Surveillance of Antimicrobial Consumption in Europe 2013-2014 SURVEILLANCE REPORT
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Role of Nanobiosensors in Therapeutic Drug Monitoring
Journal of Personalized Medicine Review Personalized Medicine for Antibiotics: The Role of Nanobiosensors in Therapeutic Drug Monitoring Vivian Garzón 1, Rosa-Helena Bustos 2 and Daniel G. Pinacho 2,* 1 PhD Biosciences Program, Universidad de La Sabana, Chía 140013, Colombia; [email protected] 2 Therapeutical Evidence Group, Clinical Pharmacology, Universidad de La Sabana, Chía 140013, Colombia; [email protected] * Correspondence: [email protected]; Tel.: +57-1-8615555 (ext. 23309) Received: 21 August 2020; Accepted: 7 September 2020; Published: 25 September 2020 Abstract: Due to the high bacterial resistance to antibiotics (AB), it has become necessary to adjust the dose aimed at personalized medicine by means of therapeutic drug monitoring (TDM). TDM is a fundamental tool for measuring the concentration of drugs that have a limited or highly toxic dose in different body fluids, such as blood, plasma, serum, and urine, among others. Using different techniques that allow for the pharmacokinetic (PK) and pharmacodynamic (PD) analysis of the drug, TDM can reduce the risks inherent in treatment. Among these techniques, nanotechnology focused on biosensors, which are relevant due to their versatility, sensitivity, specificity, and low cost. They provide results in real time, using an element for biological recognition coupled to a signal transducer. This review describes recent advances in the quantification of AB using biosensors with a focus on TDM as a fundamental aspect of personalized medicine. Keywords: biosensors; therapeutic drug monitoring (TDM), antibiotic; personalized medicine 1. Introduction The discovery of antibiotics (AB) ushered in a new era of progress in controlling bacterial infections in human health, agriculture, and livestock [1] However, the use of AB has been challenged due to the appearance of multi-resistant bacteria (MDR), which have increased significantly in recent years due to AB mismanagement and have become a global public health problem [2]. -

Faldaprevir, Pegylated Interferon, and Ribavirin for Treatment-Naïve HCV Genotype-1: Pooled Analysis of Two Phase 3 Trials
Faldaprevir, pegylated interferon and ribavirin. , 2016; 15 (3): 333-349 333 ORIGINAL ARTICLE May-June, Vol. 15 No. 3, 2016: 333-349 The Official Journal of the Mexican Association of Hepatology, the Latin-American Association for Study of the Liver and the Canadian Association for the Study of the Liver Faldaprevir, pegylated interferon, and ribavirin for treatment-naïve HCV genotype-1: pooled analysis of two phase 3 trials Donald M. Jensen,1 Tarik Asselah,2 Douglas Dieterich,3 Graham R. Foster,4 Mark S. Sulkowski,5 Stefan Zeuzem,6 Parvez Mantry,7 Eric M. Yoshida,8 Christophe Moreno,9 Denis Ouzan,10 Mark Wright,11 Luis E. Morano,12 Robert Buynak,13 Marc Bourlière,14 Tarek Hassanein,15 Shuhei Nishiguchi,16 Jia-Horng Kao,17 Masao Omata,18 Seung W. Paik,19 David K. Wong,20 Edward Tam,21 Kelly Kaita,22 S. Victor Feinman,23 Jerry O. Stern,24 Joseph Scherer,24 Anne-Marie Quinson,24 Florian Voss,25 John-Paul Gallivan,25 Wulf O. Böcher,25 Peter Ferenci26 1 University of Chicago Medicine, Chicago, IL, USA. 2 Hôpital Beaujon, APHP, University Paris-Diderot and INSERM CRB3, Clichy, France. 3 Icahn School of Medicine at Mount Sinai, New York, NY, USA. 4 Queen Mary University of London, London, UK. 5 Johns Hopkins University School of Medicine, Baltimore, MD, USA. 6 J.W.Goethe University Hospital, Frankfurt, Germany. 7 The Liver Institute at Methodist Dallas Medical Center, Dallas, TX, USA. 8 University of British Columbia, Vancouver, BC, Canada. 9 CUB Hôpital Erasme, Université Libre de Bruxelles, Brussels, Belgium. 10 Institut Arnault Tzanck, St. -

