THE CONTRIBUTION of HEADWATER STREAMS to BIODIVERSITY in RIVER Networksl
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Aquatic Insects Are Dramatically Underrepresented in Genomic Research
insects Communication Aquatic Insects Are Dramatically Underrepresented in Genomic Research Scott Hotaling 1,* , Joanna L. Kelley 1 and Paul B. Frandsen 2,3,* 1 School of Biological Sciences, Washington State University, Pullman, WA 99164, USA; [email protected] 2 Department of Plant and Wildlife Sciences, Brigham Young University, Provo, UT 84062, USA 3 Data Science Lab, Smithsonian Institution, Washington, DC 20002, USA * Correspondence: [email protected] (S.H.); [email protected] (P.B.F.); Tel.: +1-(828)-507-9950 (S.H.); +1-(801)-422-2283 (P.B.F.) Received: 20 August 2020; Accepted: 3 September 2020; Published: 5 September 2020 Simple Summary: The genome is the basic evolutionary unit underpinning life on Earth. Knowing its sequence, including the many thousands of genes coding for proteins in an organism, empowers scientific discovery for both the focal organism and related species. Aquatic insects represent 10% of all insect diversity, can be found on every continent except Antarctica, and are key components of freshwater ecosystems. However, aquatic insect genome biology lags dramatically behind that of terrestrial insects. If genomic effort was spread evenly, one aquatic insect genome would be sequenced for every ~9 terrestrial insect genomes. Instead, ~24 terrestrial insect genomes have been sequenced for every aquatic insect genome. A lack of aquatic genomes is limiting research progress in the field at both fundamental and applied scales. We argue that the limited availability of aquatic insect genomes is not due to practical limitations—small body sizes or overly complex genomes—but instead reflects a lack of research interest. We call for targeted efforts to expand the availability of aquatic insect genomic resources to empower future research. -

Research Funding (Total $2,552,481) $15,000 2019
CURRICULUM VITAE TENNESSEE AQUARIUM CONSERVATION INSTITUTE 175 BAYLOR SCHOOL RD CHATTANOOGA, TN 37405 RESEARCH FUNDING (TOTAL $2,552,481) $15,000 2019. Global Wildlife Conservation. Rediscovering the critically endangered Syr-Darya Shovelnose Sturgeon. $10,000 2019. Tennessee Wildlife Resources Agency. Propagation of the Common Logperch as a host for endangered mussel larvae. $8,420 2019. Tennessee Wildlife Resources Agency. Monitoring for the Laurel Dace. $4,417 2019. Tennessee Wildlife Resources Agency. Examining interactions between Laurel Dace (Chrosomus saylori) and sunfish $12,670 2019. Trout Unlimited. Southern Appalachian Brook Trout propagation for reintroduction to Shell Creek. $106,851 2019. Private Donation. Microplastic accumulation in fishes of the southeast. $1,471. 2019. AZFA-Clark Waldram Conservation Grant. Mayfly propagation for captive propagation programs. $20,000. 2019. Tennessee Valley Authority. Assessment of genetic diversity within Blotchside Logperch. $25,000. 2019. Riverview Foundation. Launching Hidden Rivers in the Southeast. $11,170. 2018. Trout Unlimited. Propagation of Southern Appalachian Brook Trout for Supplemental Reintroduction. $1,471. 2018. AZFA Clark Waldram Conservation Grant. Climate Change Impacts on Headwater Stream Vertebrates in Southeastern United States $1,000. 2018. Hamilton County Health Department. Step 1 Teaching Garden Grants for Sequoyah School Garden. $41,000. 2018. Riverview Foundation. River Teachers: Workshops for Educators. $1,000. 2018. Tennessee Valley Authority. Youth Freshwater Summit $20,000. 2017. Tennessee Valley Authority. Lake Sturgeon Propagation. $7,500 2017. Trout Unlimited. Brook Trout Propagation. $24,783. 2017. Tennessee Wildlife Resource Agency. Assessment of Percina macrocephala and Etheostoma cinereum populations within the Duck River Basin. $35,000. 2017. U.S. Fish and Wildlife Service. Status surveys for conservation status of Ashy (Etheostoma cinereum) and Redlips (Etheostoma maydeni) Darters. -

