Distinct Representations of Olfactory Information in Different Cortical Centres
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Born Granule Cells During Implicit Versus Explicit Olfactory Learning
RESEARCH ARTICLE Opposite regulation of inhibition by adult- born granule cells during implicit versus explicit olfactory learning Nathalie Mandairon1*, Nicola Kuczewski1, Florence Kermen1, Je´ re´ my Forest1, Maellie Midroit1, Marion Richard1, Marc Thevenet1, Joelle Sacquet1, Christiane Linster2,3, Anne Didier1 1Lyon Neuroscience Research Center, Neuroplasticity and Neuropathology of Olfactory Perception Team, CNRS UMR 5292, INSERM U1028, Universite´ de Lyon, Lyon, France; 2Computational Physiology Lab, Cornell University, Ithaca, United States; 3Department of Neurobiology and Behavior, Cornell University, Ithaca, United States Abstract Both passive exposure and active learning through reinforcement enhance fine sensory discrimination abilities. In the olfactory system, this enhancement is thought to occur partially through the integration of adult-born inhibitory interneurons resulting in a refinement of the representation of overlapping odorants. Here, we identify in mice a novel and unexpected dissociation between passive and active learning at the level of adult-born granule cells. Specifically, while both passive and active learning processes augment neurogenesis, adult-born cells differ in their morphology, functional coupling and thus their impact on olfactory bulb output. Morphological analysis, optogenetic stimulation of adult-born neurons and mitral cell recordings revealed that passive learning induces increased inhibitory action by adult-born neurons, probably resulting in more sparse and thus less overlapping odor representations. Conversely, after active learning inhibitory action is found to be diminished due to reduced connectivity. In this case, strengthened odor response might underlie enhanced discriminability. *For correspondence: DOI: https://doi.org/10.7554/eLife.34976.001 [email protected] Competing interests: The authors declare that no Introduction competing interests exist. Brain representations of the environment constantly evolve through learning mediated by different Funding: See page 13 plasticity mechanisms. -

Cell Migration in the Developing Rodent Olfactory System
Cell. Mol. Life Sci. (2016) 73:2467–2490 DOI 10.1007/s00018-016-2172-7 Cellular and Molecular Life Sciences REVIEW Cell migration in the developing rodent olfactory system 1,2 1 Dhananjay Huilgol • Shubha Tole Received: 16 August 2015 / Revised: 8 February 2016 / Accepted: 1 March 2016 / Published online: 18 March 2016 Ó The Author(s) 2016. This article is published with open access at Springerlink.com Abstract The components of the nervous system are Abbreviations assembled in development by the process of cell migration. AEP Anterior entopeduncular area Although the principles of cell migration are conserved AH Anterior hypothalamic nucleus throughout the brain, different subsystems may predomi- AOB Accessory olfactory bulb nantly utilize specific migratory mechanisms, or may aAOB Anterior division, accessory olfactory bulb display unusual features during migration. Examining these pAOB Posterior division, accessory olfactory bulb subsystems offers not only the potential for insights into AON Anterior olfactory nucleus the development of the system, but may also help in aSVZ Anterior sub-ventricular zone understanding disorders arising from aberrant cell migra- BAOT Bed nucleus of accessory olfactory tract tion. The olfactory system is an ancient sensory circuit that BST Bed nucleus of stria terminalis is essential for the survival and reproduction of a species. BSTL Bed nucleus of stria terminalis, lateral The organization of this circuit displays many evolution- division arily conserved features in vertebrates, including molecular BSTM Bed nucleus of stria terminalis, medial mechanisms and complex migratory pathways. In this division review, we describe the elaborate migrations that populate BSTMa Bed nucleus of stria terminalis, medial each component of the olfactory system in rodents and division, anterior portion compare them with those described in the well-studied BSTMpl Bed nucleus of stria terminalis, medial neocortex. -

