WO 2012/169644 Al 13 December 2012 (13.12.2012) P O P C T
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Sean Raspet – Molecules
1. Commercial name: Fructaplex© IUPAC Name: 2-(3,3-dimethylcyclohexyl)-2,5,5-trimethyl-1,3-dioxane SMILES: CC1(C)CCCC(C1)C2(C)OCC(C)(C)CO2 Molecular weight: 240.39 g/mol Volume (cubic Angstroems): 258.88 Atoms number (non-hydrogen): 17 miLogP: 4.43 Structure: Biological Properties: Predicted Druglikenessi: GPCR ligand -0.23 Ion channel modulator -0.03 Kinase inhibitor -0.6 Nuclear receptor ligand 0.15 Protease inhibitor -0.28 Enzyme inhibitor 0.15 Commercial name: Fructaplex© IUPAC Name: 2-(3,3-dimethylcyclohexyl)-2,5,5-trimethyl-1,3-dioxane SMILES: CC1(C)CCCC(C1)C2(C)OCC(C)(C)CO2 Predicted Olfactory Receptor Activityii: OR2L13 83.715% OR1G1 82.761% OR10J5 80.569% OR2W1 78.180% OR7A2 77.696% 2. Commercial name: Sylvoxime© IUPAC Name: N-[4-(1-ethoxyethenyl)-3,3,5,5tetramethylcyclohexylidene]hydroxylamine SMILES: CCOC(=C)C1C(C)(C)CC(CC1(C)C)=NO Molecular weight: 239.36 Volume (cubic Angstroems): 252.83 Atoms number (non-hydrogen): 17 miLogP: 4.33 Structure: Biological Properties: Predicted Druglikeness: GPCR ligand -0.6 Ion channel modulator -0.41 Kinase inhibitor -0.93 Nuclear receptor ligand -0.17 Protease inhibitor -0.39 Enzyme inhibitor 0.01 Commercial name: Sylvoxime© IUPAC Name: N-[4-(1-ethoxyethenyl)-3,3,5,5tetramethylcyclohexylidene]hydroxylamine SMILES: CCOC(=C)C1C(C)(C)CC(CC1(C)C)=NO Predicted Olfactory Receptor Activity: OR52D1 71.900% OR1G1 70.394% 0R52I2 70.392% OR52I1 70.390% OR2Y1 70.378% 3. Commercial name: Hyperflor© IUPAC Name: 2-benzyl-1,3-dioxan-5-one SMILES: O=C1COC(CC2=CC=CC=C2)OC1 Molecular weight: 192.21 g/mol Volume -

Human Olfactory Receptors
Human Olfactory Receptors: A journey from cell engineering for efficient in vitro functional assays to effective antagonists in human sensory assay Huysseune Sandra, Philippeau Magali, Moreau Cédric, Veithen Alex, Chatelain Pierre, Quesnel Yannick. ChemCom S.A., Route de Lennik 802, 1070 Anderlecht, Belgium. Cell lines Introduction: ChemCom's Entry ORs Literature data HEK293T HANA3 proprietary Many odorant compounds are perceived as unpleasant. They can be present in different contexts such as body-, home-, factory-, material-, or fabric-emitted 1 OR56A1 Adipietro et al., 2012 odors, so that humans are daily exposed to this olfactory pollution. In addition, odorant compounds can also taint food or beverages. Malodor and off-note 2 OR10J5 Saito et al., 2009 counteraction is a daily challenge for many different industries. 3 ORX116 - The first step of the odor perception corresponds to the interaction of olfactory receptors (ORs) with odorant molecules. Therefore, a selective inhibition of the 4 ORX069 - ORs by weakly odorant or odorless antagonists represents an innovative solution to malodor issues. The identification of such odor blockers requires an 5 OR10H5 - efficient technological platform to first fish out the receptors that interact with a malodor of interest and second, to screen libraries of potential antagonists of 6 OR8B3 Mainland et al., 2013 7 OR6P1 Mainland et al., 2013 these ORs. 8 ORX074B - 9 ORX126 - Materials and Methods: 10 ORX189 - 11 ORX081 - In vitro functional assay 12 ORX213 - Dilution-response analysis were performed in HEK293T, HANA3 and HEK293T-hRTP1S/hRTP2 cells using the CRE-luciferase reporter assay system. Briefly, each 13 OR1D2 Spehr et al., 2003 cell plated one day before was transfected with deorphanized ORs (identified at Chemcom S.A.) or empty vector plasmids using TransIT®-LT1 (Mirus) 14 OR7C1 Mainland et al., 2013 according to the manufacturer’s protocol. -

