Ethyl Alcohol
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Use of Solvents for Pahs Extraction and Enhancement of the Pahs Bioremediation in Coal- Tar-Contaminated Soils Pak-Hing Lee Iowa State University
Iowa State University Capstones, Theses and Retrospective Theses and Dissertations Dissertations 2000 Use of solvents for PAHs extraction and enhancement of the PAHs bioremediation in coal- tar-contaminated soils Pak-Hing Lee Iowa State University Follow this and additional works at: https://lib.dr.iastate.edu/rtd Part of the Environmental Engineering Commons Recommended Citation Lee, Pak-Hing, "Use of solvents for PAHs extraction and enhancement of the PAHs bioremediation in coal-tar-contaminated soils " (2000). Retrospective Theses and Dissertations. 13912. https://lib.dr.iastate.edu/rtd/13912 This Dissertation is brought to you for free and open access by the Iowa State University Capstones, Theses and Dissertations at Iowa State University Digital Repository. It has been accepted for inclusion in Retrospective Theses and Dissertations by an authorized administrator of Iowa State University Digital Repository. For more information, please contact [email protected]. INFORMATION TO USERS This manuscript has been reproduced from the microfilm master. UMI films the text directly from the original or copy submitted. Thus, some thesis and dissertation copies are in typewriter fece, while others may be from any type of computer printer. The quality of this reproduction is dependent upon the quaiity of the copy submitted. Broken or indistinct print colored or poor quality illustrations and photographs, print bleedthrough, substeindard margins, and improper alignment can adversely affect reproduction. In the unlilcely event that the author did not send UMI a complete manuscript and there are missing pages, these will be noted. Also, if unauthorized copyright material had to be removed, a note will indicate the deletion. -

Safer Cocktail for an Ethanol/Water/Ammonia Solvent
L A S C P LSC in Practice C P O C L Safer Cocktail for an Ethanol/Water/ K T I A I C Ammonia Solvent Mixture L S A Problem T A laboratory had been under pressure to move towards Using this mixture, the sample uptake capacity of I the newer generation of safer liquid scintillation ULTIMA-Flo AP, ULTIMA-Flo M and ULTIMA Gold O cocktails. Unfortunately, the sample composition of LLT (PerkinElmer 6013599, 6013579 and 6013377, one of their targets kept giving the researchers problems. respectively) at 20 °C was determined and N Before the lead researcher contacted PerkinElmer, the results were: they had tried PerkinElmer’s Opti-Fluor®, ULTIMA ULTIMA-Flo AP 4.00 mL in 10 mL cocktail N Gold LLT, and ULTIMA Gold XR. All of the safer O ™ ULTIMA-Flo M 4.25 mL in 10 mL cocktail cocktails could incorporate each of the constituents T as indicated by their sample capacity graphs. ULTIMA Gold LLT 4.25 mL in 10 mL cocktail E The problematic sample was an extraction solvent From these results, we observed that it was possible consisting of 900 mL ethanol, 50 mL ammonium to get 4.0 mL of the sample into all of these cocktails. hydroxide, 500 mL water and an enzyme containing In addition, we determined that it was not possible to 14C. The mixture had a pH in the range of 10 to 11. get 4 mL sample into 7 mL of any of these cocktails. To complete this work, we also checked for lumines- Discussion cence using 4 mL of sample in 10 mL of each cocktail. -

Perchlorates on Mars Enhance the Bacteriocidal Effects of UV Light
Edinburgh Research Explorer Perchlorates on Mars enhance the bacteriocidal effects of UV light Citation for published version: Wadsworth, J & Cockell, CS 2017, 'Perchlorates on Mars enhance the bacteriocidal effects of UV light', Scientific Reports, vol. 7, no. 1, 4662. https://doi.org/10.1038/s41598-017-04910-3 Digital Object Identifier (DOI): 10.1038/s41598-017-04910-3 Link: Link to publication record in Edinburgh Research Explorer Document Version: Publisher's PDF, also known as Version of record Published In: Scientific Reports General rights Copyright for the publications made accessible via the Edinburgh Research Explorer is retained by the author(s) and / or other copyright owners and it is a condition of accessing these publications that users recognise and abide by the legal requirements associated with these rights. Take down policy The University of Edinburgh has made every reasonable effort to ensure that Edinburgh Research Explorer content complies with UK legislation. If you believe that the public display of this file breaches copyright please contact [email protected] providing details, and we will remove access to the work immediately and investigate your claim. Download date: 02. Oct. 2021 www.nature.com/scientificreports OPEN Perchlorates on Mars enhance the bacteriocidal effects of UV light Jennifer Wadsworth & Charles S. Cockell Perchlorates have been identified on the surface of Mars. This has prompted speculation of what Received: 15 February 2017 their influence would be on habitability. We show that when irradiated with a simulated Martian UV Accepted: 22 May 2017 flux, perchlorates become bacteriocidal. At concentrations associated with Martian surface regolith, Published: xx xx xxxx vegetative cells of Bacillus subtilis in Martian analogue environments lost viability within minutes. -