Comprehensive Connectivity of the Mouse Main Olfactory Bulb: Analysis and Online Digital Atlas
ORIGINAL RESEARCH ARTICLE published: 07 August 2012 NEUROANATOMY doi: 10.3389/fnana.2012.00030 Comprehensive connectivity of the mouse main olfactory bulb: analysis and online digital atlas Houri Hintiryan , Lin Gou , Brian Zingg , Seita Yamashita , Hannah M. Lyden , Monica Y. Song , Arleen K. Grewal , Xinhai Zhang , Arthur W. Toga and Hong-Wei Dong* Laboratory of Neuro Imaging, Department of Neurology, David Geffen School of Medicine, University of California, Los Angeles, Los Angeles, CA, USA Edited by: We introduce the first open resource for mouse olfactory connectivity data produced as Jorge A. Larriva-Sahd, Universidad part of the Mouse Connectome Project (MCP) at UCLA. The MCP aims to assemble a Nacional Autónoma de México, whole-brain connectivity atlas for the C57Bl/6J mouse using a double coinjection tracing Mexico method. Each coinjection consists of one anterograde and one retrograde tracer, which Reviewed by: Marco Aurelio M. Freire, Edmond affords the advantage of simultaneously identifying efferent and afferent pathways and and Lily Safra International Institute directly identifying reciprocal connectivity of injection sites. The systematic application for Neurosciences of Natal, Brazil of double coinjections potentially reveals interaction stations between injections and Juan Andrés De Carlos, Instituto allows for the study of connectivity at the network level. To facilitate use of the data, Cajal (Consejo Superior de Investigaciones Científicas), Spain raw images are made publicly accessible through our online interactive visualization *Correspondence: tool, the iConnectome, where users can view and annotate the high-resolution, Hong-Wei Dong, Laboratory of multi-fluorescent connectivity data (www.MouseConnectome.org). Systematic double Neuro Imaging, Department of coinjections were made into different regions of the main olfactory bulb (MOB) and data Neurology, David Geffen School of from 18 MOB cases (∼72 pathways; 36 efferent/36 afferent) currently are available to Medicine, University of California, Los Angeles, 635 Charles E. -
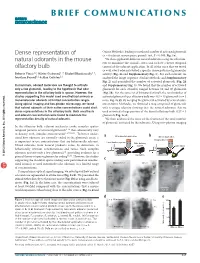
Dense Representation of Natural Odorants in the Mouse Olfactory Bulb
BRIEF COMMUNICATIONS Dense representation of Online Methods), leading to a reduced number of activated glomeruli (n = 6 odorant-mouse pairs, paired t test, P < 0.005; Fig. 1e). natural odorants in the mouse We then applied 40 different natural odorants using the olfactom- eter to minimize the animal’s stress and to have a better temporal olfactory bulb control of the odorant application. In all of the mice that we tested (n = 8), every odorant evoked a specific dense pattern of glomerular Roberto Vincis1,2, Olivier Gschwend1–3, Khaleel Bhaukaurally1–3, activity (Fig. 2a and Supplementary Fig. 1). For each odorant, we Jonathan Beroud1,2 & Alan Carleton1,2 analyzed the image sequence (Online Methods and Supplementary Fig. 2) and quantified the number of activated glomeruli (Fig. 2b In mammals, odorant molecules are thought to activate and Supplementary Fig. 3). We found that the number of activated only a few glomeruli, leading to the hypothesis that odor glomeruli for each stimulus ranged between 10 and 40 glomeruli representation in the olfactory bulb is sparse. However, the (Fig. 2b). For the same set of 30 natural stimuli, the total number of studies supporting this model used anesthetized animals or activated glomeruli per olfactory bulb was 443 ± 15 glomeruli (n = 5 monomolecular odorants at limited concentration ranges. mice; Fig. 2c,d). By merging the glomeruli activated by several odor- Using optical imaging and two-photon microscopy, we found ants (Online Methods), we obtained a map composed of glomeruli that natural odorants at their native concentrations could elicit with a unique identity showing that the natural odorants that we dense representations in the olfactory bulb. -

Functional Organization of Sensory Input to the Olfactory Bulb Glomerulus Analyzed by Two-Photon Calcium Imaging
Functional organization of sensory input to the olfactory bulb glomerulus analyzed by two-photon calcium imaging Matt Wachowiak*, Winfried Denk†, and Rainer W. Friedrich†‡ *Department of Biology, Boston University, 5 Cummington Street, Boston, MA 02215; and †Department of Biomedical Optics, Max Planck Institute for Medical Research, Jahnstrasse 29, D-69120 Heidelberg, Germany Edited by Charles F. Stevens, The Salk Institute for Biological Studies, La Jolla, CA, and approved May 10, 2004 (received for review January 20, 2004) Glomeruli in the olfactory bulb are anatomically discrete modules OSNs distinguished by histochemical markers terminate in dis- receiving input from idiotypic olfactory sensory neurons. To ex- crete subregions of the axonal compartment (36, 37). These data amine the functional organization of sensory inputs to individual raise the possibility that functionally distinct subdivisions exist .glomeruli, we loaded olfactory sensory neurons with a Ca2؉ within a glomerulus indicator and measured odorant-evoked presynaptic Ca2؉ signals Direct measurements of the activity of glomerular afferents within single glomeruli by using two-photon microscopy in anaes- have been difficult because most functional measures of intra- thetized mice. Odorants evoked patterns of discrete Ca2؉ signals glomerular activity lack sufficient spatial resolution or cellular throughout the neuropil of a glomerulus. Across glomeruli, Ca2؉ specificity. Here, we loaded OSNs with a fluorescent Ca2ϩ signals occurred with equal probability in all glomerular regions. indicator and measured patterns of afferent presynaptic activity ؉ Within single glomeruli, the pattern of intraglomerular Ca2 sig- within glomeruli by two-photon Ca2ϩ imaging in anaesthetized nals was indistinguishable for stimuli of different duration, iden- mice. Our data indicate that OSN input to individual glomeruli tity, and concentration. -

