Suppression of Cutinase Gene Expression in Isogenic Hypovirulent Strains Containing Double-Stranded Rnas D
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Ribonuclease A: Disulfide Bonds, Conformational Stability, and Cytotoxicity
Ribonuclease A: Disulfide Bonds, Conformational Stability, and Cytotoxicity by Tony A. Klink A dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy (Biochemistry) at the UNIVERSITY OF WISCONSIN-MADISON 2()()() r OJ A dissertation entitled Ribonuclease A: Disulfide Bonds, Conformational Stability and Cytotoxicity submitted to the Graduate School of the University of Wisconsin-Madison in partial fulfillment of the requirements for the degree of Doctor of Philosophy by Tony Anthony Klink Date of Final Oral Examination: August 4, 2000 Month & Year Degree to be awarded: December May August 2000 • * * * * * * • * * • * • • • • • * • • • * • • • • • • • • • • • • • * * • * • * * * • * • * • • • • • • * App oval Signature. f Dissertation Readers: Signature, Dean of Graduate School ---..., v.C~vJ,1A. 5. lJ,~k;at 1 Abstract Disulfide bonds between the side chains of cysteine residues are the only common cross links in proteins. Bovine pancreatic ribonuclease A (RNase A) is a 124-residue enzyme that contains four interweaving disulfide bonds (Cys26-Cys84, Cys40-Cys95, Cys58-CysllO, and Cys65-Cys72) and catalyzes the cleavage of RNA. The contribution of each disulfide bond to the confonnational stability and catalytic activity of RNase A was detennined using variants in which each cystine was replaced independently with a pair of alanine residues. Of the four disulfide bonds, the Cys40-Cys95 and Cys65-Cys72 cross-links are the least important to confonnational stability. Removing these disulfide bonds leads to RNase A variants that have Tm values below that of the wild-type enzyme but above physiological temperature. Unlike wild-type RNase A. G88R RNase A is toxic to cancer cells. To investigate the relationship between conformational stability and cytotoxicity, the C40AlC95A and C65A1C72A variants were made in the G88R background. -

Determination of Pk, Values of the Histidine Side
Protein Science (1997), 6:1937-1944. Cambridge University Press. Printed in the USA Copyright 0 1997 The Protein Society Determination of pK, values of the histidine side chains of phosphatidylinositol-specific phospholipase C from Bacillus cereus by NMR spectroscopy and site-directed mutagenesis TUN LIU, MARGRET RYAN, FREDERICK W. DAHLQUIST, AND 0. HAYES GRIFFITH Institute of Molecular Biology and Department of Chemistry, University of Oregon, Eugene, Oregon 97403 (RECEIVEDDecember 4, 1996: ACCEPTEDMay 19, 1997) Abstract Two active site histidine residues have been implicated in the catalysis of phosphatidylinositol-specific phospholipase C (PI-PLC). In this report, we present the first study of the pK,, values of histidines of a PI-PLC. All six histidines of Bacillus cereus PI-PLC were studied by 2D NMR spectroscopy and site-directed mutagenesis. The protein was selec- tively labeled with '3C"-histidine. A series of 'H-I3C HSQC NMR spectra were acquired over a pH range of 4.0-9.0. Five of the six histidines have been individually substituted with alanine to aid the resonance assignments in the NMR spectra. Overall, the remaining histidines in the mutants show little chemical shift changes in the 'H-"C HSQC spectra, indicating that the alanine substitution has no effect on the tertiary structure of the protein. H32A and H82A mutants are inactive enzymes, while H92A and H61A are fully active, and H81A retains about 15% of the wild-type activity. The active site histidines, His32 and His82, display pK,, values of 7.6 and 6.9, respectively. His92 and His227 exhibit pK, values of 5.4 and 6.9. -

