Aspirin and Dipyridamole
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

THE ROLE of CILOSTAZOL in ISCHEMIC STROKE
Since 1960 MedicineMedicine KoratKorat โรงพยาบาลมหาราชนครราชสีมา THE ROLE of CILOSTAZOL in ISCHEMIC STROKE PAWUT MEKAWICHAI MD DEPARTMENT of MEDICINE MAHARAT NAKHON RATCHASIMA HOSPITAL This presentation is supported by Thai Osuka CONTENT 1 Mechanism of cilostazol 2 Acute Ischemic Stroke (within 48h) 3 Secondary Prevention and Recurrence 4 Bleeding Complication CONTENT 1 Mechanism of cilostazol 2 Acute Ischemic Stroke (within 48h) 3 Secondary Prevention and Recurrence 4 Bleeding Complication Phosphodiesterase Inhibitor Increase cAMP → reduce platelet function Decrease cAMP → increase platelet function PDE enzyme change cAMP to 5-AMP Inhibit PDE enzyme → incrase cAMP → reduce platelet function Phosphodiesterase inhibitor Cilostazol Dipyridamole Classification of PDE Isozyme 9 family of PDE enzyme (PDE1-9) PDE-3 Site: platelet, heart, vascular SM, adipose tissue Inhibitor: Cilostazol PDE-5 Site: platelet, heart, vascular SM, corpus carvernosum Inhibitor: Dipyridamole, Sidenafil Distribution of PDE Isozyme Cell Platelet Heart Vascular Adipose SMC tissue Dominant III III III III Subordinate II V I II IV IV I II Inhibit PDE-3 antiplatelet action vasodilatation → headache ? inhibit vascular SMC proliferation tachycardia and palpitation increase HDL and decrease TG Which antiplatelet drug that can use in acute ischemic stroke? 1. ASA 81 mg 2. ASA 300 mg 3. Clopidogrel 4. Dipyridamole+ASA 5. Cilostazol CONTENT 1 Mechanism of cilostazol 2 Acute Ischemic Stroke (within 48h) 3 Secondary Prevention and Recurrence 4 Bleeding Complication ASPIRIN International Stroke Trial (IST) 1 ASA 300 mg-reduce mortality and recurrence in 2 wk) Chinese Acute Stroke Trial (CAST) 2 ASA 160 mg-reduce mortality and recurrence) reduce mortality rate 9/1,000 OR 0.92 (0.87-0.98) good functional outcome 7/1,000 OR 1.02 (1.01-1.04) increase severe bleeding (within 2-4 wk) 4/1,000 OR 1.69 (1.35-2.11) no evidence for difference dose of ASA 1.Lancet 1997; 349: 1569-81. -

In Vitro Modulation of Cisplatin Accumulation in Human Ovarian Carcinoma Cells by Pharmacologic Alteration of Microtubules
In vitro modulation of cisplatin accumulation in human ovarian carcinoma cells by pharmacologic alteration of microtubules. R D Christen, … , D R Shalinsky, S B Howell J Clin Invest. 1993;92(1):431-440. https://doi.org/10.1172/JCI116585. Research Article We have previously shown that forskolin and 3-isobutyl-1-methylxanthine (IBMX) increased accumulation of cisplatin (DDP) in DDP-sensitive 2008 human ovarian carcinoma cells in proportion to their ability to increase cAMP. Since the major function of cAMP is to activate protein kinase A, it was conjectured that the stimulation of DDP accumulation was mediated by a protein kinase A substrate. We now show that exposure of 2008 cells to forskolin resulted in phosphorylation of a prominent 52-kD membrane protein. Microsequencing of the band demonstrated it to be human beta-tubulin. Similarly, pretreatment of 2008 cells with the microtubule stabilizing drug taxol increased platinum accumulation in a dose-dependent manner. In 11-fold DDP-resistant 2008/C13*5.25 cells, decreased DDP accumulation was associated with enhanced spontaneous formation of microtubule bundles and decreased expression of beta-tubulin and the tubulin-associated p53 antioncogene relative to 2008 cells. 2008/C13*5.25 cells had altered sensitivity to tubulin- binding drugs, being hypersensitive to taxol and cross-resistant to colchicine. We conclude that pharmacologic alterations of tubulin enhance accumulation of DDP, and that the DDP-resistant phenotype in 2008/C13*5.25 cells is associated with tubulin abnormalities. Find the latest version: https://jci.me/116585/pdf In Vitro Modulation of Cisplatin Accumulation in Human Ovarian Carcinoma Cells by Pharmacologic Alteration of Microtubules Randolph D. -