Medial Temporal Lobe (The Limbic System)
MEDIAL TEMPORAL LOBE (THE LIMBIC SYSTEM) On the medial surface of the temporal lobe are three structures critical for normal human functioning. From rostral to caudal, they are the olfactory cortex, the amygdala, and the hippocampus. We will look at the anatomy and function of each separately, although they are often grouped together as "the limbic system". A. The olfactory system: The olfactory system actually begins in the roof of the nasal cavity. The olfactory receptors are ciliated epithelial cells with an array of receptors capable of detecting thousands of different odors. However, just as with any sensory system, the receptor neurons themselves do not project to the cerebral hemispheres. Their axons project up through the cribiform plate of the skull to synapse on the dendrites of the mitral cells of the olfactory bulb. The axons of the olfactory receptors make up the elusive cranial nerve I. This fragile tract is susceptible to shearing forces in head trauma, and loss of smell is a surprisingly debilitating injury. Here is an example of a section through olfactory bulb. The olfactory bulb is not a simple relay (something which passively transmits the signal), but is a sophisticated structure in itself. The mitral cell- olfactory neuron synapse is actually within a tangle of axons and dendrites that is called a glomerulus. There is a second cell type tucked around these glomeruli which probably affects how the signal is transmitted. These cells are small and densely packed, which gives them the name "granule cells". However, they bear no relation to the granule cells of the cerebellum or cerebral cortex. -

Taste and Smell Disorders in Clinical Neurology
TASTE AND SMELL DISORDERS IN CLINICAL NEUROLOGY OUTLINE A. Anatomy and Physiology of the Taste and Smell System B. Quantifying Chemosensory Disturbances C. Common Neurological and Medical Disorders causing Primary Smell Impairment with Secondary Loss of Food Flavors a. Post Traumatic Anosmia b. Medications (prescribed & over the counter) c. Alcohol Abuse d. Neurodegenerative Disorders e. Multiple Sclerosis f. Migraine g. Chronic Medical Disorders (liver and kidney disease, thyroid deficiency, Diabetes). D. Common Neurological and Medical Disorders Causing a Primary Taste disorder with usually Normal Olfactory Function. a. Medications (prescribed and over the counter), b. Toxins (smoking and Radiation Treatments) c. Chronic medical Disorders ( Liver and Kidney Disease, Hypothyroidism, GERD, Diabetes,) d. Neurological Disorders( Bell’s Palsy, Stroke, MS,) e. Intubation during an emergency or for general anesthesia. E. Abnormal Smells and Tastes (Dysosmia and Dysgeusia): Diagnosis and Treatment F. Morbidity of Smell and Taste Impairment. G. Treatment of Smell and Taste Impairment (Education, Counseling ,Changes in Food Preparation) H. Role of Smell Testing in the Diagnosis of Neurodegenerative Disorders 1 BACKGROUND Disorders of taste and smell play a very important role in many neurological conditions such as; head trauma, facial and trigeminal nerve impairment, and many neurodegenerative disorders such as Alzheimer’s, Parkinson Disorders, Lewy Body Disease and Frontal Temporal Dementia. Impaired smell and taste impairs quality of life such as loss of food enjoyment, weight loss or weight gain, decreased appetite and safety concerns such as inability to smell smoke, gas, spoiled food and one’s body odor. Dysosmia and Dysgeusia are very unpleasant disorders that often accompany smell and taste impairments. -