Families and the Structural Relatedness Among Globular Proteins
Protein Science (1993), 2, 884-899. Cambridge University Press. Printed in the USA. Copyright 0 1993 The Protein Society -~~ ~~~~ ~ Families and the structural relatedness among globular proteins DAVID P. YEE AND KEN A. DILL Department of Pharmaceutical Chemistry, University of California, San Francisco, California94143-1204 (RECEIVEDJanuary 6, 1993; REVISEDMANUSCRIPT RECEIVED February 18, 1993) Abstract Protein structures come in families. Are families “closely knit” or “loosely knit” entities? We describe a mea- sure of relatedness among polymer conformations. Based on weighted distance maps, this measure differs from existing measures mainly in two respects: (1) it is computationally fast, and (2) it can compare any two proteins, regardless of their relative chain lengths or degree of similarity. It does not require finding relative alignments. The measure is used here to determine the dissimilarities between all 12,403 possible pairs of 158 diverse protein structures from the Brookhaven Protein Data Bank (PDB). Combined with minimal spanning trees and hier- archical clustering methods,this measure is used to define structural families. It is also useful for rapidly searching a dataset of protein structures for specific substructural motifs.By using an analogy to distributions of Euclid- ean distances, we find that protein families are not tightly knit entities. Keywords: protein family; relatedness; structural comparison; substructure searches Pioneering work over the past 20 years has shown that positions after superposition. RMS is a useful distance proteins fall into families of related structures (Levitt & metric for comparingstructures that arenearly identical: Chothia, 1976; Richardson, 1981; Richardson & Richard- for example, when refining or comparing structures ob- son, 1989; Chothia & Finkelstein, 1990). -

Structures, Functions, and Mechanisms of Filament Forming Enzymes: a Renaissance of Enzyme Filamentation
Structures, Functions, and Mechanisms of Filament Forming Enzymes: A Renaissance of Enzyme Filamentation A Review By Chad K. Park & Nancy C. Horton Department of Molecular and Cellular Biology University of Arizona Tucson, AZ 85721 N. C. Horton ([email protected], ORCID: 0000-0003-2710-8284) C. K. Park ([email protected], ORCID: 0000-0003-1089-9091) Keywords: Enzyme, Regulation, DNA binding, Nuclease, Run-On Oligomerization, self-association 1 Abstract Filament formation by non-cytoskeletal enzymes has been known for decades, yet only relatively recently has its wide-spread role in enzyme regulation and biology come to be appreciated. This comprehensive review summarizes what is known for each enzyme confirmed to form filamentous structures in vitro, and for the many that are known only to form large self-assemblies within cells. For some enzymes, studies describing both the in vitro filamentous structures and cellular self-assembly formation are also known and described. Special attention is paid to the detailed structures of each type of enzyme filament, as well as the roles the structures play in enzyme regulation and in biology. Where it is known or hypothesized, the advantages conferred by enzyme filamentation are reviewed. Finally, the similarities, differences, and comparison to the SgrAI system are also highlighted. 2 Contents INTRODUCTION…………………………………………………………..4 STRUCTURALLY CHARACTERIZED ENZYME FILAMENTS…….5 Acetyl CoA Carboxylase (ACC)……………………………………………………………………5 Phosphofructokinase (PFK)……………………………………………………………………….6 -

Purification and Characterization of Cutinase from Venturia Inaequalis
Physiology and Biochemistry Purification and Characterization of Cutinase from Venturia inaequalis Wolfram K611er and Diana M. Parker Assistant professor and research assistant, Department of Plant Pathology, Cornell University, New York State Agricultural Experiment Station, Geneva 14456. We thank Professor A. L. Jones for the isolates of Venturia inaequalis, Mr. Robert W. Ennis, Jr. for his help in cutin preparation, and Comstock Foods, Alton, NY, for the apple peels. Accepted for publication 16 August 1988. ABSTRACT K61ler, W., and Parker, D. M. 1989. Purification and characterization of cutinase from Venturia inaequalis. Phytopathology 79:278-283. Venturia inaequalis was grown in a culture medium containing purified cutinase from V. inaequalisis optimal at a pH of 6 and thus different from apple cutin as the sole carbon source. After 8 wk of growth an esterase was the alkaline pH-optimum reported for other purified cutinases. The isolated from the culture fluid and purified to apparent homogeneity. The hydrolysis of the model esterp-nitrophenyl butyrate was less affected by the enzyme hydrolyzed tritiated cutin and thus was identified as cutinase. The pH. The esterase activity was strongly inhibited by diisopropyl purified cutinase is a glycoprotein with a molecular mass of 21-23 kg/ mol, fluorophosphate, and the phosphorylation of one serine was sufficient for as determined by various procedures. Remarkable structural features are a complete inhibition. Thus, cutinase from V. inaequalis belongs to the class high content of glycine, a high content of nonpolar amino acids, two of serine hydrolases, a characterisitic shared with other fungal cutinases. disulfide bridges, and a high degree of hydrophobicity. -