The Hypothalamus As a Hub for SARS-Cov-2 Brain Infection and Pathogenesis
bioRxiv preprint doi: https://doi.org/10.1101/2020.06.08.139329; this version posted June 19, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. The hypothalamus as a hub for SARS-CoV-2 brain infection and pathogenesis Sreekala Nampoothiri1,2#, Florent Sauve1,2#, Gaëtan Ternier1,2ƒ, Daniela Fernandois1,2 ƒ, Caio Coelho1,2, Monica ImBernon1,2, Eleonora Deligia1,2, Romain PerBet1, Vincent Florent1,2,3, Marc Baroncini1,2, Florence Pasquier1,4, François Trottein5, Claude-Alain Maurage1,2, Virginie Mattot1,2‡, Paolo GiacoBini1,2‡, S. Rasika1,2‡*, Vincent Prevot1,2‡* 1 Univ. Lille, Inserm, CHU Lille, Lille Neuroscience & Cognition, DistAlz, UMR-S 1172, Lille, France 2 LaBoratorY of Development and PlasticitY of the Neuroendocrine Brain, FHU 1000 daYs for health, EGID, School of Medicine, Lille, France 3 Nutrition, Arras General Hospital, Arras, France 4 Centre mémoire ressources et recherche, CHU Lille, LiCEND, Lille, France 5 Univ. Lille, CNRS, INSERM, CHU Lille, Institut Pasteur de Lille, U1019 - UMR 8204 - CIIL - Center for Infection and ImmunitY of Lille (CIIL), Lille, France. # and ƒ These authors contriButed equallY to this work. ‡ These authors directed this work *Correspondence to: [email protected] and [email protected] Short title: Covid-19: the hypothalamic hypothesis 1 bioRxiv preprint doi: https://doi.org/10.1101/2020.06.08.139329; this version posted June 19, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. -

Analysis of Single-Cell Transcriptomes Links Enrichment of Olfactory Receptors with Cancer Cell Differentiation Status and Prognosis
ARTICLE https://doi.org/10.1038/s42003-020-01232-5 OPEN Analysis of single-cell transcriptomes links enrichment of olfactory receptors with cancer cell differentiation status and prognosis Siddhant Kalra1,7, Aayushi Mittal1,7, Krishan Gupta 1,2, Vrinda Singhal1, Anku Gupta2, Tripti Mishra3, ✉ ✉ 1234567890():,; Srivatsava Naidu4, Debarka Sengupta 1,2,5,6 & Gaurav Ahuja 1 Ectopically expressed olfactory receptors (ORs) have been linked with multiple clinically- relevant physiological processes. Previously used tissue-level expression estimation largely shadowed the potential role of ORs due to their overall low expression levels. Even after the introduction of the single-cell transcriptomics, a comprehensive delineation of expression dynamics of ORs in tumors remained unexplored. Our targeted investigation into single malignant cells revealed a complex landscape of combinatorial OR expression events. We observed differentiation-dependent decline in expressed OR counts per cell as well as their expression intensities in malignant cells. Further, we constructed expression signatures based on a large spectrum of ORs and tracked their enrichment in bulk expression profiles of tumor samples from The Cancer Genome Atlas (TCGA). TCGA tumor samples stratified based on OR-centric signatures exhibited divergent survival probabilities. In summary, our compre- hensive analysis positions ORs at the cross-road of tumor cell differentiation status and cancer prognosis. 1 Department of Computational Biology, Indraprastha Institute of Information Technology-Delhi (IIIT-Delhi), Okhla, Phase III, New Delhi 110020, India. 2 Department of Computer Science and Engineering, Indraprastha Institute of Information Technology-Delhi (IIIT-Delhi), Okhla, Phase III, New Delhi 110020, India. 3 Pathfinder Research and Training Foundation, 30/7 and 8, Knowledge Park III, Greater Noida, Uttar Pradesh 201308, India. -

