoclinal structure, synclinal structure and block-faulting structure. Karst water system in the north is characterized by large scales, numerous components of water resources, the complexity of transformation between water resources, coexistence of water and coal and so on. The karst water resource in the north is not only important water resource, but also is threatening the coal resources. -

People's Republic of China: Shanxi Road Development II Project
Completion Report Project Number: 34097 Loan Number: 1967 August 2008 People’s Republic of China: Shanxi Road Development II Project CURRENCY EQUIVALENTS Currency Unit – yuan (CNY) At Appraisal At Project Completion (14 November 2002) (as of 6 March 2008) CNY1.00 = $0.1208 $0.14047 $1.00 = CNY8.277 CNY7.119 ABBREVIATIONS AADT – average annual daily traffic ADB – Asian Development Bank CSE – chief supervision engineer CSEO – chief supervision engineer office DCSE – deputy chief supervision engineer EIA – environmental impact assessment EIRR – economic internal rate of return FIRR – financial internal rate of return GDP – gross domestic product HDM-4 – highway design and maintenance standards model, version 4 ICB – international competitive bidding IDC – interest and other charges during construction IEE – initial environmental examination IRI – international roughness index MOC – Ministry of Communications NCB – national competitive bidding NTHS – national trunk highway system O&M – operation and maintenance PCR – project completion review PPMS – project performance management system PRC – People’s Republic of China PRIS – poverty reduction impact study PRMP – poverty reduction monitoring program REO – resident engineer office RP – resettlement plan SCD – Shanxi Communications Department SCF – standard conversion factor SEIA – summary environmental impact assessment SEPA – State Environment Protection Administration SFB – Shanxi Finance Bureau SHEC – Shanxi Hou-yu Expressway Construction Company Limited SKCC – Shaanxi Kexin Consultant Company SPG – Shanxi provincial government VOC – vehicle operating cost YWNR – Yuncheng Wetlands Nature Reserve WEIGHTS AND MEASURES mu – A traditional land area measurement, it is equivalent to 666.66 square meters, or 0.1647 acres, or 0.066 of a hectare. m/km – meters per kilometer mg/m3 – milligram per meter cube p.a. -
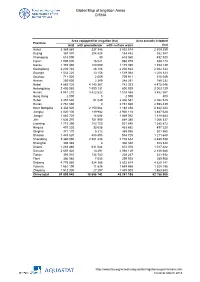
Global Map of Irrigation Areas CHINA
Global Map of Irrigation Areas CHINA Area equipped for irrigation (ha) Area actually irrigated Province total with groundwater with surface water (ha) Anhui 3 369 860 337 346 3 032 514 2 309 259 Beijing 367 870 204 428 163 442 352 387 Chongqing 618 090 30 618 060 432 520 Fujian 1 005 000 16 021 988 979 938 174 Gansu 1 355 480 180 090 1 175 390 1 153 139 Guangdong 2 230 740 28 106 2 202 634 2 042 344 Guangxi 1 532 220 13 156 1 519 064 1 208 323 Guizhou 711 920 2 009 709 911 515 049 Hainan 250 600 2 349 248 251 189 232 Hebei 4 885 720 4 143 367 742 353 4 475 046 Heilongjiang 2 400 060 1 599 131 800 929 2 003 129 Henan 4 941 210 3 422 622 1 518 588 3 862 567 Hong Kong 2 000 0 2 000 800 Hubei 2 457 630 51 049 2 406 581 2 082 525 Hunan 2 761 660 0 2 761 660 2 598 439 Inner Mongolia 3 332 520 2 150 064 1 182 456 2 842 223 Jiangsu 4 020 100 119 982 3 900 118 3 487 628 Jiangxi 1 883 720 14 688 1 869 032 1 818 684 Jilin 1 636 370 751 990 884 380 1 066 337 Liaoning 1 715 390 783 750 931 640 1 385 872 Ningxia 497 220 33 538 463 682 497 220 Qinghai 371 170 5 212 365 958 301 560 Shaanxi 1 443 620 488 895 954 725 1 211 648 Shandong 5 360 090 2 581 448 2 778 642 4 485 538 Shanghai 308 340 0 308 340 308 340 Shanxi 1 283 460 611 084 672 376 1 017 422 Sichuan 2 607 420 13 291 2 594 129 2 140 680 Tianjin 393 010 134 743 258 267 321 932 Tibet 306 980 7 055 299 925 289 908 Xinjiang 4 776 980 924 366 3 852 614 4 629 141 Yunnan 1 561 190 11 635 1 549 555 1 328 186 Zhejiang 1 512 300 27 297 1 485 003 1 463 653 China total 61 899 940 18 658 742 43 241 198 52 -