MEDICINE to TREAT: HEART DISEASES Antiplatelet Agents Aspirin Dipyridamole Clopidogrel Ticlopidine 1. What Are These Medicines U
PATIENT INFORMATION LEAFLET MEDICINE TO TREAT: HEART DISEASES Antiplatelet Agents Aspirin Dipyridamole Clopidogrel Ticlopidine 1. What are these medicines used for? Medicine Purpose of medicine Brand Names This medicine makes blood less sticky to reduce the chance of blood forming Aspirin Cardiprin® clots which will lead to stroke or heart Disprin® attack. This medicine makes the blood less Dipyridamole sticky to reduce the chance of stroke. It Persantin® is used together with aspirin. These medicines make your blood less Clopidogrel (Plavix®) Clopidogrel sticky to reduce chance of stroke or Ticlopidine (Ticlid®), Ticlopidine heart attack. They can be used alone or together with aspirin. 2. How should I take the medicines? • Do not stop taking your medicines without checking with your doctors. • If you miss a dose, take the missed dose as soon as you remember. If it is almost time for your next dose, take only the usual dose. Do not double your dose or use extra medicine to make up for the missed dose. • You should take your medicine after a meal to prevent stomach upset. • How you take aspirin depends on the brand and type of aspirin the doctor gives you: o For tablets which can be chewed: Take with food or glass of water or milk These tablets may be chewed or swallowed whole The solution may also be used as a gargle. Please check with your pharmacist on how to use it as a gargle. o For tablets which enteric coated: Enteric coated forms of aspirin reduce stomach upset that is caused by the medicine Take with a glass of water after food. -

Pyridylisatogen Tosylate of ATP-Responses at a Recombinant P2y1 Purinoceptor 1B.F
British Journal of Pharmacology (I996) 117, 1111 1118 1996 Stockton Press All rights reserved 0007-1188/96 $12.00 0 Potentiation by 2,2'-pyridylisatogen tosylate of ATP-responses at a recombinant P2y1 purinoceptor 1B.F. King, *C. Dacquet, A.U. Ziganshin, tD.F. Weetman, G. Burnstock, *P.M. Vanhoutte & *M. Spedding Department ofAnatomy and Developmental Biology, University College London, Gower Street, London WClE 6BT; *Institute de Recherches Servier, 125 Chemin de Ronde, Croissy sur Seine, 72890, France and tDepartment of Pharmacology, University of Sunderland, Tyne and Wear SRI 3SD 1 2,2'-Pyridylisatogen tosylate (PIT) has been reported to be an irreversible antagonist of responses to adenosine 5'-triphosphate (ATP) at metabotropic purinoceptors (of the P2Y family) in some smooth muscles. When a recombinant P2Y purinoceptor (derived from chick brain) is expressed in Xenopus oocytes, ATP and 2-methylthioATP (2-MeSATP) evoke calcium-activated chloride currents (IC1,ca) in a concentration-dependent manner. The effects of PIT on these agonist responses were examined at this cloned P2Y purinoceptor. 2 PIT (0.1-100 rM) failed to stimulate P2y, purinoceptors directly but, over a narrow concentration range (0.1-3 tM), caused a time-dependent potentiation (2-5 fold) of responses to ATP. The potentiation of ATP-responses by PIT was not caused by inhibition of oocyte ecto-ATPase. At high concentrations (3-100 gM), PIT irreversibly inhibited responses to ATP with a IC_0 value of 13 +9 gM (pKB= 4.88 + 0.22; n = 3). PIT failed to potentiate inward currents evoked by 2-MeSATP and only inhibited the responses to this agonist in an irreversible manner. -