Data Quality, Performance, and Uncertainty in Taxonomic Identification for Biological Assessments
J. N. Am. Benthol. Soc., 2008, 27(4):906–919 Ó 2008 by The North American Benthological Society DOI: 10.1899/07-175.1 Published online: 28 October 2008 Data quality, performance, and uncertainty in taxonomic identification for biological assessments 1 2 James B. Stribling AND Kristen L. Pavlik Tetra Tech, Inc., 400 Red Brook Blvd., Suite 200, Owings Mills, Maryland 21117-5159 USA Susan M. Holdsworth3 Office of Wetlands, Oceans, and Watersheds, US Environmental Protection Agency, 1200 Pennsylvania Ave., NW, Mail Code 4503T, Washington, DC 20460 USA Erik W. Leppo4 Tetra Tech, Inc., 400 Red Brook Blvd., Suite 200, Owings Mills, Maryland 21117-5159 USA Abstract. Taxonomic identifications are central to biological assessment; thus, documenting and reporting uncertainty associated with identifications is critical. The presumption that comparable results would be obtained, regardless of which or how many taxonomists were used to identify samples, lies at the core of any assessment. As part of a national survey of streams, 741 benthic macroinvertebrate samples were collected throughout the eastern USA, subsampled in laboratories to ;500 organisms/sample, and sent to taxonomists for identification and enumeration. Primary identifications were done by 25 taxonomists in 8 laboratories. For each laboratory, ;10% of the samples were randomly selected for quality control (QC) reidentification and sent to an independent taxonomist in a separate laboratory (total n ¼ 74), and the 2 sets of results were compared directly. The results of the sample-based comparisons were summarized as % taxonomic disagreement (PTD) and % difference in enumeration (PDE). Across the set of QC samples, mean values of PTD and PDE were ;21 and 2.6%, respectively. -

( ) Hydropsychidae (Insecta: Trichoptera) As Bio-Indicators Of
ว.วิทย. มข. 40(3) 654-666 (2555) KKU Sci. J. 40(3) 654-666 (2012) แมลงน้ําวงศ!ไฮดรอบไซคิดี้ (อันดับไทรคอบเทอร-า) เพื่อเป2นตัวบ-งชี้ทางชีวภาพของคุณภาพน้ํา Hydropsychidae (Insecta: Trichoptera) as Bio-indicators of Water QuaLity แตงออน พรหมมิ1 บทคัดยอ การประเมินคุณภาพน้ําในแมน้ําและลําธารควรที่จะมีการใชปจจัยทางกายภาพ เคมีและชีวภาพควบคูกัน ไป ปจจัยทางชีวภาพที่มีศักยภาพในการประเมินคุณภาพน้ําในแหลงน้ําคือกลุมสัตว+ไมมีกระดูกสันหลังขนาดใหญที่ อาศัยอยูตามพื้นทองน้ํา โดยเฉพาะแมลงน้ําอันดับไทรคอบเทอรา ซึ่งเป3นกลุมสัตว+ที่มีความหลากหลายมากกลุม หนึ่งในแหลงน้ํา ระยะตัวออนของแมลงกลุมนี้ทุกชนิดอาศัยอยูในแหลงน้ํา เป3นองค+ประกอบหลักในแหลงน้ําและ เป3นตัวหมุนเวียนสารอาหารในแหลงน้ํา ระยะตัวออนของแมลงน้ํากลุมนี้จะตอบสนองตอปจจัยของสภาพแวดลอม ในแหลงน้ําทุกรูปแบบ ระยะตัวเต็มวัยอาศัยอยูบนบกบริเวณตนไมซึ่งไมไกลจากแหลงน้ํามากนัก หากินเวลา กลางคืน ความรูทางดานอนุกรมวิธานและชีววิทยาไมวาจะเป3นระยะตัวออนหรือตัวเต็มวัยของแมลงน้ําอันดับไทร คอบเทอราในประเทศแถบยุโรปตะวันตกและอเมริกาเหนือสามารถวินิจฉัยไดถึงระดับชนิด โดยเฉพาะแมลงน้ํา วงศ+ไฮดรอบไซคิดี้ มีการประยุกต+ใชในการติดตามตรวจสอบทางชีวภาพของคุณภาพน้ํา เนื่องจากชนิดของตัวออน แมลงน้ําวงศ+นี้มีความทนทานตอมลพิษในชวงกวางมากกวาแมลงน้ําชนิดอื่น ๆ 1สายวิชาวิทยาศาสตร+ คณะศิลปศาสตร+และวิทยาศาสตร+ มหาวิทยาลัยเกษตรศาสตร+ วิทยาเขตกําแพงแสน จ.นครปฐม 73140 E-mail: [email protected] บทความ วารสารวิทยาศาสตร+ มข. ปQที่ 40 ฉบับที่ 3 655 ABSTRACT Assessment on rivers and streams water quality should incorporate aspects of chemical, physical, and biological. Of all the potential groups of freshwater organisms that have been considered for -

Insecta: Trichoptera) from the Ecoregion Hellenic Western Balkans