Ribonuclease A: Exploring the Function of the Active-Site Lysine
Ribonuclease A: Exploring the Function of the Active-Site Lysine Residue in Catalysis and Inhibition By June M. Messmore A dissertation submitted in partial fulfillment of the degree of Doctor of Philosophy (Biochemistry) UNIVERSITY OF WISCONSIN-MADISON 1999 A dissertation entitled Ribonuclease A: Exploring the Function of the Active-Site Lysine Residue in Catalysis and Inhibition submitted to the Graduate School of the University of Wisconsin-Madison Jp'part.i~l f~lfillment of ~~ereq~ire~~nt$ for the degree of Doctor·of Philosophy by June M. Messmore Degree to be awarded: December 19_ May 19_ August 19~ th May 28 , 1999 Date of Examination . ~ r:i~ b lli{\~~ Dean. Graduate School 1 Ribonuclease A: Exploring the Function of an Active-Site Lysine in Catalysis and Inhibition June M. Messmore Under the supervision of Professor Ronald T. Raines at the University of Wisconsin-Madison ABSTRACT Structural analyses had suggested that the active-site lysine residue of RNase A (Lys41) may interact preferentially with the transition state for covalent bond cleavage, thus facilitating catalysis. Site-directed mutagenesis and chemical modification were combined (1) to probe the role of Lys41 in catalysis, (2) to provide a chemical on/off switch for RNase A activity in in vitro applications, and (3) to probe the analogy ofuridine 2',3'-cyclic vanadate to the catalytic transition state. In addition to studying inhibition by the uridine vanadate, inhibition by a polyvanadate species was also characterized. Results indicate the importance of positive charge and the donation of a single hydrogen bond in catalysis. Chemical modification can reversibly modulate the activity of ribonuclease by a factor of 30,000. -

Hypoxic Regulation of the 6-Phosphofructo-2-Kinase/Fructose
FEBS Letters 554 (2003) 264^270 FEBS 27797 CORE Metadata, citation and similar papers at core.ac.uk Provided by Elsevier -Hypoxic Publisher Connector regulation of the 6-phosphofructo-2-kinase/fructose-2,6- bisphosphatase gene family (PFKFB-1^4) expression in vivo Oleksandr Minchenkoa;b, Iryna Opentanovaa, Jaime Carob;Ã aDepartment of Molecular Biology, Institute of Biochemistry, National Academy of Science of Ukraine, Kyiv 01601, Ukraine bCardeza Foundation for Hematologic Research, Department of Medicine, Je¡erson Medical College of Thomas Je¡erson University, Curtis Bldg. 810, 1015 Walnut Street, Philadelphia, PA 19107, USA Received 12 May 2003; revised 10 September 2003; accepted 6 October 2003 First published online 21October 2003 Edited by Vladimir Skulachev 1. Introduction Abstract When oxygen becomes limiting, cells shift primarily to a glycolytic mode for generation of energy. A key regulator of glycolytic £ux is fructose-2,6-bisphosphate (F-2,6-BP), a po- The ability to respond to hypoxia is an essential evolution- tent allosteric regulator of 6-phosphofructo-1-kinase (PFK-1). ary adaptation in higher vertebrates. Hypoxia could be caused The levels of F-2,6-BP are maintained by a family of bifunc- by a generalized reduction in oxygen delivery, such as in alti- tional enzymes, 6-phosphofructo-2-kinase/fructose-2,6-bisphos- tude and pulmonary diseases, or by disruption in the local phatase (PFKFBor PFK-2), which have both kinase and phos- blood supply, such as in ischemic disorders. Important in phatase activities. Each member of the enzyme family is the adaptations to hypoxia is the activation of genes that characterized by their phosphatase:kinase activity ratio (K:B) ameliorate or compensate for the oxygen de¢cit. -

