Prevalence, Intensity and Control Strategies of Soil-Transmitted Helminth
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
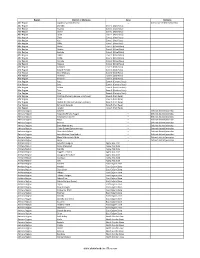
Districts of Ethiopia
Region District or Woredas Zone Remarks Afar Region Argobba Special Woreda -- Independent district/woredas Afar Region Afambo Zone 1 (Awsi Rasu) Afar Region Asayita Zone 1 (Awsi Rasu) Afar Region Chifra Zone 1 (Awsi Rasu) Afar Region Dubti Zone 1 (Awsi Rasu) Afar Region Elidar Zone 1 (Awsi Rasu) Afar Region Kori Zone 1 (Awsi Rasu) Afar Region Mille Zone 1 (Awsi Rasu) Afar Region Abala Zone 2 (Kilbet Rasu) Afar Region Afdera Zone 2 (Kilbet Rasu) Afar Region Berhale Zone 2 (Kilbet Rasu) Afar Region Dallol Zone 2 (Kilbet Rasu) Afar Region Erebti Zone 2 (Kilbet Rasu) Afar Region Koneba Zone 2 (Kilbet Rasu) Afar Region Megale Zone 2 (Kilbet Rasu) Afar Region Amibara Zone 3 (Gabi Rasu) Afar Region Awash Fentale Zone 3 (Gabi Rasu) Afar Region Bure Mudaytu Zone 3 (Gabi Rasu) Afar Region Dulecha Zone 3 (Gabi Rasu) Afar Region Gewane Zone 3 (Gabi Rasu) Afar Region Aura Zone 4 (Fantena Rasu) Afar Region Ewa Zone 4 (Fantena Rasu) Afar Region Gulina Zone 4 (Fantena Rasu) Afar Region Teru Zone 4 (Fantena Rasu) Afar Region Yalo Zone 4 (Fantena Rasu) Afar Region Dalifage (formerly known as Artuma) Zone 5 (Hari Rasu) Afar Region Dewe Zone 5 (Hari Rasu) Afar Region Hadele Ele (formerly known as Fursi) Zone 5 (Hari Rasu) Afar Region Simurobi Gele'alo Zone 5 (Hari Rasu) Afar Region Telalak Zone 5 (Hari Rasu) Amhara Region Achefer -- Defunct district/woredas Amhara Region Angolalla Terana Asagirt -- Defunct district/woredas Amhara Region Artuma Fursina Jile -- Defunct district/woredas Amhara Region Banja -- Defunct district/woredas Amhara Region Belessa -- -

Revisiting Gamo: Linguists’ Classification Versus Self Identification of the Community
Vol. 5(9), pp. 373-380, December, 2013 DOI: 10.5897/IJSA2013.0471 International Journal of Sociology and ISSN 2006- 988x © 2013 Academic Journals Anthropology http://www.academicjournals.org/IJSA Full Length Research Paper Revisiting Gamo: Linguists’ classification versus self identification of the community Hirut Woldemariam Department of Linguistics, Institute of Language Studies, Addis Ababa University, P. O. Box 1176, Addis Ababa, Ethiopia. Accepted 17 September, 2013 This study attempts to contribute to our knowledge about Gamo, a member of the North Ometo subgroup, which is one of the four subgroups that constitute the Ometo group of the Omotic language family (Fleming, 1976; Bender, 2000). This paper characterizes some of the issues in the research of language and identity. It will attempt to employ the complementary perspectives of sameness and difference between Gamo, its sisters in the North Ometo sub-branch and its dialects. North Ometo comprises of several related languages and dialects of which Gamo is one. The exact relationship amongst the Ometo languages is not well known. Not equally well known is the relationship Gamo has with its sisters and daughters. The study tries to address issues concerning with misrepresentation of the Gamo language by the existing classification in one hand and what the self perception of the Gamo community likes on the other hand. This study aimed at examining linguistic facts and the Gamo speakers’ own understandings of their identities. To this end, the study has used linguistic, anthropological and sociolinguists attempt to characterize membership of Gamo based on linguistic facts and members’ self ethno-linguistic identificationi. -