List Item Withdrawal Assessment Report for Garenoxacin Mesylate
European Medicines Agency Pre-authorisation Evaluation of Medicines for Human Use London, 18 October 2007 Doc. Ref: EMEA/CHMP/363573/2007 WITHDRAWAL ASSESSMENT REPORT FOR Garenoxacin Mesylate (garenoxacin) EMEA/H/C/747 Day 120 Assessment Report as adopted by the CHMP with all information of a commercially confidential nature deleted. This should be read in conjunction with th e "Question and Answer" document on the withdrawal of the application: the Assessment Report may not include all available information on the product if the CHMP assessment of the latest submitted information was still ongoing at the time of the withdrawal of the application. 7 Westferry Circus, Canary Wharf, London, E14 4HB, UK Tel. (44-20) 74 18 84 00 Fax (44-20) 74 18 84 16 E-mail: [email protected] http://www.emea.europa.eu ©EMEA 2007 Reproduction and/or distribution of this document is authorised for non commercial purposes only provided the EMEA is acknowledged TABLE OF CONTENTS I. RECOMMENDATION ........................................................................................................... 3 II. EXECUTIVE SUMMARY...................................................................................................... 3 II.1 Problem statement............................................................................................. .. ..................... 3 II.2 About the product ............................................................................................. .. ..................... 4 II.3 The development programme/Compliance with -
(12) United States Patent (10) Patent No.: US 8,993,581 B2 Perrine Et Al
US00899.3581B2 (12) United States Patent (10) Patent No.: US 8,993,581 B2 Perrine et al. (45) Date of Patent: Mar. 31, 2015 (54) METHODS FOR TREATINGVIRAL (58) Field of Classification Search DSORDERS CPC ... A61K 31/00; A61K 31/166; A61K 31/185: A61K 31/233; A61K 31/522: A61K 38/12: (71) Applicant: Trustees of Boston University, Boston, A61K 38/15: A61K 45/06 MA (US) USPC ........... 514/263.38, 21.1, 557, 565, 575, 617; 424/2011 (72) Inventors: Susan Perrine, Weston, MA (US); Douglas Faller, Weston, MA (US) See application file for complete search history. (73) Assignee: Trustees of Boston University, Boston, (56) References Cited MA (US) U.S. PATENT DOCUMENTS (*) Notice: Subject to any disclaimer, the term of this 3,471,513 A 10, 1969 Chinn et al. patent is extended or adjusted under 35 3,904,612 A 9/1975 Nagasawa et al. U.S.C. 154(b) by 0 days. (Continued) (21) Appl. No.: 13/915,092 FOREIGN PATENT DOCUMENTS (22) Filed: Jun. 11, 2013 CA 1209037 A 8, 1986 CA 2303268 A1 4f1995 (65) Prior Publication Data (Continued) US 2014/OO45774 A1 Feb. 13, 2014 OTHER PUBLICATIONS Related U.S. Application Data (63) Continuation of application No. 12/890,042, filed on PCT/US 10/59584 Search Report and Written Opinion mailed Feb. Sep. 24, 2010, now abandoned. 11, 2011. (Continued) (60) Provisional application No. 61/245,529, filed on Sep. 24, 2009, provisional application No. 61/295,663, filed on Jan. 15, 2010. Primary Examiner — Savitha Rao (74) Attorney, Agent, or Firm — Nixon Peabody LLP (51) Int. -

Faldaprevir Combined with Peginterferon Alfa2a and Ribavirin In