Clinical Update
Atualização Clínica Antiagregantes Plaquetários na Prevenção Primária e Secundária de Eventos Aterotrombóticos Platelet Antiaggregants in Primary and Secondary Prevention of Atherothrombotic Events Marcos Vinícius Ferreira Silva1, Luci Maria SantAna Dusse1, Lauro Mello Vieira, Maria das Graças Carvalho1 Departamento de Análises Clínicas e Toxicológicas, Faculdade de Farmácia, Universidade Federal de Minas Gerais1, Belo Horizonte, MG – Brasil Resumo prevenção primária e secundária de tais eventos. Os fatores A aterotrombose e suas complicações correspondem, de risco associados ao desenvolvimento desses eventos hoje, à principal causa de mortalidade no mundo todo, estão intimamente associados à exacerbação da ativação e sua incidência encontra-se em franca expansão. plaquetária que, por sua vez, favorece a formação de As plaquetas desempenham um papel essencial na agregados plaquetários e geração de trombina, resultando patogênese dos eventos aterotrombóticos, justificando a em trombos ricos em plaquetas (trombos brancos). Dessa utilização dos antiagregantes plaquetários na prevenção forma, o uso de antiagregantes plaquetários tem sido dos mesmos. Desse modo, é essencial que se conheça o benéfico na prevenção primária e secundária de eventos perfil de eficácia e segurança desses fármacos em prevenção mediados por trombos. primária e secundária de eventos aterotrombóticos. Dentro As características dos principais antiagregantes plaquetários desse contexto, a presente revisão foi realizada com o utilizados na prática clínica e em fase de estudos estão objetivo de descrever e sintetizar os resultados dos principais descritos na Tabela 13-9, e as proteínas de membrana com as ensaios, envolvendo a utilização de antiagregantes nos dois quais eles interagem e as vias metabólicas nas quais atuam níveis de prevenção, e avaliando a eficácia e os principais são ilustrados Figura 110. -
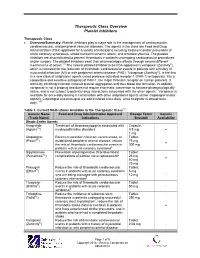
Therapeutic Class Overview Platelet Inhibitors
Therapeutic Class Overview Platelet Inhibitors Therapeutic Class • Overview/Summary: Platelet inhibitors play a major role in the management of cardiovascular, cerebrovascular, and peripheral vascular diseases. The agents in the class are Food and Drug Administration (FDA)-approved for a variety of indications including treatment and/or prevention of acute coronary syndromes, stroke/transient ischemic attack, and thrombocythemia. The platelet inhibitors are also indicated to prevent thrombosis in patients undergoing cardiovascular procedures and/or surgery. The platelet inhibitors exert their pharmacologic effects through several different mechanisms of action.1-8 The newest platelet inhibitor to be FDA-approved is vorapaxar (Zontivity®), which is indicated for the reduction of thrombotic cardiovascular events in patients with a history of myocardial infarction (MI) or with peripheral arterial disease (PAD).7 Vorapaxar (Zontivity®), is the first in a new class of antiplatelet agents called protease-activated receptor-1 (PAR-1) antagonists. It is a competitive and selective antagonist of PAR-1, the major thrombin receptor on human platelets. It works by inhibiting thrombin-induced platelet aggregation and thus blood clot formation. In addition, vorapaxar is not a prodrug and does not require enzymatic conversion to become pharmacologically active, and is not subject to potential drug interactions associated with the other agents.7 Vorapaxar is available for once-daily dosing in combination with other antiplatelet agents (either clopidogrel -