Nat. Croat. Vol. 26(2), 2017 197 NAT. CROAT. VOL. 26 No 2 197-204 ZAGREB DECEMBER 31, 2017 original scientific paper / izvorni znanstveni rad DOI 10.20302/NC.2017.26.16 FIRST RECORD OF TRIAENODES BICOLOR (CURTIS, 1834) (INSECTA: TRICHOPTERA) FROM THE ECOREGION HELLENIC WESTERN BALKANS Halil Ibrahimi1, Ruzhdi Kuçi2*, Astrit Bilalli3 & Ermira Gashi1 1Department of Biology, Faculty of Mathematical and Natural Sciences, University of Prishtina “Hasan Prishtina”, “Mother Theresa” p.n., 10 000 Prishtinë, Republic of Kosovo 2Faculty of Education, University of Prishtina “Hasan Prishtina”, “Mother Teresa” p.n., 10 000 Prishtinë, Republic of Kosovo 3Faculty of Agribusiness, University of Peja “Haxhi Zeka”, “UÇK” street, 30 000 Pejë, Republic of Kosovo Ibrahimi, H., Kuçi, R., Bilalli, A. & Gashi, E.: First record of Triaenodes bicolor (Curtis, 1834) (Insecta: Trichoptera) from the Ecoregion Hellenic Western Balkans. Nat. Croat., Vol. 26, No. 2., 197-204, Zagreb, 2017. We collected adult caddisfly specimens with entomological nets and ultraviolet light traps monthly from May to November 2012 in Brezne Lake situated in Dragash Municipality. During this investigation we found the Leptocerid species Triaenodes bicolor for the first time in Kosovo; it is also the first record for Ecoregion 6, Hellenic Western Balkans. Additionally, this is the first record of the genus Triaenodes from Kosovo. In total seven males and three females of this species were found. Triaenodes bicolor is present all over the European continent but has been rarely sampled in southeastern Europe. Other taxa sympatric with Triaenodes bicolor in the investigated locality are: Hydropsyche instabilis, Hydropsyche spp., Plectrocnemia conspersa, Plectrocnemia spp., Micropterna nycterobia, Micropterna sequax, Limnephilus vittatus, Limnephilus auricula and Thremma anomalum. -
![A SUMMARY of the LIFE HISTORY and DISTRIBUTION of the SPRING CAVEFISH, Chologaster ]Gassizi, PUTNAM, with POPULATION ESTIMATES for the SPECIES in SOUTHERN ILLINOIS](https://docslib.b-cdn.net/cover/8157/a-summary-of-the-life-history-and-distribution-of-the-spring-cavefish-chologaster-gassizi-putnam-with-population-estimates-for-the-species-in-southern-illinois-288157.webp)
A SUMMARY of the LIFE HISTORY and DISTRIBUTION of the SPRING CAVEFISH, Chologaster ]Gassizi, PUTNAM, with POPULATION ESTIMATES for the SPECIES in SOUTHERN ILLINOIS
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Illinois Digital Environment for Access to Learning and Scholarship Repository A SUMMARY OF THE LIFE HISTORY AND DISTRIBUTION OF THE SPRING CAVEFISH, Chologaster ]gassizi, PUTNAM, WITH POPULATION ESTIMATES FOR THE SPECIES IN SOUTHERN ILLINOIS PHILIP W. SMITH -NORBERT M. WELCH Biological Notes No.104 Illinois Natural History Survey Urbana, Illinois • May 1978 State of Illinois Department of Registration and Education Natural History Survey Division A Summary of the life History and Distribution of the Spring Cavefish~ Chologasfer agassizi Putnam~ with Population Estimates for the Species in Southern Illinois Philip W. Smith and Norbert M. Welch The genus Chologaster, which means mutilated belly various adaptations and comparative metabolic rates of in reference to the absence of pelvic fins, was proposed all known amblyopsids. The next major contribution to by Agassiz ( 1853: 134) for a new fish found in ditches our knowledge was a series of papers by Hill, who worked and rice fields of South Carolina and described by him with the Warren County, Kentucky, population of spring as C. cornutus. Putnam (1872:30) described a second cave fish and described oxygen preferences ( 1968), food species of the genus found in a well at Lebanon, Tennes and feeding habits ( 1969a), effects of isolation upon see, naming it C. agassizi for the author of the generic meristic characters ( 1969b ), and the development of name. Forbes ( 1881:232) reported one specimen of squamation in the young ( 1971). Whittaker & Hill Chologaster from a spring in western Union County, ( 1968) described a new species of cestode parasite, nam Illinois, and noted that it differed from known specimens ing it Proteocephalus chologasteri. -