Faldaprevir Combined With Peginterferon Alfa-2a and Ribavirin in Chronic Hepatitis C Virus Genotype-1 Patients With Prior Nonresponse: SILEN-C2 Trial Mark S. Sulkowski,1 Marc Bourlie`re,2 Jean-Pierre Bronowicki,3 Tarik Asselah,4 Jean-Michel Pawlotsky,5 Stephen D. Shafran,6 Stanislas Pol,7 Stefan Mauss,8 Dominique Larrey,9 Yakov Datsenko,10 Jerry O. Stern,11 George Kukolj,12 Joseph Scherer,11 Gerhard Nehmiz,10 Gerhard G. Steinmann,10 and Wulf O. B€ocher13 Faldaprevir (BI 201335) is a potent, hepatitis C virus (HCV) NS3/4A protease inhibitor. In all, 290 noncirrhotic HCV genotype (GT)-1 patients with prior null (<1log10 viral load [VL] drop at any time on treatment) or partial response (1log10 VL drop but never undetectable on treatment) were randomized 2:1:1 to receive 48 weeks of peginterferon alfa-2a and ribavirin (PegIFN/RBV) in combination with faldaprevir 240 mg once daily (QD) with 3 days PegIFN/RBV lead-in (LI), 240 mg QD without LI, or 240 mg twice daily (BID) with LI. Patients in the 240 mg QD/LI group achieving maintained rapid virologic response (mRVR; VL <25 IU/mL [Roche TaqMan] at week 4 and undetectable at weeks 8 to 20) were rerandomized to cease all treatment at week 24 or continue PegIFN/RBV up to week 48. Sustained virologic response (SVR) rates were 32%, 50%, and 42% in prior partial responders, and 21%, 35%, and 29% in prior null responders in the faldaprevir 240 mg QD/LI, 240 mg QD, and 240 mg BID/LI groups, respectively. -

The Protean Neurologic Manifestations of Varicella-Zoster Virus Infection
REVIEW MARIA A. NAGEL, MD DONALD H. GILDEN, MD Department of Neurology, University of Departments of Neurology and Microbiology, Colorado Health Sciences Center, Denver University of Colorado Health Sciences Center, Denver The protean neurologic manifestations of varicella-zoster virus infection ■ ABSTRACT ARICELLA-ZOSTER VIRUS (VZV) is an V exclusively human, highly neurotropic Multiple neurologic complications may follow the alphaherpesvirus. Primary infection causes reactivation of varicella-zoster virus (VZV), including chickenpox, after which the virus becomes herpes zoster (also known as zoster or shingles), latent in cranial nerve ganglia, dorsal root postherpetic neuralgia, vasculopathy, myelitis, necrotizing ganglia, and autonomic ganglia along the retinitis, and zoster sine herpete (pain without rash). entire neuraxis. Decades later, VZV can reac- These conditions can be difficult to recognize, especially tivate to cause a number of neurologic condi- as several can occur without rash. tions. This article discusses the protean manifes- ■ KEY POINTS tations of VZV reactivation and their diagno- sis and treatment. The most common sequela of herpes zoster is postherpetic neuralgia, which can persist for months and ■ VZV REACTIVATION sometimes years after the rash resolves. HAS MANY MANIFESTATIONS VZV vasculopathy can present as transient ischemic VZV reactivation can produce a number of neurologic complications: attacks, ischemic or hemorrhagic stroke, or aneurysm. •Herpes zoster (shingles), manifesting as pain and rash in up to three dermatomes Vasculopathy and neurologic complications of VZV are • Postherpetic neuralgia, the most common best diagnosed by detecting VZV DNA in bodily fluids or sequela of zoster, is pain that persists for tissues or anti-VZV immunoglobulin G in the months and sometimes years after the rash cerebrospinal fluid. -

Allosteric Drug Transport Mechanism of Multidrug Transporter Acrb