Preventive Drug List
PREVENTIVE DRUG LIST Preventive medications are used for the prevention of conditions such as high blood pressure, high cholesterol, diabetes, asthma, osteoporosis, heart attack, stroke and prenatal nutrient deficiency. Following is a list of generic preventive medications covered at 100%, arranged by type of condition. diltiazem sotalol AF glipizide Asthma related diltiazem ER sotalol HCl glipizide ER albuterol sulfate doxazosin mesylate spironolactone glipizide/metformin HCl albuterol sulfate (nebulizer enalapril maleate spironolactone/hctz glyburide solution) enalapril maleate/hctz telmisartan glyburide micronized albuterol sulfate/ipratropium eplerenone telmisartan/amlodipine glyburide/metformin nebulizer solution eprosartan mesylate telmisartan/hctz Humalog budesonide felodipine ER terazosin HCl Humulin caffeine citrate fosinopril sodium timolol maleate Invokamet cromolyn sodium inhalation fosinopril sodium/hctz torsemide Invokamet XR solution furosemide trandolapril Invokana ipratropium bromide guanfacine HCl trandolapril/verapamil Janumet levalbuterol HCl hydralazine HCl triamterene/hctz Janumet XR levalbuterol tartrate HFA hydrochlorothiazide valsartan Januvia metaproterenol sulfate indapamide valsartan/hctz Kombiglyze XR montelukast irbesartan Vecamyl – mecamylamine HCl Lantus terbutaline sulfate irbesartan/hctz Verapamil Lantus SoloStar TheoChron isradipine Levemir theophylline anhydrous labetalol HCl Blood thinner related Levemir FlexTouch zafirlukast lisinopril aspirin/dipyridamole ER metformin HCl lisinopril/hctz cilostazol -

Persantine® (Dipyridamole USP) 25 Mg, 50 Mg, and 75 Mg Tablets
® Persantine (dipyridamole USP) 25 mg, 50 mg, and 75 mg tablets Rx only Prescribing Information DESCRIPTION PERSANTINE® (dipyridamole USP) is a platelet inhibitor chemically described as 2,2',2'',2'''-[(4,8 Dipiperidinopyrimido[5,4-d]pyrimidine-2,6-diyl)dinitrilo]-tetraethanol. It has the following structural formula: OH N N N N HO N N N OH N OH C24H40N8O4 Mol. Wt. 504.63 Dipyridamole is an odorless yellow crystalline powder, having a bitter taste. It is soluble in dilute acids, methanol and chloroform, and practically insoluble in water. PERSANTINE tablets for oral administration contain: Active Ingredient TABLETS 25 mg, 50 mg, and 75 mg: dipyridamole USP 25 mg, 50 mg and 75 mg, respectively. Inactive Ingredients TABLETS 25 mg, 50 mg, and 75 mg: acacia, carnauba wax, corn starch, edible white ink, lactose monohydrate, magnesium stearate, D&C yellow #10 aluminum lake, D&C red #30, helendon aluminum pink lake, sodium benzoate, methylparaben, propylparaben, polyethylene glycol, povidone, sucrose, talc, titanium dioxide, and white wax. CLINICAL PHARMACOLOGY It is believed that platelet reactivity and interaction with prosthetic cardiac valve surfaces, resulting in abnormally shortened platelet survival time, is a significant factor in thromboembolic complications occurring in connection with prosthetic heart valve replacement. PERSANTINE tablets have been found to lengthen abnormally shortened platelet survival time in a dose-dependent manner. In three randomized controlled clinical trials involving 854 patients who had undergone surgical placement of a prosthetic heart valve, PERSANTINE tablets, in combination with warfarin, decreased the incidence of postoperative thromboembolic events by 62 to 91% compared to warfarin treatment alone. -