Plume Dynamics Structure the Spatiotemporal Activity of Mitral/Tufted Cell Networks in the Mouse Olfactory Bulb
ORIGINAL RESEARCH published: 30 April 2021 doi: 10.3389/fncel.2021.633757 Plume Dynamics Structure the Spatiotemporal Activity of Mitral/Tufted Cell Networks in the Mouse Olfactory Bulb Suzanne M. Lewis 1*, Lai Xu 1, Nicola Rigolli 2,3, Mohammad F. Tariq 4, Lucas M. Suarez 1, Merav Stern 5, Agnese Seminara 3 and David H. Gire 1* 1 Department of Psychology, University of Washington, Seattle, WA, United States, 2 Dipartimento di Fisica, Istituto Nazionale Fisica Nucleare (INFN) Genova, Universitá di Genova, Genova, Italy, 3 CNRS, Institut de Physique de Nice, Université Côte d’Azur, Nice, France, 4 Graduate Program in Neuroscience, University of Washington, Seattle, WA, United States, 5 Department of Applied Mathematics, University of Washington, Seattle, WA, United States Although mice locate resources using turbulent airborne odor plumes, the stochasticity and intermittency of fluctuating plumes create challenges for interpreting odor cues in natural environments. Population activity within the olfactory bulb (OB) is thought to process this complex spatial and temporal information, but how plume dynamics Edited by: impact odor representation in this early stage of the mouse olfactory system is unknown. Shaina M. Short, The University of Utah, United States Limitations in odor detection technology have made it difficult to measure plume Reviewed by: fluctuations while simultaneously recording from the mouse’s brain. Thus, previous Andreas Schaefer, studies have measured OB activity following controlled odor pulses of varying profiles or Francis Crick Institute and University frequencies, but this approach only captures a subset of features found within olfactory College London, London, United Kingdom plumes. Adequately sampling this feature space is difficult given a lack of knowledge Shawn Denver Burton, regarding which features the brain extracts during exposure to natural olfactory scenes. -

Smell & Taste.Pdf
Smell and Taste 428 Special senses 1. SMELL (OLFACTION) 1.1 Overview Smell is the least Understood sense. It is mainly subjective. In dogs and other animals, it is more developed than humans. - There are dfferent stimuli that can be smelled such as: camphoraceous, musky, flora (flower), pepperminty, ethereal, pungent, putrid 1.2 Structure of Olfactory epithelium and bulb See the figure on the next page! 1.2.1 Olfactory mucous membrane It is the upper lining of the nasal cavity (near the septum), containing olfactory (odorant) receptors that are responsible for smelling. o Olfactory receptors are bipolar neurons which receive stimuli in the nasal cavity (through cilia) and transmits them through axons, leave the olfactory epithelium and travel into CNS (olfactory bulb). o Although they are nerve cells, olfactory receptor cells are replaced every 60 days or so, and they grow their axon into the correct place in CNS. Olfactory epithelium contains three types of cells (the olfactory receptors cells discussed) as well as two other types of cells: o Olfactory (Bowman’s) glands: produce mucus that dissolves odorants o Supporting cell o Basal cells: regenerate olfactory receptor cells. 1 Smell and Taste 428 1.2.2 Olfactory bulb The olfactory bulb is made up of nerves that receive olfactory signals from axons of olfactory receptor cells. These nerves are of two cell types: o Mitral cells (most important) (M) o Tufted cells (smaller than mitral cells) (T) Mitral and tufted cells release glutamate The synapse between the axons of olfactory receptor cells and dendrites of mitral cells occur in clusters called olphactory glomeruli (OG) In a glomerulus, about 1000 olfactory receptor axons converge onto 1 mitral cell. -