Us 2018 / 0305689 A1
US 20180305689A1 ( 19 ) United States (12 ) Patent Application Publication ( 10) Pub . No. : US 2018 /0305689 A1 Sætrom et al. ( 43 ) Pub . Date: Oct. 25 , 2018 ( 54 ) SARNA COMPOSITIONS AND METHODS OF plication No . 62 /150 , 895 , filed on Apr. 22 , 2015 , USE provisional application No . 62/ 150 ,904 , filed on Apr. 22 , 2015 , provisional application No. 62 / 150 , 908 , (71 ) Applicant: MINA THERAPEUTICS LIMITED , filed on Apr. 22 , 2015 , provisional application No. LONDON (GB ) 62 / 150 , 900 , filed on Apr. 22 , 2015 . (72 ) Inventors : Pål Sætrom , Trondheim (NO ) ; Endre Publication Classification Bakken Stovner , Trondheim (NO ) (51 ) Int . CI. C12N 15 / 113 (2006 .01 ) (21 ) Appl. No. : 15 /568 , 046 (52 ) U . S . CI. (22 ) PCT Filed : Apr. 21 , 2016 CPC .. .. .. C12N 15 / 113 ( 2013 .01 ) ; C12N 2310 / 34 ( 2013. 01 ) ; C12N 2310 /14 (2013 . 01 ) ; C12N ( 86 ) PCT No .: PCT/ GB2016 /051116 2310 / 11 (2013 .01 ) $ 371 ( c ) ( 1 ) , ( 2 ) Date : Oct . 20 , 2017 (57 ) ABSTRACT The invention relates to oligonucleotides , e . g . , saRNAS Related U . S . Application Data useful in upregulating the expression of a target gene and (60 ) Provisional application No . 62 / 150 ,892 , filed on Apr. therapeutic compositions comprising such oligonucleotides . 22 , 2015 , provisional application No . 62 / 150 ,893 , Methods of using the oligonucleotides and the therapeutic filed on Apr. 22 , 2015 , provisional application No . compositions are also provided . 62 / 150 ,897 , filed on Apr. 22 , 2015 , provisional ap Specification includes a Sequence Listing . SARNA sense strand (Fessenger 3 ' SARNA antisense strand (Guide ) Mathew, Si Target antisense RNA transcript, e . g . NAT Target Coding strand Gene Transcription start site ( T55 ) TY{ { ? ? Targeted Target transcript , e . -
Explorations in Olfactory Receptor Structure and Function by Jianghai
Explorations in Olfactory Receptor Structure and Function by Jianghai Ho Department of Neurobiology Duke University Date:_______________________ Approved: ___________________________ Hiroaki Matsunami, Supervisor ___________________________ Jorg Grandl, Chair ___________________________ Marc Caron ___________________________ Sid Simon ___________________________ [Committee Member Name] Dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy in the Department of Neurobiology in the Graduate School of Duke University 2014 ABSTRACT Explorations in Olfactory Receptor Structure and Function by Jianghai Ho Department of Neurobiology Duke University Date:_______________________ Approved: ___________________________ Hiroaki Matsunami, Supervisor ___________________________ Jorg Grandl, Chair ___________________________ Marc Caron ___________________________ Sid Simon ___________________________ [Committee Member Name] An abstract of a dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy in the Department