Food Supply Prospects - 2009
FOOD SUPPLY PROSPECTS - 2009 Disaster Management and Food Security Sector (DMFSS) Ministry of Agriculture and Rural Development (MoARD) Addis Ababa Ethiopia February 10, 2009 TABLE OF CONTENTS Pages LIST OF GLOSSARY OF LOCAL NAMES 2 ACRONYMS 3 EXECUTIVE SUMMARY 5 - 8 INTRODUCTION 9 - 12 REGIONAL SUMMARY 1. SOMALI 13 - 17 2. AMHARA 18 – 22 3. SNNPR 23 – 28 4. OROMIYA 29 – 32 5. TIGRAY 33 – 36 6. AFAR 37 – 40 7. BENSHANGUL GUMUZ 41 – 42 8. GAMBELLA 43 - 44 9. DIRE DAWA ADMINISTRATIVE COUNSEL 44 – 46 10. HARARI 47 - 48 ANNEX – 1 NEEDY POPULATION AND FOOD REQUIREMENT BY WOREDA 2 Glossary Azmera Rains from early March to early June (Tigray) Belg Short rainy season from February/March to June/July (National) Birkads cemented water reservoir Chat Mildly narcotic shrub grown as cash crop Dega Highlands (altitude>2500 meters) Deyr Short rains from October to November (Somali Region) Ellas Traditional deep wells Enset False Banana Plant Gena Belg season during February to May (Borena and Guji zones) Gu Main rains from March to June ( Somali Region) Haga Dry season from mid July to end of September (Southern zone of of Somali ) Hagaya Short rains from October to November (Borena/Bale) Jilal Long dry season from January to March ( Somali Region) Karan Rains from mid-July to September in the Northern zones of Somali region ( Jijiga and Shinile zones) Karma Main rains fro July to September (Afar) Kolla Lowlands (altitude <1500meters) Meher/Kiremt Main rainy season from June to September in crop dependent areas Sugum Short rains ( not more than 5 days -

Full Length Research Article DEVELOPMENT RESEARCH
Available online at http://www.journalijdr.com International Journal of DEVELOPMENT RESEARCH ISSN: 2230-9926 International Journal of Development Research Vol. 07, Issue, 01, pp.11119-11130, January, 2017 Full Length Research Article DETERMINANTS OF RURAL HOUSEHOLDS’ VULNERABILITY TO POVERTY IN CHENCHA AND ABAYA DISTRICTS, SOUTHERN ETHIOPIA *Fassil Eshetu Abebe Department of Economics, College of Business and Economics, Arba Minch University ARTICLE INFO ABSTRACT Article History: This study primarily aimed to examine the determinants of rural households’ vulnerability to Received 27th October, 2016 poverty and to profile the households according to their level of vulnerability using Feasible Received in revised form Generalized Least Square (FGLS) and Logistic Regression analysis with the help of data collected 28th November, 2016 from a sample of 500 households in two Woredas. The general poverty line of the study area was Accepted 14th December, 2016 determined to be Birr 248 per month per adult equivalent and 29.8 percent of the population in the th Published online 30 January, 2017 study areas were found to be poor. The projected consumption percapita after the three step FGLS estimation revealed that, the incidence of vulnerability to poverty in the area was 34.2 percent and Key Words: therefore, vulnerability was more spread in the study areas than ex post poverty. Using the two Poverty, Vulnerability, vulnerability thresholds, observed poverty rate (0.298) and vulnerability of 0.5, about 28.6%, Feasible Generalized Least Square, 5.6% and 65.8% of households were highly vulnerable, low vulnerable and not vulnerable Logit Model and Ethiopia. respectively. Most importantly, from the total poor households about 81.75%, 3.25% and 15% were highly vulnerable, low vulnerable and not vulnerable respectively. -

Land Use Patterns and Its Implication for Climate Change: the Case of Gamo Gofa, Southern Ethiopia
Defaru Debebe. et al., IJSRR 2013, 2(3), 155-173 Research article Available online www.ijsrr.org ISSN: 2279–0543 International Journal of Scientific Research and Reviews Land Use Patterns and its Implication for Climate Change: The Case of Gamo Gofa, Southern Ethiopia Defaru Debebe* and Tuma Ayele Arba Minch University P.O.Box 21, Arba Minch, Ethiopia ABSTRACT Land is one of three major factors of production in classical economics (along with labor and capital) and an essential input for housing and crop production. Land use is the backbone of agriculture and it provides substantial economic and social benefits. Assessing past-to present land use patterns associated with the crop production helps to understand which climatic effects might arise due to expanding crop cultivation. This study was conducted to evaluate the land use pattern and its implication for climate change in Gamo Gofa, Southern Ethiopia. For evaluation, correlation and time series trend analysis were used. Results revealed that a significant reduction in cultivable land, which was converted into cropland and might increase deforestation and greenhouse gas emission, in turn induce climate change. The correlation between cropland and fertile (cultivable) land (r=0.22674) in 2005 improved to (r=0.75734) in 2012 indicating major shift of fertile land to cropland in seven years interval. On other side, twelve years (1987-1999 and 2000-2011) average maximum temperature difference in Gamo Gafa was increased 0.425oC with standard deviation 0.331. It is statistically significant (t =1.284, alpha=0.10) at 10% level of error. Moreover, the spatial differences in climate change are likely to imply a heterogeneous pattern of land use responses. -