A Novel Cell Death Gene Acts to Repair Patterning Defects in Drosophila Melanogaster
NOTE A Novel Cell Death Gene Acts to Repair Patterning Defects in Drosophila melanogaster Kentaro M. Tanaka,*,†,1 Aya Takahashi,*,‡,§ Naoyuki Fuse,** and Toshiyuki Takano-Shimizu-Kouno*,††,1 *Department of Population Genetics, National Institute of Genetics, Yata 1111, Mishima, Shizuoka, 411-8540, Japan, †Department of Biological and Medical Sciences, Oxford Brookes University, Oxford, OX3 0BP, United Kingdom, ‡Department of Biological Sciences and §Research Center for Genomics and Bioinformatics, Tokyo Metropolitan University, Hachioji, 192-0397, Japan, **Department of Biophysics, Kyoto University, Kitashirakawa-Oiwake-cho, Sakyo-ku, Kyoto, 606-8502, Japan, and ††Drosophila Genetic Resource Center, Kyoto Institute of Technology, Saga Ippongi-cho, Ukyo-ku, Kyoto, 616-8354, Japan ABSTRACT Cell death is a mechanism utilized by organisms to eliminate excess cells during development. Here, we describe a novel regulator of caspase-independent cell death, Mabiki (Mabi), that is involved in the repair of the head patterning defects caused by extra copies of bicoid in Drosophila melanogaster. Mabiki functions together with caspase-dependent cell death mechanisms to provide robustness during development. RGANISMS are surprisingly robust to various stresses and embryos. However, the mechanism leading to this compensa- Operturbations. Elimination of undesirable cells is one tory cell death remains largely unexplored. mechanism that ensures robust development. For example, To identify genes involved in this repair, we first screened compensatory cell death is observed in the expanded prospec- a panel of 152 autosomal deficiencies for those with signifi- tive head region of embryos from mothers carrying extra (six) cantly lower viability in 6xbcd than in normal (2xbcd)condi- copies of bicoid (6xbcd)inDrosophila melanogaster. -

Supplemental Table 7. Every Significant Association
Supplemental Table 7. Every significant association between an individual covariate and functional group (assigned to the KO level) as determined by CPGLM regression analysis. Variable Unit RelationshipLabel See also CBCL Aggressive Behavior K05914 + CBCL Emotionally Reactive K05914 + CBCL Externalizing Behavior K05914 + K15665 K15658 CBCL Total K05914 + K15660 K16130 KO: E1.13.12.7; photinus-luciferin 4-monooxygenase (ATP-hydrolysing) [EC:1.13.12.7] :: PFAMS: AMP-binding enzyme; CBQ Inhibitory Control K05914 - K12239 K16120 Condensation domain; Methyltransferase domain; Thioesterase domain; AMP-binding enzyme C-terminal domain LEC Family Separation/Social Services K05914 + K16129 K16416 LEC Poverty Related Events K05914 + K16124 LEC Total K05914 + LEC Turmoil K05914 + CBCL Aggressive Behavior K15665 + CBCL Anxious Depressed K15665 + CBCL Emotionally Reactive K15665 + K05914 K15658 CBCL Externalizing Behavior K15665 + K15660 K16130 KO: K15665, ppsB, fenD; fengycin family lipopeptide synthetase B :: PFAMS: Condensation domain; AMP-binding enzyme; CBCL Total K15665 + K12239 K16120 Phosphopantetheine attachment site; AMP-binding enzyme C-terminal domain; Transferase family CBQ Inhibitory Control K15665 - K16129 K16416 LEC Poverty Related Events K15665 + K16124 LEC Total K15665 + LEC Turmoil K15665 + CBCL Aggressive Behavior K11903 + CBCL Anxiety Problems K11903 + CBCL Anxious Depressed K11903 + CBCL Depressive Problems K11903 + LEC Turmoil K11903 + MODS: Type VI secretion system K01220 K01058 CBCL Anxiety Problems K11906 + CBCL Depressive -

Biochemical and Structural Analysis of Thermostable Esterases
Biochemical and Structural Analysis of Thermostable Esterases Mark Levisson Thesis supervisors: Prof. dr. J. van der Oost Personal chair at the Laboratory of Microbiology Wageningen University Prof. dr. W.M. de Vos Professor of Microbiology Wageningen University Thesis co-supervisor: Dr. S.W.M. Kengen Assistant Professor at the Laboratory of Microbiology Wageningen University Other members: Prof. dr. ir. H. Gruppen Wageningen University Dr. M.C.R Franssen Wageningen University Dr. T. Sonke DSM, Geleen Prof. dr. B.W. Dijkstra University of Groningen Dr. A.M.W.H Thunnissen University of Groningen This research was conducted under the auspices of the Graduate School VLAG Biochemical and Structural Analysis of Thermostable Esterases Mark Levisson Thesis submitted in partial fulfilment of the requirements for the degree of doctor at Wageningen University by the authority of the Rector Magnificus Prof. dr. M.J. Kropff, in the presence of the Thesis Committee appointed by the Doctorate Board to be defended in public on Monday 28 September 2009 at 4 PM in the Aula Levisson, M. Biochemical and Structural Analysis of Thermostable Esterases PhD Thesis, Wageningen University, Wageningen, The Netherlands (2009) 170 pages - With references, and with summaries in English and Dutch ISBN: 978-90-8585-459-3 TABLE OF contents Aim and outline 1 Chapter 1 Introduction 5 Chapter 2 Characterization and structural modeling of a new type of 21 thermostable esterase from Thermotoga maritima Chapter 3 Crystallization and preliminary crystallographic analysis of -