of Neurobiology in the Graduate School of Duke University 2014 Copyright by Jianghai Ho 2014 Abstract Olfaction is one of the most primitive of our senses, and the olfactory receptors that mediate this very important chemical sense comprise the largest family of genes in the mammalian genome. It is therefore surprising that we understand so little of how olfactory receptors work. In particular we have a poor idea of what chemicals are detected by most of the olfactory receptors in the genome, and for those receptors which we have paired with ligands, we know relatively little about how the structure of these ligands can either activate or inhibit the activation of these receptors. Furthermore the large repertoire of olfactory receptors, which belong to the G protein coupled receptor (GPCR) superfamily, can serve as a model to contribute to our broader understanding of GPCR-ligand binding, especially since GPCRs are important pharmaceutical targets. -

SUPPLEMENTARY APPENDIX Whole Exome Sequencing Identifies Mutational Signatures of Vitreoretinal Lymphoma
SUPPLEMENTARY APPENDIX Whole exome sequencing identifies mutational signatures of vitreoretinal lymphoma Junwon Lee, 1* Borahm Kim, 2* Hyeonah Lee, 3 Heejung Park, 4 Suk Ho Byeon, 1 Jong Rak Choi, 2 Sung Chul Lee, 1 Seung-Tae Lee 2 and Christopher Seungkyu Lee 1 *JL and BK contributed equally as co-first authors 1Department of Ophthalmology, Severance Hospital, Institute of Vision Research, Yonsei University College of Medicine; 2Department of Laboratory Medi - cine, Yonsei University College of Medicine; 3Brain Korea 21 PLUS Project for Medical Science, Yonsei University and 4Department of pathology, Yonsei Uni - versity College of medicine, Seoul, South Korea Correspondence: CHRISTOPHER SEUNGKYU LEE - [email protected] SEUNG-TAE LEE - [email protected] doi:10.3324/haematol.2019.233783 Supplemental Data Supplemental Methods Supplemental Table 1 Supplemental Figure 1-5 Supplementary Methods Whole exome sequencing (WES) Genomic DNA was extracted using a QIAamp DNA Blood Mini Kit (Qiagen). The sequencing libraries for Exome‐sequencing were prepared using the Twist Human Core Exome Kit (Twist Bioscience). Paired‐end 100 bp read sequencing was performed on a NovaSeq system (Illumina). The paired‐end reads were mapped to the human genome (NCBI build 37) using BWA (version 0.7.12).1 The alignment was further refined by the functions of local realignment, base quality recalibration and indel realignment provided by GATK software 3.8-0. In order to identify single nucleotide variations (SNVs) and indels, we used HaplotypeCaller and Mutect2 from the GATK package (3.8-0), and VarScan2 (2.4.0). The results of these three algorithms were compared and merged.2-4 An R package, ExomeDepth (version 1.1.10), was used to detect exon- or gene-level copy number variation (CNV) in the target regions,5 followed by visualization using a base-level read depth normalization algorithm implemented in the DxSeq Analyzer (Dxome). -
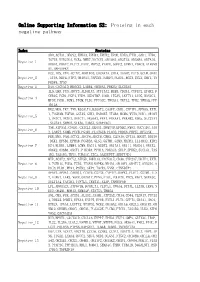
Online Supporting Information S2: Proteins in Each Negative Pathway