Hygienic Practice Among Milk and Cottage Cheese Handlers in Districts of Gamo and Gofa Zone, Southern Ethiopia
Research Article Volume 12:2, 2021 Journal of Veterinary Science & Technology ISSN: 2157-7579 Open Access Knowledge; Hygienic Practice among Milk and Cottage Cheese Handlers in Districts of Gamo and Gofa Zone, Southern Ethiopia Edget Alembo* Department of Animal Science, Arba Minch University, Arba Minch, Ethiopia Abstract A cross-sectional questionnaire survey was conducted in Arba Minch Zuria and Demba Gofa districts of Gamo and Gofa Zone of the Southern nation nationalities and people’s regional state with the objectives of assessing knowledge of hygienic practice of milk and cheese handlers in both study area. For this a total of 102 farmers who involved in milking, collecting and retailing of milk were included in the study area. Data obtained from questionnaire survey were analyzed by descriptive statistics and Chi –square test, using the Statistical package for social science (SPSS Version 17). The participants of this study were woman of different age group and 27(52.9%) of participants in Arba Minch Zuria and 32(64.7%) in Demba Gofa were >36 years old. The majority of participants 21(41.2%) and 22(43.1%) were educated up to grade 1-8 in Arba Minch Zuria and Demba Gofa, respectively. This had an impact on hygienic practice of milking and milk handling. The difference in hygienic handling, training obtained and cheese making practice among the study areas were statistically significant (p<0.05). There was also a statistically significant difference in hand washing and utensil as well as manner of washing between the two study areas (p<0.01). Finally this study revealed that there were no variation in Antibiotic usage and Practice of treating sick animal in both study area (p>0.05) with significant difference in Prognosis, Level of skin infection and Selling practice among study participants in both study areas (p<0.05). -

Visceral and Cutaneous Larva Migrans PAUL C
Visceral and Cutaneous Larva Migrans PAUL C. BEAVER, Ph.D. AMONG ANIMALS in general there is a In the development of our concepts of larva II. wide variety of parasitic infections in migrans there have been four major steps. The which larval stages migrate through and some¬ first, of course, was the discovery by Kirby- times later reside in the tissues of the host with¬ Smith and his associates some 30 years ago of out developing into fully mature adults. When nematode larvae in the skin of patients with such parasites are found in human hosts, the creeping eruption in Jacksonville, Fla. (6). infection may be referred to as larva migrans This was followed immediately by experi¬ although definition of this term is becoming mental proof by numerous workers that the increasingly difficult. The organisms impli¬ larvae of A. braziliense readily penetrate the cated in infections of this type include certain human skin and produce severe, typical creep¬ species of arthropods, flatworms, and nema¬ ing eruption. todes, but more especially the nematodes. From a practical point of view these demon¬ As generally used, the term larva migrans strations were perhaps too conclusive in that refers particularly to the migration of dog and they encouraged the impression that A. brazil¬ cat hookworm larvae in the human skin (cu¬ iense was the only cause of creeping eruption, taneous larva migrans or creeping eruption) and detracted from equally conclusive demon¬ and the migration of dog and cat ascarids in strations that other species of nematode larvae the viscera (visceral larva migrans). In a still have the ability to produce similarly the pro¬ more restricted sense, the terms cutaneous larva gressive linear lesions characteristic of creep¬ migrans and visceral larva migrans are some¬ ing eruption. -

Agency Deyr/Karan 2012 Seasonal
Food Supply Prospects FOR THE YEAR 2013 ______________________________________________________________________________ Disaster Risk Management and Food Security Sector (DRMFSS) Ministry of Agriculture (MoA) March 2013 Addis Ababa, Ethiopia Table of Contents Glossary ................................................................................................................. 2 Acronyms ............................................................................................................... 3 EXCUTIVE SUMMARY ............................................................................................. 4 INTRODUCTION.................................................................................................... 11 REGIONAL SUMMARY OF FOOD SUPPLY PROSPECT ............................................. 14 SOMALI ............................................................................................................. 14 OROMIA ........................................................................................................... 21 TIGRAY .............................................................................................................. 27 AMHARA ........................................................................................................... 31 AFAR ................................................................................................................. 34 BENISHANGUL GUMUZ ..................................................................................... 37 SNNP ............................................................................................................... -