Investigation on the Remaining Situation of West Post Road in the Ancient City of Jinyang—Taking the Ancient Village of Zhoujiazhuang, the First Station in The
2018 International Conference on Advanced Chemical Engineering and Environmental Sustainability (ICACEES 2018) ISBN: 978-1-60595-571-1 Investigation on the Remaining Situation of West Post Road in the Ancient City of Jinyang—Taking the Ancient Village of Zhoujiazhuang, The First Station in the West of Taiyuan as an Example Chong-En Wang and Wen Bin Fan ABSTRACT Through the field investigation, site visit and other methods, this paper analyzes the ancient village of Zhoujiazhuang, Jinyuan District, Taiyuan City, Shanxi Province. It is found that the ancient village is located in the ancient post road when going out of the ancient city of Jinyang in the west, and it is a very important link in the post road. There are many historical and cultural relics around the ancient village. Through the collection and arrangement of the building information and documents, the paper analyzes the surrounding environment and architectural layout of the ancient village, so as to provide the basis for the protection and utilization of the traditional village as well as the sustainable development strategy.1 KEYWORDS The Ancient Village of Zhoujiazhuang, Fengyu Valley; The Ancient Post Road, Engraved Rock. INTRODUCTION In ancient times, Taiyuan area was surrounded by mountains from east, west to the north, so the trade between inside and outside as well as the deployment of 1Chong en Wang, College of Architecture and Civil Engineering, TUT, Taiyuan 030024, China Wen bin Fan, College of Architecture and Civil Engineering, TUT, Taiyuan 030024,China E-Mail:[email protected] 346 military hubs were mostly through the valley between the two mountains. -

The Fen River in Taiyuan, China: Ecology, Revitalization, and Urban Culture
The Fen River in Taiyuan, China: Ecology, Revitalization, and Urban Culture Matthias Falke Location of Taiyuan, Shanxi province, and the Fen River Basin This map was created by Matthias Falke in 2016, using Arc Map and the basemap layer World Topographic Map; map materials are from Openstreetmap contributors. This work is licensed under a Creative Commons Attribution-ShareAlike 2.0 Generic License . Shanxi (山西) and its fertile loess-covered landscapes are also known as the cradle of China’s civilization. The 716-km-long Fen River (汾河) or Mother River (母亲河) drains most of the province via the basin of Taiyuan. The river’s stunning scenery, once the subject of poetry during the Jin (金朝, 1125–1234) and Yuan (元朝, 1279–1368) dynasties, quickly deteriorated after industrialization in the late 1950s. The construction of dams, extensive irrigation of farmland, and wastewater discharge severely impacted the river’s ecosystem. From 1956 to 2013, the average surface runoff fell from 2.65 billion m³ to 1.33 m³. Moreover, overexploitation of groundwater dropped the groundwater level in the Fen River basin by 81.4 meters. Source URL: http://www.environmentandsociety.org/node/7679 Print date: 09 July 2019 11:15:32 Falke, Matthias. "The Fen River in Taiyuan, China: Ecology, Revitalization, and Urban Culture ." Arcadia (Autumn 2016), no. 17. View of four cokeries in the Gujiao area with an annual capacity of Traditional dwelling in the rural part of Gujiao 3.8 million metric tons per year before their integration into a Photo taken by Harald Zepp, 2010. combined coke processing facility This work is used by permission of the copyright holder. -