ARTICLE https://doi.org/10.1038/s41467-021-24151-3 OPEN Allosteric drug transport mechanism of multidrug transporter AcrB ✉ Heng-Keat Tam 1,3,4 , Wuen Ee Foong 1,4, Christine Oswald1,2, Andrea Herrmann1, Hui Zeng1 & ✉ Klaas M. Pos 1 Gram-negative bacteria maintain an intrinsic resistance mechanism against entry of noxious compounds by utilizing highly efficient efflux pumps. The E. coli AcrAB-TolC drug efflux pump + 1234567890():,; contains the inner membrane H /drug antiporter AcrB comprising three functionally inter- dependent protomers, cycling consecutively through the loose (L), tight (T) and open (O) state during cooperative catalysis. Here, we present 13 X-ray structures of AcrB in inter- mediate states of the transport cycle. Structure-based mutational analysis combined with drug susceptibility assays indicate that drugs are guided through dedicated transport chan- nels toward the drug binding pockets. A co-structure obtained in the combined presence of erythromycin, linezolid, oxacillin and fusidic acid shows binding of fusidic acid deeply inside the T protomer transmembrane domain. Thiol cross-link substrate protection assays indicate that this transmembrane domain-binding site can also accommodate oxacillin or novobiocin but not erythromycin or linezolid. AcrB-mediated drug transport is suggested to be allos- terically modulated in presence of multiple drugs. 1 Institute of Biochemistry, Goethe-University Frankfurt, Frankfurt am Main, Germany. 2 Sosei Heptares, Steinmetz Building, Granta Park, Great Abington, Cambridge, UK. 3Present -

Clinically Isolated Chlamydia Trachomatis Strains
ANTIMICROBIAL AGENTS AND CHEMOTHERAPY, JUIY 1988, p. 1080-1081 Vol. 32, No. 7 0066-4804/88/071080-02$02.00/0 Copyright © 1988, American Society for Microbiology In Vitro Activities of T-3262, NY-198, Fleroxacin (AM-833; RO 23-6240), and Other New Quinolone Agents against Clinically Isolated Chlamydia trachomatis Strains HIROSHI MAEDA,* AKIRA FUJII, KATSUHISA NAKATA, SOICHI ARAKAWA, AND SADAO KAMIDONO Department of Urology, School of Medicine, Kobe University, 7-5-1 Kusunoki-cho, Chuo-ku, Kobe-city, Japan Received 9 December 1987/Accepted 29 March 1988 The in vitro activities of three newly developed quinolone drugs (T-3262, NY-198, and fleroxacin [AM-833; RO 23-6240]) against 10 strains of clinically isolated Chiamydia trachomatis were assessed and compared with those of other quinolones and minocycline. T-3262 (MIC for 90% of isolates tested, 0.1 ,ug/ml) was the most active of the quinolones. The NY-198 and fleroxacin MICs for 90% of isolates were 3.13 and 62.5 ,ug/ml, respectively. Recently, it has become well known that Chlamydia 1-ml sample of suspension was seeded into flat-bottomed trachomatis is an important human pathogen. It is respon- tubes with glass cover slips and incubated at 37°C in 5% CO2 sible not only for trachoma but also for sexually transmitted for 24 h. The monolayer was inoculated with 103 inclusion- infections, including lymphogranuloma venereum. In forming units of C. trachomatis. The tubes were centrifuged women, it causes cervicitis, endometritis, and salpingitis at 2,000 x g at 25°C for 45 min and left undisturbed at room asymptomatically (19), while in men it causes nongono- temperature for 2 h. -

Treatment of Bacterial Urinary Tract Infections: Presence and Future