Online Supporting Information S2: Proteins in each negative pathway Index Proteins ADO,ACTA1,DEGS2,EPHA3,EPHB4,EPHX2,EPOR,EREG,FTH1,GAD1,HTR6, IGF1R,KIR2DL4,NCR3,NME7,NOTCH1,OR10S1,OR2T33,OR56B4,OR7A10, Negative_1 OR8G1,PDGFC,PLCZ1,PROC,PRPS2,PTAFR,SGPP2,STMN1,VDAC3,ATP6V0 A1,MAPKAPK2 DCC,IDS,VTN,ACTN2,AKR1B10,CACNA1A,CHIA,DAAM2,FUT5,GCLM,GNAZ Negative_2 ,ITPA,NEU4,NTF3,OR10A3,PAPSS1,PARD3,PLOD1,RGS3,SCLY,SHC1,TN FRSF4,TP53 Negative_3 DAO,CACNA1D,HMGCS2,LAMB4,OR56A3,PRKCQ,SLC25A5 IL5,LHB,PGD,ADCY3,ALDH1A3,ATP13A2,BUB3,CD244,CYFIP2,EPHX2,F CER1G,FGD1,FGF4,FZD9,HSD17B7,IL6R,ITGAV,LEFTY1,LIPG,MAN1C1, Negative_4 MPDZ,PGM1,PGM3,PIGM,PLD1,PPP3CC,TBXAS1,TKTL2,TPH2,YWHAQ,PPP 1R12A HK2,MOS,TKT,TNN,B3GALT4,B3GAT3,CASP7,CDH1,CYFIP1,EFNA5,EXTL 1,FCGR3B,FGF20,GSTA5,GUK1,HSD3B7,ITGB4,MCM6,MYH3,NOD1,OR10H Negative_5 1,OR1C1,OR1E1,OR4C11,OR56A3,PPA1,PRKAA1,PRKAB2,RDH5,SLC27A1 ,SLC2A4,SMPD2,STK36,THBS1,SERPINC1 TNR,ATP5A1,CNGB1,CX3CL1,DEGS1,DNMT3B,EFNB2,FMO2,GUCY1B3,JAG Negative_6 2,LARS2,NUMB,PCCB,PGAM1,PLA2G1B,PLOD2,PRDX6,PRPS1,RFXANK FER,MVD,PAH,ACTC1,ADCY4,ADCY8,CBR3,CLDN16,CPT1A,DDOST,DDX56 ,DKK1,EFNB1,EPHA8,FCGR3A,GLS2,GSTM1,GZMB,HADHA,IL13RA2,KIR2 Negative_7 DS4,KLRK1,LAMB4,LGMN,MAGI1,NUDT2,OR13A1,OR1I1,OR4D11,OR4X2, OR6K2,OR8B4,OXCT1,PIK3R4,PPM1A,PRKAG3,SELP,SPHK2,SUCLG1,TAS 1R2,TAS1R3,THY1,TUBA1C,ZIC2,AASDHPPT,SERPIND1 MTR,ACAT2,ADCY2,ATP5D,BMPR1A,CACNA1E,CD38,CYP2A7,DDIT4,EXTL Negative_8 1,FCER1G,FGD3,FZD5,ITGAM,MAPK8,NR4A1,OR10V1,OR4F17,OR52D1,O R8J3,PLD1,PPA1,PSEN2,SKP1,TACR3,VNN1,CTNNBIP1 APAF1,APOA1,CARD11,CCDC6,CSF3R,CYP4F2,DAPK1,FLOT1,GSTM1,IL2 -

Supplemental Table S18. Cellular Process Enrichment Analysis Output
Supplemental Table S18. -

Method for Identifying a Malodor Inhibitor Verfahren Zur Identifizierung Eines Geruchshemmers Procédé D’Identification D’Un Inhibiteur De Mauvaises Odeurs
(19) TZZ ___T (11) EP 2 718 718 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publication and mention (51) Int Cl.: of the grant of the patent: G01N 33/566 (2006.01) G01N 33/50 (2006.01) 29.04.2015 Bulletin 2015/18 (86) International application number: (21) Application number: 12728839.7 PCT/JP2012/064862 (22) Date of filing: 05.06.2012 (87) International publication number: WO 2012/169644 (13.12.2012 Gazette 2012/50) (54) METHOD FOR IDENTIFYING A MALODOR INHIBITOR VERFAHREN ZUR IDENTIFIZIERUNG EINES GERUCHSHEMMERS PROCÉDÉ D’IDENTIFICATION D’UN INHIBITEUR DE MAUVAISES ODEURS (84) Designated Contracting States: • SAITO, Naoko AL AT BE BG CH CY CZ DE DK EE ES FI FR GB Haga-gun GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO Tochigi 321-3497 (JP) PL PT RO RS SE SI SK SM TR (74) Representative: Vossius & Partner (30) Priority: 06.06.2011 JP 2011126637 Patentanwälte Rechtsanwälte mbB Siebertstrasse 3 (43) Date of publication of application: 81675 München (DE) 16.04.2014 Bulletin 2014/16 (56) References cited: (73) Proprietor: Kao Corporation WO-A1-2012/029922 WO-A2-2005/046632 Chuo-ku US-A1- 2003 207 337 Tokyo 103-8210 (JP) • H. ZHUANG ET AL: "Synergism of Accessory (72) Inventors: Factors in Functional Expression of Mammalian • KATO, Aya Odorant Receptors", JOURNAL OF BIOLOGICAL Haga-gun CHEMISTRY, vol. 282, no. 20, 1 May 2007 Tochigi 321-3497 (JP) (2007-05-01), pages 15284-15293, XP55036876, ISSN: 0021-9258, DOI: 10.1074/jbc.M700386200 Note: Within nine months of the publication of the mention of the grant of the European patent in the European Patent Bulletin, any person may give notice to the European Patent Office of opposition to that patent, in accordance with the Implementing Regulations. -

Characterization of the Genomic Features and Expressed Fusion Genes In