Distribution, Genetic Diversity and Population Structure of Aegilops Tauschii Coss. in Major Whea
Supplementary materials Title: Distribution, Genetic Diversity and Population Structure of Aegilops tauschii Coss. in Major Wheat Growing Regions in China Table S1. The geographic locations of 192 Aegilops tauschii Coss. populations used in the genetic diversity analysis. Population Location code Qianyuan Village Kongzhongguo Town Yancheng County Luohe City 1 Henan Privince Guandao Village Houzhen Town Liantian County Weinan City Shaanxi 2 Province Bawang Village Gushi Town Linwei County Weinan City Shaanxi Prov- 3 ince Su Village Jinchengban Town Hancheng County Weinan City Shaanxi 4 Province Dongwu Village Wenkou Town Daiyue County Taian City Shandong 5 Privince Shiwu Village Liuwang Town Ningyang County Taian City Shandong 6 Privince Hongmiao Village Chengguan Town Renping County Liaocheng City 7 Shandong Province Xiwang Village Liangjia Town Henjin County Yuncheng City Shanxi 8 Province Xiqu Village Gujiao Town Xinjiang County Yuncheng City Shanxi 9 Province Shishi Village Ganting Town Hongtong County Linfen City Shanxi 10 Province 11 Xin Village Sansi Town Nanhe County Xingtai City Hebei Province Beichangbao Village Caohe Town Xushui County Baoding City Hebei 12 Province Nanguan Village Longyao Town Longyap County Xingtai City Hebei 13 Province Didi Village Longyao Town Longyao County Xingtai City Hebei Prov- 14 ince 15 Beixingzhuang Town Xingtai County Xingtai City Hebei Province Donghan Village Heyang Town Nanhe County Xingtai City Hebei Prov- 16 ince 17 Yan Village Luyi Town Guantao County Handan City Hebei Province Shanqiao Village Liucun Town Yaodu District Linfen City Shanxi Prov- 18 ince Sabxiaoying Village Huqiao Town Hui County Xingxiang City Henan 19 Province 20 Fanzhong Village Gaosi Town Xiangcheng City Henan Province Agriculture 2021, 11, 311. -

Shanxi Small Cities and Towns Development Demonstration Sector Project
Environmental Assessment Report Summary Environmental Impact Assessment Project Number: 42383 October 2008 People’s Republic of China: Shanxi Small Cities and Towns Development Demonstration Sector Project Prepared by the Shanxi provincial government for the Asian Development Bank (ADB). The summary environmental impact assessment is a document of the borrower. The views expressed herein do not necessarily represent those of ADB’s Board of Directors, Management, or staff, and may be preliminary in nature. CURRENCY EQUIVALENTS (as of 17 October 2008) Currency Unit – yuan (CNY) CNY1.00 = $0.1461 $1.00 = CNY6.8435 ABBREVIATIONS ADB – Asian Development Bank BOD5 – 5-day biochemical oxygen demand ClO2 – chlorine dioxide CO2 – carbon dioxide CODCr – chemical oxygen demand determined through the dichromate reflux method CSC – construction supervision company DMF – design and monitoring framework EA – executing agency EAMF – environmental assessment and management framework EIA – environmental impact assessment EMC – environmental management consultant EMP – environmental management plan EPB – environmental protection bureau FSR – feasibility study report GDP – gross domestic product GHG – greenhouse gas HDPE – high-density polyethylene IA – implementing agency LDI – local design institute MSW – municipal solid waste NH3-N – ammonia nitrogen NOx – nitrogen oxides O&M – operation and maintenance pH – a unit of acidity PM10 – particulate matter ≤10 micrometers in diameter PMO – project management office PPTA – project preparatory technical assistance -

Resource-Based City Type and Reforming Strategy Discussion and Research
2016 4th International Conference on Advances in Social Science, Humanities, and Management (ASSHM 2016) ISBN: 978-1-60595-412-7 Resource-based City Type and Reforming Strategy Discussion and Research Junwei Xing1 Abstract The resource-based city can divides according to the Israeli resource type and development phase two big standards. The resource-based city economic transformation strategy first should from the macroscopic level, seek for the regional economic development the new superiority, next should act according to the new regional development favorable condition that establishes the pattern of industrial transformation from the microscopic level. Keywords. Resource-based City; Type; Reforming Strategy. 1. INTRODUCTION The resource-based city is our country important foundation energy and raw material supplying place, development display excessively significant role to our country national economy. But as a result of the non-renewability of finiteness and resources of resource, the resource-based city sooner or later must face the issue of economic transformation, otherwise possibly enters the winter. At present, a considerable number of resource-based cities in China have been facing a series of economic and social problems such as resource depletion, economic recession, environmental degradation, unemployment and the increase of the poor population. These resource-based cities have become the problem areas in which regional contradictions are concentrated. Therefore, how to guide these cities to successfully transform and achieve sustainable development has become an important topic of concern for both academia and government. 2. Classification of Resource-based Cities Resource based cities are mainly built up by the development of resources. Although the overall pattern of urban growth and development is similar, because of the differences of the economic development factors such as natural resources, geographical conditions and other economic development factors, the difference between different resource cities is very large. -