Platelet-Inhibiting Drugs: a Hematologist’S Perspective
Platelet-Inhibiting Drugs: A Hematologist’s Perspective A. Koneti Rao, M.D., Sol Sherry Professor of Medicine Chief, Hematology Section Co-Director, Sol Sherry Thrombosis Research Center Temple University School of Medicine 2012 Chile Akkerman JW, Bouma BN, Sixma JJ. Atlas of Hemostasis, 1979. STRATEGIES FOR ANTITHROMBOTIC THERAPY VASCULAR INJURY RISK FACTOR REDUCTION PLATELET ADHERANCE PLATELET AGGREGATION INHIBITORS COAGULATION ACTIVATION THROMBIN GENERATION ANTICOAGULANTS FIBRIN FORMATION PLASMIN GENERATION THROMBOLYTICS FIBRINOLYSIS AKR/2003 Platelet Responses to Activation Membrane Shape Change Aggregation Dense AGONIST Receptor Granule Secretion Thromboxane Alpha Granule Production TxA2 Acid Hydrolase ADP / Serotonin Rao/2010 GPIIb/IIIa GPIIb/IIIa Fibrinogen GPIIb/IIIa GPVI GPIa/IIa a5b1 GPIb/V/IX GPIV Damaged vWF Fibronectin Endothelium Collagen AKR/2004 Platelet-Inhibiting Drugs Platelet Inhibiting Drugs Aspirin Sulfinpyrazone Dipyridamole P2Y12 Antagonists • Thienopyridines - Ticlopidine, Clopidogrel, Prasugrel (Effient) • Non-Thienopyridine - Ticagrelor (Brilinta) GPIIb/IIIa Inhibitors Abciximab (c7E3 Fab, ReoPro) Eptifibatide (Integrilin) Tirofiban (Aggrastat) Cilostazol (Pletal) AKR-2012 Cyclooxygenase Arachidonic Acid Ser OH 529 COOH OCOCH 3 PGG2, PGH2 Aspirin Cyclooxygenase Thromboxane A2 Ser OCOCH 3 AKR/09 529 GPIIb-IIIa cAMP Fibrinogen ADP (P2Y12) Gi AC Aggregation Ticlopidine P ATP Pleckstrin Clopidogrel GP IIb/IIIa Prasugrel PKC Antagonists TxA2 ADP Gq Secretion (P2Y1) DG TS Gq PGG2/PGH2 Thrombin PLC PIP2 CO Aspirin -

A Randomized, Controlled Trial Comparing Cilostazol with Extended Release Dipyridamole in the Management of Intermittent Claudication in Peripheral Artery Disease
1 IRB PROTOCOL Geetha F. Pinto, MD Dec 18th, 2007 2 A Randomized, Controlled Trial Comparing Cilostazol with Extended Release Dipyridamole in the Management of Intermittent Claudication in Peripheral Artery Disease A. Study Purpose: The main goal of this prospective, double-blinded, randomized, controlled, trial is to compare the efficacy of extended-release dipyridamole with cilostazol in the management of lower extremity claudication in patients with peripheral vascular disease (PAD). B. Rationale: Intermittent claudication (IC) is defined as a reproducible discomfort of a defined group of muscles which is induced by exercise and relieved with rest. The symptoms result from an imbalance between supply and demand of blood flow that fails to satisfy ongoing metabolic requirements. [1] Not only is PAD now considered coronary heart disease (CHD) equivalent, symptomatic PAD contributes significantly to the morbidity and quality of life in patients with IC. The prevalence of IC would appear to increase from about 3% in patients aged 40 to 6% in patients aged 60 years. [2] Despite this prevalence, the treatment options for IC are limited. The Trans-Atlantic Inter-Society Consensus Document on Management of Peripheral Arterial Disease II (TASC II), a working group of expert representatives recently published a summary of all the evidence for the particular drug therapies available for the management of IC in the United States being limited to cilostazol, a phosphodiesterase III inhibitor with vasodilator, metabolic and antiplatelet activity. Though many other drugs have been studied including other vasoactive and antiplatelet agents, only one other drug, naftidrofuryl ( l5- hydroxytryptamine type 2 antagonist), a drug available only in Europe has shown comparable efficacy in the management of IC. -