Public Health Significance of Intestinal Parasitic Infections*
Articles in the Update series Les articles de la rubrique give a concise, authoritative, Le pointfournissent un bilan and up-to-date survey of concis et fiable de la situa- the present position in the tion actuelle dans les do- Update selectedfields, coveringmany maines consideres, couvrant different aspects of the de nombreux aspects des biomedical sciences and sciences biomedicales et de la , po n t , , public health. Most of santepublique. Laplupartde the articles are written by ces articles auront donc ete acknowledged experts on the redigeis par les specialistes subject. les plus autorises. Bulletin of the World Health Organization, 65 (5): 575-588 (1987) © World Health Organization 1987 Public health significance of intestinal parasitic infections* WHO EXPERT COMMITTEE' Intestinal parasitic infections are distributed virtually throughout the world, with high prevalence rates in many regions. Amoebiasis, ascariasis, hookworm infection and trichuriasis are among the ten most common infections in the world. Other parasitic infections such as abdominal angiostrongyliasis, intestinal capil- lariasis, and strongyloidiasis are of local or regional public health concern. The prevention and control of these infections are now more feasible than ever before owing to the discovery of safe and efficacious drugs, the improvement and sim- plification of some diagnostic procedures, and advances in parasite population biology. METHODS OF ASSESSMENT The amount of harm caused by intestinal parasitic infections to the health and welfare of individuals and communities depends on: (a) the parasite species; (b) the intensity and course of the infection; (c) the nature of the interactions between the parasite species and concurrent infections; (d) the nutritional and immunological status of the population; and (e) numerous socioeconomic factors. -

Ethiopia Round 6 SDP Questionnaire
Ethiopia Round 6 SDP Questionnaire Always 001a. Your name: [NAME] Is this your name? ◯ Yes ◯ No 001b. Enter your name below. 001a = 0 Please record your name 002a = 0 Day: 002b. Record the correct date and time. Month: Year: ◯ TIGRAY ◯ AFAR ◯ AMHARA ◯ OROMIYA ◯ SOMALIE BENISHANGUL GUMZ 003a. Region ◯ ◯ S.N.N.P ◯ GAMBELA ◯ HARARI ◯ ADDIS ABABA ◯ DIRE DAWA filter_list=${this_country} ◯ NORTH WEST TIGRAY ◯ CENTRAL TIGRAY ◯ EASTERN TIGRAY ◯ SOUTHERN TIGRAY ◯ WESTERN TIGRAY ◯ MEKELE TOWN SPECIAL ◯ ZONE 1 ◯ ZONE 2 ◯ ZONE 3 ZONE 5 003b. Zone ◯ ◯ NORTH GONDAR ◯ SOUTH GONDAR ◯ NORTH WELLO ◯ SOUTH WELLO ◯ NORTH SHEWA ◯ EAST GOJAM ◯ WEST GOJAM ◯ WAG HIMRA ◯ AWI ◯ OROMIYA 1 ◯ BAHIR DAR SPECIAL ◯ WEST WELLEGA ◯ EAST WELLEGA ◯ ILU ABA BORA ◯ JIMMA ◯ WEST SHEWA ◯ NORTH SHEWA ◯ EAST SHEWA ◯ ARSI ◯ WEST HARARGE ◯ EAST HARARGE ◯ BALE ◯ SOUTH WEST SHEWA ◯ GUJI ◯ ADAMA SPECIAL ◯ WEST ARSI ◯ KELEM WELLEGA ◯ HORO GUDRU WELLEGA ◯ Shinile ◯ Jijiga ◯ Liben ◯ METEKEL ◯ ASOSA ◯ PAWE SPECIAL ◯ GURAGE ◯ HADIYA ◯ KEMBATA TIBARO ◯ SIDAMA ◯ GEDEO ◯ WOLAYITA ◯ SOUTH OMO ◯ SHEKA ◯ KEFA ◯ GAMO GOFA ◯ BENCH MAJI ◯ AMARO SPECIAL ◯ DAWURO ◯ SILTIE ◯ ALABA SPECIAL ◯ HAWASSA CITY ADMINISTRATION ◯ AGNEWAK ◯ MEJENGER ◯ HARARI ◯ AKAKI KALITY ◯ NEFAS SILK-LAFTO ◯ KOLFE KERANIYO 2 ◯ GULELE ◯ LIDETA ◯ KIRKOS-SUB CITY ◯ ARADA ◯ ADDIS KETEMA ◯ YEKA ◯ BOLE ◯ DIRE DAWA filter_list=${level1} ◯ TAHTAY ADIYABO ◯ MEDEBAY ZANA ◯ TSELEMTI ◯ SHIRE ENIDASILASE/TOWN/ ◯ AHIFEROM ◯ ADWA ◯ TAHTAY MAYCHEW ◯ NADER ADET ◯ DEGUA TEMBEN ◯ ABIYI ADI/TOWN/ ◯ ADWA/TOWN/ ◯ AXUM/TOWN/ ◯ SAESI TSADAMBA ◯ KLITE -