1 SUPPLEMENTARY INFORMATION (ONLINE SUPPORTING INFORMATION) Characterization of the genomic features and expressed fusion genes in micropapillary carcinomas of the breast Natrajan et al. Supplementary Methods Supplementary Figures S1-S6 Supplementary Tables S1-S7 2 SUPPLEMENTARY METHODS Tumor samples Two cohorts of micropapillary carcinomas (MPCs) were analyzed; the first cohort comprised 16 consecutive formalin fixed paraffin embedded (FFPE) MPCs, 11 pure and 5 mixed, which were retrieved from the authors' institutions (Table 1), and a second, validation cohort comprised 14 additional consecutive FFPE MPCs, retrieved from Molinette Hospital, Turin, Italy. Frozen samples were available from five out of the 16 cases from the first cohort of MPCs. As a comparator for the results of the Sequenom mutation profiling, a cohort of 16 consecutive IC-NSTs matched to the first cohort of 16 MPCs according to ER and HER2 status and histological grade were retrieved from a series of breast cancers previously analyzed by aCGH[1]. In addition, 14 IC-NSTs matched according to grade, and ER and HER2 status to tumors from the second cohort of 14 MPCs, and 48 grade 3 IC-NSTs were retrieved from Hospital La Paz, Madrid, Spain[1] (Supplementary Table S1). Power calculation For power calculations, we have assumed that if MPCs were driven by a recurrent fusion gene in a way akin to secretory carcinomas (which harbor the ETV6-NTRK3 fusion gene in >95% of cases[2-4]) or adenoid cystic carcinomas of the breast (which harbor the MYB-NFIB fusion gene in >90% of cases[5]), a ‘pathognomonic’ driver event would be present in at least ≥70% of cases (an estimate that is conservative). -

Supplementary Methods
doi: 10.1038/nature06162 SUPPLEMENTARY INFORMATION Supplementary Methods Cloning of human odorant receptors 423 human odorant receptors were cloned with sequence information from The Olfactory Receptor Database (http://senselab.med.yale.edu/senselab/ORDB/default.asp). Of these, 335 were predicted to encode functional receptors, 45 were predicted to encode pseudogenes, 29 were putative variant pairs of the same genes, and 14 were duplicates. We adopted the nomenclature proposed by Doron Lancet 1. OR7D4 and the six intact odorant receptor genes in the OR7D4 gene cluster (OR1M1, OR7G2, OR7G1, OR7G3, OR7D2, and OR7E24) were used for functional analyses. SNPs in these odorant receptors were identified from the NCBI dbSNP database (http://www.ncbi.nlm.nih.gov/projects/SNP) or through genotyping. OR7D4 single nucleotide variants were generated by cloning the reference sequence from a subject or by inducing polymorphic SNPs by site-directed mutagenesis using overlap extension PCR. Single nucleotide and frameshift variants for the six intact odorant receptors in the same gene cluster as OR7D4 were generated by cloning the respective genes from the genomic DNA of each subject. The chimpanzee OR7D4 orthologue was amplified from chimpanzee genomic DNA (Coriell Cell Repositories). Odorant receptors that contain the first 20 amino acids of human rhodopsin tag 2 in pCI (Promega) were expressed in the Hana3A cell line along with a short form of mRTP1 called RTP1S, (M37 to the C-terminal end), which enhances functional expression of the odorant receptors 3. For experiments with untagged odorant receptors, OR7D4 RT and S84N variants without the Rho tag were cloned into pCI.