Adenosine-Mediated Effects of Ticagrelor Evidence and Potential Clinical Relevance
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Elsevier - Publisher Connector Journal of the American College of Cardiology Vol. 63, No. 23, 2014 Ó 2014 by the American College of Cardiology Foundation ISSN 0735-1097/$36.00 Published by Elsevier Inc. http://dx.doi.org/10.1016/j.jacc.2014.03.031 Adenosine-Mediated Effects of Ticagrelor Evidence and Potential Clinical Relevance Marco Cattaneo, MD,* Rainer Schulz, MD, PHD,y Sven Nylander, PHDz Milan, Italy; Giessen, Germany; and Mölndal, Sweden This review constitutes a critical evaluation of recent publications that have described an additional mode of action of the P2Y12 receptor antagonist ticagrelor. The effect is mediated by inhibition of the adenosine transporter ENT1 (type 1 equilibrative nucleoside transporter), which provides protection for adenosine from intracellular metabolism, thus increasing its concentration and biological activity, particularly at sites of ischemia and tissue injury where it is formed. Understanding the mode of action of ticagrelor is of particular interest given that its clinical profile, both in terms of efficacy and adverse events, differs from that of thienopyridine P2Y12 antagonists. (J Am Coll Cardiol 2014;63:2503–9) ª 2014 by the American College of Cardiology Foundation Ticagrelor is a direct-acting, reversibly binding P2Y12 Outcomes) study, ticagrelor was superior to clopidogrel in antagonist that provides rapid onset of antiplatelet effects preventing cardiovascular death, myocardial infarction, or after oral administration. P2Y12, 1 of the 2 purinergic stroke (9.8% vs. 11.7%, a 16% reduction) in patients with receptors for adenosine diphosphate (ADP) expressed by acute coronary syndrome (ACS); two-thirds of these pa- platelets, is essential for normal ADP-induced platelet tients had undergone percutaneous coronary intervention fi aggregation. -

CLINICAL POLICY Regadenoson Pharmacologic Stress Test
CLINICAL POLICY Regadenoson Pharmacologic Stress Test A. EFFECTIVE DATE : March 16, 2021 B. PURPOSE : Pharmacologic stress testing is utilized to diagnose overt or latent coronary artery disease in individuals unable to undergo exercise stress testing. C. POLICY : A Cardiology Fellow, Physician’s Assistant, Advanced Practice Registered Nurse, or Registered Nurse will supervise individual tests with oversight by a credentialed cardiologist. All clinicians supervising stress tests must be ACLS-certified. D. SCOPE : INDICATIONS: Regadenoson is a selective A2A adenosine receptor antagonist causing coronary vasodilation and increased coronary blood flow. A regadenoson pharmacologic stress test with nuclear myocardial perfusion imagining is indicated for patients unable to undergo adequate exercise stress testing and in the presence of the following conditions: 1. Inability to perform adequate exercise due to non-cardiac physical or mental limitations. 2. LBBB or ventricular pacing baseline ECG abnormalities. 3. Severe aortic stenosis CONTRAINDICATIONS: Absolute: • Patients with second or third degree AV block or sinus node dysfunction without a functioning pacemaker. • Known hypersensitivity to adenosine or regadenoson. • Systolic blood pressure less than 90mmHg. • Use of dipyridamole or dipyridamole containing medications within 48 hours of test. • Use of aminophylline within 24 hours of test. • Ingestion of caffeinated foods or beverages within 12 hours of the test. • Use of products containing methylxanthines or theophylline containing medications. • Unstable angina or acute coronary syndrome. • Patients with severe COPD or asthma with active wheezing. Relative: • Profound sinus bradycardia (HR <40 BPM) • Patients with mild COPD, asthma or reactive airway disease. The safety of regadenoson is not definitely established in patients with bronchoconstrictive lung disease such as asthma or COPD.