Comparative Osteology and Evolution of the Lungless Salamanders, Family Plethodontidae David B
COMPARATIVE OSTEOLOGY AND EVOLUTION OF THE LUNGLESS SALAMANDERS, FAMILY PLETHODONTIDAE DAVID B. WAKE1 ABSTRACT: Lungless salamanders of the family Plethodontidae comprise the largest and most diverse group of tailed amphibians. An evolutionary morphological approach has been employed to elucidate evolutionary rela tionships, patterns and trends within the family. Comparative osteology has been emphasized and skeletons of all twenty-three genera and three-fourths of the one hundred eighty-three species have been studied. A detailed osteological analysis includes consideration of the evolution of each element as well as the functional unit of which it is a part. Functional and developmental aspects are stressed. A new classification is suggested, based on osteological and other char acters. The subfamily Desmognathinae includes the genera Desmognathus, Leurognathus, and Phaeognathus. Members of the subfamily Plethodontinae are placed in three tribes. The tribe Hemidactyliini includes the genera Gyri nophilus, Pseudotriton, Stereochilus, Eurycea, Typhlomolge, and Hemidac tylium. The genera Plethodon, Aneides, and Ensatina comprise the tribe Pleth odontini. The highly diversified tribe Bolitoglossini includes three super genera. The supergenera Hydromantes and Batrachoseps include the nominal genera only. The supergenus Bolitoglossa includes Bolitoglossa, Oedipina, Pseudoeurycea, Chiropterotriton, Parvimolge, Lineatriton, and Thorius. Manculus is considered to be congeneric with Eurycea, and Magnadig ita is congeneric with Bolitoglossa. Two species are assigned to Typhlomolge, which is recognized as a genus distinct from Eurycea. No. new information is available concerning Haptoglossa. Recognition of a family Desmognathidae is rejected. All genera are defined and suprageneric groupings are defined and char acterized. Range maps are presented for all genera. Relationships of all genera are discussed. -

Endangered Species
FEATURE: ENDANGERED SPECIES Conservation Status of Imperiled North American Freshwater and Diadromous Fishes ABSTRACT: This is the third compilation of imperiled (i.e., endangered, threatened, vulnerable) plus extinct freshwater and diadromous fishes of North America prepared by the American Fisheries Society’s Endangered Species Committee. Since the last revision in 1989, imperilment of inland fishes has increased substantially. This list includes 700 extant taxa representing 133 genera and 36 families, a 92% increase over the 364 listed in 1989. The increase reflects the addition of distinct populations, previously non-imperiled fishes, and recently described or discovered taxa. Approximately 39% of described fish species of the continent are imperiled. There are 230 vulnerable, 190 threatened, and 280 endangered extant taxa, and 61 taxa presumed extinct or extirpated from nature. Of those that were imperiled in 1989, most (89%) are the same or worse in conservation status; only 6% have improved in status, and 5% were delisted for various reasons. Habitat degradation and nonindigenous species are the main threats to at-risk fishes, many of which are restricted to small ranges. Documenting the diversity and status of rare fishes is a critical step in identifying and implementing appropriate actions necessary for their protection and management. Howard L. Jelks, Frank McCormick, Stephen J. Walsh, Joseph S. Nelson, Noel M. Burkhead, Steven P. Platania, Salvador Contreras-Balderas, Brady A. Porter, Edmundo Díaz-Pardo, Claude B. Renaud, Dean A. Hendrickson, Juan Jacobo Schmitter-Soto, John Lyons, Eric B. Taylor, and Nicholas E. Mandrak, Melvin L. Warren, Jr. Jelks, Walsh, and Burkhead are research McCormick is a biologist with the biologists with the U.S. -