european urology 49 (2006) 235–244 available at www.sciencedirect.com journal homepage: www.europeanurology.com Review - Infections Treatment of Bacterial Urinary Tract Infections: Presence and Future Florian M.E. Wagenlehner *, Kurt G. Naber Urologic Clinic, Hospital St. Elisabeth, Straubing, Germany Article info Abstract Article history: Bacterial urinary tract infections (UTIs) are frequent infections in the Accepted December 12, 2005 outpatient as well as in the nosocomial setting. The stratification into Published online ahead of uncomplicated and complicated UTIs has proven to be clinically useful. print on January 4, 2006 Bacterial virulence factors on the one side and the integrity of the host defense mechanisms on the other side determine the course of the Keywords: infection. In uncomplicated UTIs Escherichia coli is the leading organism, Urinary tract infections (UTI) whereas in complicated UTIs the bacterial spectrum is much broader Uncomplicated and including Gram-negative and Gram-positive and often multiresistant complicated UTI organisms. The therapy of uncomplicated UTIs is almost exclusively Antibiotic resistance of antibacterial, whereas in complicated UTIs the complicating factors uropathogens have to be treated as well. There are two predominant aims in the Antibiotic treatment antimicrobial treatment of both uncomplicated and complicated UTIs: New antiinfectives for (i) rapid and effective response to therapy and prevention of recurrence treatment of UTI of the individual patient treated; (ii) prevention of emergence of resis- tance to antimicrobial chemotherapy in the microbial environment. The main drawback of current antibiotic therapies is the emergence and rapid increase of antibiotic resistance. To combat this development several strategies can be followed. Decrease the amount of antibiotics administered, optimal dosing, prevention of infection and development of new antibiotic substances. -
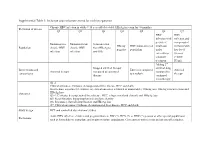
Inclusion and Exclusion Criteria for Each Key Question
Supplemental Table 1: Inclusion and exclusion criteria for each key question Chronic HBV infection in adults ≥ 18 year old (detectable HBsAg in serum for >6 months) Definition of disease Q1 Q2 Q3 Q4 Q5 Q6 Q7 HBV HBV infection with infection and persistent compensated Immunoactive Immunotolerant Seroconverted HBeAg HBV mono-infected viral load cirrhosis with Population chronic HBV chronic HBV from HBeAg to negative population under low level infection infection anti-HBe entecavir or viremia tenofovir (<2000 treatment IU/ml) Adding 2nd Stopped antiviral therapy antiviral drug Interventions and Entecavir compared Antiviral Antiviral therapy compared to continued compared to comparisons to tenofovir therapy therapy continued monotherapy Q1-2: Clinical outcomes: Cirrhosis, decompensated liver disease, HCC and death Intermediate outcomes (if evidence on clinical outcomes is limited or unavailable): HBsAg loss, HBeAg seroconversion and Outcomes HBeAg loss Q3-4: Cirrhosis, decompensated liver disease, HCC, relapse (viral and clinical) and HBsAg loss Q5: Renal function, hypophosphatemia and bone density Q6: Resistance, flare/decompensation and HBeAg loss Q7: Clinical outcomes: Cirrhosis, decompensated liver disease, HCC and death Study design RCT and controlled observational studies Acute HBV infection, children and pregnant women, HIV (+), HCV (+) or HDV (+) persons or other special populations Exclusions such as hemodialysis, transplant, and treatment failure populations. Co treatment with steroids and uncontrolled studies. Supplemental Table 2: Detailed Search Strategy: Ovid Database(s): Embase 1988 to 2014 Week 37, Ovid MEDLINE(R) In-Process & Other Non- Indexed Citations and Ovid MEDLINE(R) 1946 to Present, EBM Reviews - Cochrane Central Register of Controlled Trials August 2014, EBM Reviews - Cochrane Database of Systematic Reviews 2005 to July 2014 Search Strategy: # Searches Results 1 exp Hepatitis B/dt 26410 ("hepatitis B" or "serum hepatitis" or "hippie hepatitis" or "injection hepatitis" or 2 178548 "hepatitis type B").mp. -