Imaging Parasitic Diseases
Insights Imaging (2017) 8:101–125 DOI 10.1007/s13244-016-0525-2 REVIEW Unexpected hosts: imaging parasitic diseases Pablo Rodríguez Carnero1 & Paula Hernández Mateo2 & Susana Martín-Garre2 & Ángela García Pérez3 & Lourdes del Campo1 Received: 8 June 2016 /Revised: 8 September 2016 /Accepted: 28 September 2016 /Published online: 23 November 2016 # The Author(s) 2016. This article is published with open access at Springerlink.com Abstract Radiologists seldom encounter parasitic dis- • Some parasitic diseases are still endemic in certain regions eases in their daily practice in most of Europe, although in Europe. the incidence of these diseases is increasing due to mi- • Parasitic diseases can have complex life cycles often involv- gration and tourism from/to endemic areas. Moreover, ing different hosts. some parasitic diseases are still endemic in certain • Prompt diagnosis and treatment is essential for patient man- European regions, and immunocompromised individuals agement in parasitic diseases. also pose a higher risk of developing these conditions. • Radiologists should be able to recognise and suspect the This article reviews and summarises the imaging find- most relevant parasitic diseases. ings of some of the most important and frequent human parasitic diseases, including information about the para- Keywords Parasitic diseases . Radiology . Ultrasound . site’s life cycle, pathophysiology, clinical findings, diag- Multidetector computed tomography . Magnetic resonance nosis, and treatment. We include malaria, amoebiasis, imaging toxoplasmosis, trypanosomiasis, leishmaniasis, echino- coccosis, cysticercosis, clonorchiasis, schistosomiasis, fascioliasis, ascariasis, anisakiasis, dracunculiasis, and Introduction strongyloidiasis. The aim of this review is to help radi- ologists when dealing with these diseases or in cases Parasites are organisms that live in another organism at the where they are suspected. -

Dracunculiasis in Oral and Maxillofacial Surgery
http://dx.doi.org/10.5125/jkaoms.2016.42.2.67 INVITED SPECIAL ARTICLE pISSN 2234-7550·eISSN 2234-5930 Dracunculiasis in oral and maxillofacial surgery Soung Min Kim1,2 1Oral and Maxillofacial Microvascular Reconstruction Lab, Sunyani Regional Hospital, Sunyani, Brong Ahafo, Ghana, 2Department of Oral and Maxillofacial Surgery, Dental Research Institute, School of Dentistry, Seoul National University, Seoul, Korea Abstract (J Korean Assoc Oral Maxillofac Surg 2016;42:67-76) Dracunculiasis, otherwise known as guinea worm disease (GWD), is caused by infection with the nematode Dracunculus medinensis. This nematode is transmitted to humans exclusively via contaminated drinking water. The transmitting vectors are Cyclops copepods (water fleas), which are tiny free- swimming crustaceans usually found abundantly in freshwater ponds. Humans can acquire GWD by drinking water that contains vectors infected with guinea worm larvae. This disease is prevalent in some of the most deprived areas of the world, and no vaccine or medicine is currently available. Inter- national efforts to eradicate dracunculiasis began in the early 1980s. Most dentists and maxillofacial surgeons have neglected this kind of parasite infec- tion. However, when performing charitable work in developing countries near the tropic lines or other regions where GWD is endemic, it is important to consider GWD in cases of swelling or tumors of unknown origin. This paper reviews the pathogenesis, epidemiology, clinical criteria, diagnostic criteria, treatment, and prevention of dracunculiasis. It also summarizes important factors for maxillofacial surgeons to consider. Key words: Dracunculiasis, Dracunculus medinensis, Guinea worm disease, Neglected tropical diseases, Swelling of unknown origin [paper submitted 2016. 3. 24 / accepted 2016.