1 Southeastern Us Coastal Plain
1 In Press: 2000. Chapter XX. Pages __-__ in Richard C. Bruce, Robert J. Jaeger, and Lynn D. Houck, editors. The Biology of the Plethodontidae. Plenum Publishing Corp., New York, N. Y. SOUTHEASTERN U. S. COASTAL PLAIN HABITATS OF THE PLETHODONTIDAE: THE IMPORTANCE OF RELIEF, RAVINES, AND SEEPAGE D. BRUCE MEANS Coastal Plains Institute and Land Conservancy, 1313 N. Duval Street, Tallahassee, FL 32303, USA 1. INTRODUCTION Because the Coastal Plain is geologically and biologically a very distinct region of the southeastern United States -- the southern portion having a nearly subtropical climate -- the life cycles, ecology, and evolutionary relationships of its plethodontid salamanders may be significantly different from plethodontids in the Appalachians and Piedmont. Little attention has been paid, however, to Coastal Plain plethodontids. For instance, of the 133 scientific papers and posters presented at the four plethodontid salamander conferences held in Highlands, North Carolina, since 1972, only three (2%) dealt with Coastal Plain plethodontids. And yet, while it possesses fewer total species than the Appalachians and Piedmont, the Coastal Plain boasts of slightly more plethodontid diversity at the generic level. The genera Phaeognathus, Haideotriton, and Stereochilus are Coastal Plain endemics, whereas in the Appalachians and Piedmont Gyrinophilus is the only endemic genus unless one accepts Leurognathus apart from Desmognathus. In addition, Aneides is found in the Appalachians and not the Coastal Plain, but the genus also occurs in the western U. S. All the rest of the plethodontid genera east of the Mississippi River are shared by the Coastal Plain with the Appalachians and Piedmont. Geographically, the Coastal Plain includes Long Island in New York, and stretches south from the New Jersey Pine Barrens to include all of Florida, then west to Texas (Fig. -

Geological Survey of Alabama Calibration of The
GEOLOGICAL SURVEY OF ALABAMA Berry H. (Nick) Tew, Jr. State Geologist WATER INVESTIGATIONS PROGRAM CALIBRATION OF THE INDEX OF BIOTIC INTEGRITY FOR THE SOUTHERN PLAINS ICHTHYOREGION IN ALABAMA OPEN-FILE REPORT 0908 by Patrick E. O'Neil and Thomas E. Shepard Prepared in cooperation with the Alabama Department of Environmental Management and the Alabama Department of Conservation and Natural Resources Tuscaloosa, Alabama 2009 TABLE OF CONTENTS Abstract ............................................................ 1 Introduction.......................................................... 1 Acknowledgments .................................................... 6 Objectives........................................................... 7 Study area .......................................................... 7 Southern Plains ichthyoregion ...................................... 7 Methods ............................................................ 8 IBI sample collection ............................................. 8 Habitat measures............................................... 10 Habitat metrics ........................................... 12 The human disturbance gradient ................................... 15 IBI metrics and scoring criteria..................................... 19 Designation of guilds....................................... 20 Results and discussion................................................ 22 Sampling sites and collection results . 22 Selection and scoring of Southern Plains IBI metrics . 41 1. Number of native species ................................ -

Louisiana's Animal Species of Greatest Conservation Need (SGCN)