Legionella Pneumophila Type II Secretome Reveals Unique Exoproteins and a Chitinase That Promotes Bacterial Persistence in the Lung
Legionella pneumophila type II secretome reveals unique exoproteins and a chitinase that promotes bacterial persistence in the lung Sruti DebRoy, Jenny Dao, Maria So¨ derberg, Ombeline Rossier, and Nicholas P. Cianciotto* Department of Microbiology–Immunology, Northwestern University Medical School, Chicago, IL 60611 Edited by Thomas J. Silhavy, Princeton University, Princeton, NJ, and approved October 27, 2006 (received for review September 20, 2006) Type II protein secretion is critical for Legionella pneumophila acid phosphatases, lipases, phospholipase A, phospholipase C, infection of amoebae, macrophages, and mice. Previously, we lysophospholipase A, cholesterol acyltransferase, and ribonucle- found several enzymes to be secreted by this (Lsp) secretory ase (4–6, 10–15). Both the Sec and Tat pathways are implicated pathway. To better define the L. pneumophila type II secretome, a in the processing of L. pneumophila type II substrates (16). 2D electrophoresis proteomic approach was used to compare Because mutants lacking individual type II effectors infect proteins in wild-type and type II mutant supernatants. We identi- normally (10, 12–14, 17, 18), we hypothesize that more proteins fied 20 proteins that are type II-dependent, including aminopep- are secreted by Lsp, and that one or more of them are virulence tidases, an RNase, and chitinase, as well as proteins with no determinants. Although L. pneumophila has been sequenced (19, homology to known proteins. Because a chitinase had not been 20), it is not possible to readily identify effectors by bioinfor- previously reported in Legionella, we determined that wild type matics, because there is no motif that specifically defines a secretes activity against both p-nitrophenyl triacetyl chitotriose protein as a type II substrate (3). -
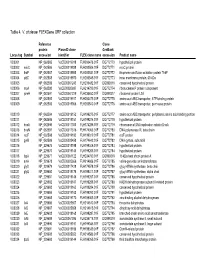
Table 4. V. Cholerae Flexgene ORF Collection
Table 4. V. cholerae FLEXGene ORF collection Reference Clone protein PlasmID clone GenBank Locus tag Symbol accession identifier FLEX clone name accession Product name VC0001 NP_062585 VcCD00019918 FLH200476.01F DQ772770 hypothetical protein VC0002 mioC NP_062586 VcCD00019938 FLH200506.01F DQ772771 mioC protein VC0003 thdF NP_062587 VcCD00019958 FLH200531.01F DQ772772 thiophene and furan oxidation protein ThdF VC0004 yidC NP_062588 VcCD00019970 FLH200545.01F DQ772773 inner membrane protein, 60 kDa VC0005 NP_062589 VcCD00061243 FLH236482.01F DQ899316 conserved hypothetical protein VC0006 rnpA NP_062590 VcCD00025697 FLH214799.01F DQ772774 ribonuclease P protein component VC0007 rpmH NP_062591 VcCD00061229 FLH236450.01F DQ899317 ribosomal protein L34 VC0008 NP_062592 VcCD00019917 FLH200475.01F DQ772775 amino acid ABC transporter, ATP-binding protein VC0009 NP_062593 VcCD00019966 FLH200540.01F DQ772776 amino acid ABC transproter, permease protein VC0010 NP_062594 VcCD00019152 FLH199275.01F DQ772777 amino acid ABC transporter, periplasmic amino acid-binding portion VC0011 NP_062595 VcCD00019151 FLH199274.01F DQ772778 hypothetical protein VC0012 dnaA NP_062596 VcCD00017363 FLH174286.01F DQ772779 chromosomal DNA replication initiator DnaA VC0013 dnaN NP_062597 VcCD00017316 FLH174063.01F DQ772780 DNA polymerase III, beta chain VC0014 recF NP_062598 VcCD00019182 FLH199319.01F DQ772781 recF protein VC0015 gyrB NP_062599 VcCD00025458 FLH174642.01F DQ772782 DNA gyrase, subunit B VC0016 NP_229675 VcCD00019198 FLH199346.01F DQ772783 hypothetical protein