A TWO-YEAR RETROSPECTIVE ANALYSIS of ADVERSE DRUG REACTIONS with 5PSQ-031 FLUOROQUINOLONE and QUINOLONE ANTIBIOTICS 24Th Congress Of
A TWO-YEAR RETROSPECTIVE ANALYSIS OF ADVERSE DRUG REACTIONS WITH 5PSQ-031 FLUOROQUINOLONE AND QUINOLONE ANTIBIOTICS 24th Congress of V. Borsi1, M. Del Lungo2, L. Giovannetti1, M.G. Lai1, M. Parrilli1 1 Azienda USL Toscana Centro, Pharmacovigilance Centre, Florence, Italy 2 Dept. of Neurosciences, Psychology, Drug Research and Child Health (NEUROFARBA), 27-29 March 2019 Section of Pharmacology and Toxicology , University of Florence, Italy BACKGROUND PURPOSE On 9 February 2017, the Pharmacovigilance Risk Assessment Committee (PRAC) initiated a review1 of disabling To review the adverse drugs and potentially long-lasting side effects reported with systemic and inhaled quinolone and fluoroquinolone reactions (ADRs) of antibiotics at the request of the German medicines authority (BfArM) following reports of long-lasting side effects systemic and inhaled in the national safety database and the published literature. fluoroquinolone and quinolone antibiotics that MATERIAL AND METHODS involved peripheral and central nervous system, Retrospective analysis of ADRs reported in our APVD involving ciprofloxacin, flumequine, levofloxacin, tendons, muscles and joints lomefloxacin, moxifloxacin, norfloxacin, ofloxacin, pefloxacin, prulifloxacin, rufloxacin, cinoxacin, nalidixic acid, reported from our pipemidic given systemically (by mouth or injection). The period considered is September 2016 to September Pharmacovigilance 2018. Department (PVD). RESULTS 22 ADRs were reported in our PVD involving fluoroquinolone and quinolone antibiotics in the period considered and that affected peripheral or central nervous system, tendons, muscles and joints. The mean patient age was 67,3 years (range: 17-92 years). 63,7% of the ADRs reported were serious, of which 22,7% caused hospitalization and 4,5% caused persistent/severe disability. 81,8% of the ADRs were reported by a healthcare professional (physician, pharmacist or other) and 18,2% by patient or a non-healthcare professional. -

Maxaquin® Lomefloxacin Hydrochloride Tablets to Reduce The
Maxaquin® lomefloxacin hydrochloride tablets To reduce the development of drug-resistant bacteria and maintain the effectiveness of Maxaquin and other antibacterial drugs, Maxaquin should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. DESCRIPTION Maxaquin (lomefloxacin HCl) is a synthetic broad-spectrum antimicrobial agent for oral administration. Lomefloxacin HCl, a difluoroquinolone, is the monohydrochloride salt of (±)-1-ethyl-6, 8-difluoro-1, 4-dihydro-7-(3-methyl-1-piperazinyl)-4-oxo-3- quinolinecarboxylic acid. Its empirical formula is C17H19F2N3O3·HCl, and its structural formula is: Lomefloxacin HCl is a white to pale yellow powder with a molecular weight of 387.8. It is slightly soluble in water and practically insoluble in alcohol. Lomefloxacin HCl is stable to heat and moisture but is sensitive to light in dilute aqueous solution. Maxaquin is available as a film-coated tablet formulation containing 400 mg of lomefloxacin base, present as the hydrochloride salt. The base content of the hydrochloride salt is 90.6%. The inactive ingredients are carboxymethylcellulose calcium, hydroxypropyl cellulose, hypromellose, lactose, magnesium stearate, polyethylene glycol, polyoxyl 40 stearate, and titanium dioxide. CLINICAL PHARMACOLOGY Pharmacokinetics in healthy volunteers: In 6 fasting healthy male volunteers, approximately 95% to 98% of a single oral dose of lomefloxacin was absorbed. Absorption was rapid following single doses of 200 and 400 mg (Tmax 0.8 to 1.4 hours). Mean