Minimum Wage Standards in China August 11, 2020
Minimum Wage Standards in China August 11, 2020 Contents Heilongjiang ................................................................................................................................................. 3 Jilin ............................................................................................................................................................... 3 Liaoning ........................................................................................................................................................ 4 Inner Mongolia Autonomous Region ........................................................................................................... 7 Beijing......................................................................................................................................................... 10 Hebei ........................................................................................................................................................... 11 Henan .......................................................................................................................................................... 13 Shandong .................................................................................................................................................... 14 Shanxi ......................................................................................................................................................... 16 Shaanxi ...................................................................................................................................................... -

A New Griesbachian–Dienerian (Induan, Early Triassic) Ammonoid Fauna from Gujiao, South China
Journal of Paleontology, 93(1), 2019, p. 48–71 Copyright © 2018, The Paleontological Society 0022-3360/15/0088-0906 doi: 10.1017/jpa.2018.46 A new Griesbachian–Dienerian (Induan, Early Triassic) ammonoid fauna from Gujiao, South China Xu Dai,1 Haijun Song,1 Arnaud Brayard,2 and David Ware3 1State Key Laboratory of Biogeology and Environmental Geology, School of Earth Sciences, China University of Geosciences, Wuhan 430074, China 〈[email protected]〉, 〈[email protected]〉 2Biogéosciences, UMR 6282, CNRS, Université Bourgogne Franche-Comté, 6 Boulevard Gabriel, 21000 Dijon, France 〈[email protected]〉 3Museum für Naturkunde, Leibniz Institute for Evolution and Biodiversity Science, Invalidenstrasse 43, 10115 Berlin, Germany 〈[email protected]〉 Abstract.—Bed-by-bed sampling of the lower portion of the Daye Formation at Gujiao, Guizhou Province, South China, yielded new Griesbachian–Dienerian (Induan, Early Triassic) ammonoid faunas showing a new regional Induan ammonoid succession. This biostratigraphic scheme includes in chronological order the late Griesbachian Ophiceras medium and Jieshaniceras guizhouense beds, and the middle Dienerian Ambites radiatus bed. The latter is recognized for the first time as a separate biozone in South China. Eight genera and 13 species are identified, includ- ing one new species, Mullericeras gujiaoense n. sp. The new data show that a relatively high level of ammonoid taxonomic richness occurred rather rapidly after the Permian/Triassic mass extinction in the late Griesbachian, echo- ing similar observations in other basins, such as in the Northern Indian Margin. UUID: http://zoobank.org/a24a3387-f3dd-4da4-a134-84372352a63d Introduction Early Triassic (Brayard et al., 2006, 2009a; Brühwiler et al., 2010a; Ware et al., 2015). -

Natural Resources, Rent Dependence, and Public Goods Provision in China: Evidence from Shanxi’S County-Level Governments Yuyi Zhuang* and Guang Zhang
Zhuang and Zhang The Journal of Chinese Sociology (2016) 3:20 The Journal of DOI 10.1186/s40711-016-0040-3 Chinese Sociology RESEARCH Open Access Natural resources, rent dependence, and public goods provision in China: evidence from Shanxi’s county-level governments Yuyi Zhuang* and Guang Zhang * Correspondence: [email protected] Abstract School of Public Affairs, Xiamen University, Xiamen, Fujian Province This paper investigates how natural-resource endowments affect the provision of 361005, People’s Republic of China local public goods in China. According to fiscal sociology, due to the rentier effect, resource-rich local governments tend to have more state autonomy and are less responsive to society, resulting in poor governance. Moreover, due to political myopia, resource-abundant local governments tend to neglect the accumulation of human capital. Shanxi’s county-level governments are excellent samples to test these hypotheses. Statistical results show that resource-abundant local governments tend to spend less on social expenditures as well as specific education, social security and healthcare, and environmental protection expenditures. Meanwhile, coal-rich governments spend significantly more on self-serving administrative expenditures. The results suggest that negative impacts of natural resources on governmental fiscal extraction and expenditure behaviors are an important causal mechanism of the resource-curse hypothesis. To curb this problem, the current fiscal system needs to be reformed accordingly. Keywords: Resource curse, Coal, Public goods provision, Shanxi Province Introduction Why are developing countries and regions rich in natural resources often characterized by slow economic growth? Exploring this “resource curse,” economists, political scien- tists, and sociologists have put forward various causal mechanisms and explanations.