Arc-Expressing Neuronal Ensembles
14070 • The Journal of Neuroscience, October 14, 2015 • 35(41):14070–14075 Brief Communications Arc-Expressing Neuronal Ensembles Supporting Pattern Separation Require Adrenergic Activity in Anterior Piriform Cortex: An Exploration of Neural Constraints on Learning X Amin MD. Shakhawat,1 XAli Gheidi,1 X Iain T. MacIntyre,1 Melissa L. Walsh,1 XCarolyn W. Harley,2 and XQi Yuan1 1Division of Biomedical Sciences, Faculty of Medicine, and 2Department of Psychology, Faculty of Science, Memorial University of Newfoundland, St. John’s, Newfoundland A1B 3V6, Canada Arc ensembles in adult rat olfactory bulb (OB) and anterior piriform cortex (PC) were assessed after discrimination training on highly similar odor pairs. Nonselective ␣- and -adrenergic antagonists or saline were infused in the OB or anterior PC during training. OB adrenergic blockade slowed, but did not prevent, odor discrimination learning. After criterion performance, Arc ensembles in anterior piriform showed enhanced stability for the rewarded odor and pattern separation for the discriminated odors as described previously. Anterior piriform adrenergic blockade prevented acquisition of similar odor discrimination and of OB ensemble changes, even with extended overtraining. Mitral and granule cell Arc ensembles in OB showed enhanced stability for rewarded odor only in the saline group. Pattern separation was not seen in the OB. Similar odor discrimination co-occurs with increased stability in rewarded odor representa- tions and pattern separation to reduce encoding overlap. The difficulty of similar discriminations may relate to the necessity to both strengthen rewarded representations and weaken overlap across similar representations. Key words: Arc; norepinephrine; odor discrimination; olfactory bulb; pattern separation; piriform cortex Significance Statement We show for the first time that adrenoceptors in anterior piriform cortex (aPC) must be engaged for adult rats to learn to discriminate highly similar odors. -

Clusters of Secretagogin-Expressing Neurons in the Aged Human Olfactory Tract Lack Terminal Differentiation
Clusters of secretagogin-expressing neurons in the aged human olfactory tract lack terminal differentiation Johannes Attemsa,1, Alan Alparb,c,1, Lauren Spenceb, Shane McParlanda, Mathias Heikenwalderd, Mathias Uhléne, Heikki Tanilaf, Tomas G. M. Hökfeltg,2, and Tibor Harkanyb,c,2 aInstitute for Ageing and Health, Newcastle University, Newcastle upon Tyne NE4 5PL, United Kingdom; bEuropean Neuroscience Institute at Aberdeen, University of Aberdeen, Aberdeen AB25 2ZD, United Kingdom; cDivision of Molecular Neurobiology, Department of Medical Biochemistry and Biophysics, and gDepartment of Neuroscience, Karolinska Institutet, SE-17177 Stockholm, Sweden; dInstitute of Virology, Technische Universität/Helmholtz Zentrum München, D-81675 Munich, Germany; eScience for Life Laboratory, Royal Institute of Technology, SE-17121 Stockholm, Sweden; and fDepartment of Neurology, Kuopio University Hospital and A. I. Virtanen Institute, University of Eastern Finland, FI-70211, Kuopio, Finland Contributed by Tomas G. M. Hökfelt, March 6, 2012 (sent for review November 10, 2011) Expanding the repertoire of molecularly diverse neurons in the olfactory system, we found secretagogin-positive (secretagogin+) human nervous system is paramount to characterizing the neuro- neurons in the RMS and olfactory bulb (16). Therefore, we hy- nal networks that underpin sensory processing. Defining neuronal pothesized that secretagogin may reveal previously undescribed identities is particularly timely in the human olfactory system, cellular identities and cytoarchitectural -

Asymmetric Neurotransmitter Release Enables Rapid Odour Lateralization in Drosophila
SUPPLEMENTARY INFORMATION doi:10.1038/nature11747 SUPPLEMENTARY NOTES Note S1: Light offset produced a precise compensatory turn in the direction opposite to the light-evoked turn (Fig. 1g). As a result, the fly’s net lateral displacement after this compensatory turn was close to zero (Fig. 1i). A train of light pulses produced a larger turn, and the fly’s compensatory turn after light offset was commensurately larger, although less precise (Fig. 1i). This suggests that the fly keeps track of how far it has turned away from its original path, and the accuracy of its estimate depends on how far it has strayed. Note S2: Resting GCaMP fluorescence (F) was not significantly different in the right and left glomeruli (p = 0.67, n = 6, paired t-test). This is expected because each glomerulus contains the axons arising from both the right and left antennae. Although one antenna is removed, the cut axons arising from that antenna are still present in the neuropil. Thus, both right and left glomeruli must contain the same number of GCaMP molecules, and so resting fluorescence is the same. Spontaneous ORN activity evidently makes little contribution to F. What is the mechanistic basis for the significant ipsi/contra difference in F/F? A previous study reported that an ORN plasma membrane marker is two-fold more abundant in the ipsilateral glomerulus as compared to the contralateral glomerulus1. This suggests that the ipsilateral arbor is simply larger than the contralateral arbor of the same ORN. If the properties of ipsi- and contralateral branches are the same, then the ipsi/contra asymmetry in F/F should be roughly proportional to the ipsi/contra asymmetry in arbor size.