Louisiana's Animal Species of Greatest Conservation Need (SGCN) ‐ Rare, Threatened, and Endangered Animals ‐ 2020 MOLLUSKS Common Name Scientific Name G‐Rank S‐Rank Federal Status State Status Mucket Actinonaias ligamentina G5 S1 Rayed Creekshell Anodontoides radiatus G3 S2 Western Fanshell Cyprogenia aberti G2G3Q SH Butterfly Ellipsaria lineolata G4G5 S1 Elephant‐ear Elliptio crassidens G5 S3 Spike Elliptio dilatata G5 S2S3 Texas Pigtoe Fusconaia askewi G2G3 S3 Ebonyshell Fusconaia ebena G4G5 S3 Round Pearlshell Glebula rotundata G4G5 S4 Pink Mucket Lampsilis abrupta G2 S1 Endangered Endangered Plain Pocketbook Lampsilis cardium G5 S1 Southern Pocketbook Lampsilis ornata G5 S3 Sandbank Pocketbook Lampsilis satura G2 S2 Fatmucket Lampsilis siliquoidea G5 S2 White Heelsplitter Lasmigona complanata G5 S1 Black Sandshell Ligumia recta G4G5 S1 Louisiana Pearlshell Margaritifera hembeli G1 S1 Threatened Threatened Southern Hickorynut Obovaria jacksoniana G2 S1S2 Hickorynut Obovaria olivaria G4 S1 Alabama Hickorynut Obovaria unicolor G3 S1 Mississippi Pigtoe Pleurobema beadleianum G3 S2 Louisiana Pigtoe Pleurobema riddellii G1G2 S1S2 Pyramid Pigtoe Pleurobema rubrum G2G3 S2 Texas Heelsplitter Potamilus amphichaenus G1G2 SH Fat Pocketbook Potamilus capax G2 S1 Endangered Endangered Inflated Heelsplitter Potamilus inflatus G1G2Q S1 Threatened Threatened Ouachita Kidneyshell Ptychobranchus occidentalis G3G4 S1 Rabbitsfoot Quadrula cylindrica G3G4 S1 Threatened Threatened Monkeyface Quadrula metanevra G4 S1 Southern Creekmussel Strophitus subvexus -
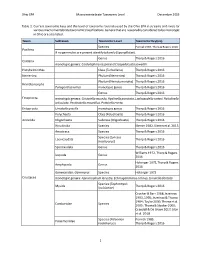
Ohio EPA Macroinvertebrate Taxonomic Level December 2019 1 Table 1. Current Taxonomic Keys and the Level of Taxonomy Routinely U
Ohio EPA Macroinvertebrate Taxonomic Level December 2019 Table 1. Current taxonomic keys and the level of taxonomy routinely used by the Ohio EPA in streams and rivers for various macroinvertebrate taxonomic classifications. Genera that are reasonably considered to be monotypic in Ohio are also listed. Taxon Subtaxon Taxonomic Level Taxonomic Key(ies) Species Pennak 1989, Thorp & Rogers 2016 Porifera If no gemmules are present identify to family (Spongillidae). Genus Thorp & Rogers 2016 Cnidaria monotypic genera: Cordylophora caspia and Craspedacusta sowerbii Platyhelminthes Class (Turbellaria) Thorp & Rogers 2016 Nemertea Phylum (Nemertea) Thorp & Rogers 2016 Phylum (Nematomorpha) Thorp & Rogers 2016 Nematomorpha Paragordius varius monotypic genus Thorp & Rogers 2016 Genus Thorp & Rogers 2016 Ectoprocta monotypic genera: Cristatella mucedo, Hyalinella punctata, Lophopodella carteri, Paludicella articulata, Pectinatella magnifica, Pottsiella erecta Entoprocta Urnatella gracilis monotypic genus Thorp & Rogers 2016 Polychaeta Class (Polychaeta) Thorp & Rogers 2016 Annelida Oligochaeta Subclass (Oligochaeta) Thorp & Rogers 2016 Hirudinida Species Klemm 1982, Klemm et al. 2015 Anostraca Species Thorp & Rogers 2016 Species (Lynceus Laevicaudata Thorp & Rogers 2016 brachyurus) Spinicaudata Genus Thorp & Rogers 2016 Williams 1972, Thorp & Rogers Isopoda Genus 2016 Holsinger 1972, Thorp & Rogers Amphipoda Genus 2016 Gammaridae: Gammarus Species Holsinger 1972 Crustacea monotypic genera: Apocorophium lacustre, Echinogammarus ischnus, Synurella dentata Species (Taphromysis Mysida Thorp & Rogers 2016 louisianae) Crocker & Barr 1968; Jezerinac 1993, 1995; Jezerinac & Thoma 1984; Taylor 2000; Thoma et al. Cambaridae Species 2005; Thoma & Stocker 2009; Crandall & De Grave 2017; Glon et al. 2018 Species (Palaemon Pennak 1989, Palaemonidae kadiakensis) Thorp & Rogers 2016 1 Ohio EPA Macroinvertebrate Taxonomic Level December 2019 Taxon Subtaxon Taxonomic Level Taxonomic Key(ies) Informal grouping of the Arachnida Hydrachnidia Smith 2001 water mites Genus Morse et al.