Chemical Formula
Chemical Formula Jean Brainard, Ph.D. Say Thanks to the Authors Click http://www.ck12.org/saythanks (No sign in required) AUTHOR Jean Brainard, Ph.D. To access a customizable version of this book, as well as other interactive content, visit www.ck12.org CK-12 Foundation is a non-profit organization with a mission to reduce the cost of textbook materials for the K-12 market both in the U.S. and worldwide. Using an open-content, web-based collaborative model termed the FlexBook®, CK-12 intends to pioneer the generation and distribution of high-quality educational content that will serve both as core text as well as provide an adaptive environment for learning, powered through the FlexBook Platform®. Copyright © 2013 CK-12 Foundation, www.ck12.org The names “CK-12” and “CK12” and associated logos and the terms “FlexBook®” and “FlexBook Platform®” (collectively “CK-12 Marks”) are trademarks and service marks of CK-12 Foundation and are protected by federal, state, and international laws. Any form of reproduction of this book in any format or medium, in whole or in sections must include the referral attribution link http://www.ck12.org/saythanks (placed in a visible location) in addition to the following terms. Except as otherwise noted, all CK-12 Content (including CK-12 Curriculum Material) is made available to Users in accordance with the Creative Commons Attribution-Non-Commercial 3.0 Unported (CC BY-NC 3.0) License (http://creativecommons.org/ licenses/by-nc/3.0/), as amended and updated by Creative Com- mons from time to time (the “CC License”), which is incorporated herein by this reference. -

Fuel Properties Comparison
Alternative Fuels Data Center Fuel Properties Comparison Compressed Liquefied Low Sulfur Gasoline/E10 Biodiesel Propane (LPG) Natural Gas Natural Gas Ethanol/E100 Methanol Hydrogen Electricity Diesel (CNG) (LNG) Chemical C4 to C12 and C8 to C25 Methyl esters of C3H8 (majority) CH4 (majority), CH4 same as CNG CH3CH2OH CH3OH H2 N/A Structure [1] Ethanol ≤ to C12 to C22 fatty acids and C4H10 C2H6 and inert with inert gasses 10% (minority) gases <0.5% (a) Fuel Material Crude Oil Crude Oil Fats and oils from A by-product of Underground Underground Corn, grains, or Natural gas, coal, Natural gas, Natural gas, coal, (feedstocks) sources such as petroleum reserves and reserves and agricultural waste or woody biomass methanol, and nuclear, wind, soybeans, waste refining or renewable renewable (cellulose) electrolysis of hydro, solar, and cooking oil, animal natural gas biogas biogas water small percentages fats, and rapeseed processing of geothermal and biomass Gasoline or 1 gal = 1.00 1 gal = 1.12 B100 1 gal = 0.74 GGE 1 lb. = 0.18 GGE 1 lb. = 0.19 GGE 1 gal = 0.67 GGE 1 gal = 0.50 GGE 1 lb. = 0.45 1 kWh = 0.030 Diesel Gallon GGE GGE 1 gal = 1.05 GGE 1 gal = 0.66 DGE 1 lb. = 0.16 DGE 1 lb. = 0.17 DGE 1 gal = 0.59 DGE 1 gal = 0.45 DGE GGE GGE Equivalent 1 gal = 0.88 1 gal = 1.00 1 gal = 0.93 DGE 1 lb. = 0.40 1 kWh = 0.027 (GGE or DGE) DGE DGE B20 DGE DGE 1 gal = 1.11 GGE 1 kg = 1 GGE 1 gal = 0.99 DGE 1 kg = 0.9 DGE Energy 1 gallon of 1 gallon of 1 gallon of B100 1 gallon of 5.66 lb., or 5.37 lb. -

Liquichek Ethanol/Ammonia Control SERUM CHEMISTRY CONTROLS Liquichek Ethanol/Ammonia Control
Bio-Rad Laboratories SERUM CHEMISTRY CONTROLS Liquichek Ethanol/Ammonia Control SERUM CHEMISTRY CONTROLS Liquichek Ethanol/Ammonia Control A liquid control used to monitor the precision of Ethanol and Ammonia test procedures in the clinical laboratory. • Liquid • Normal, elevated and toxic concentrations of Ammonia • 2 year shelf life at 2–8°C • 20 day open-vial stability at 2–8°C Analytes Ammonia Ethanol Refer to the package insert of currently available lots for specific analyte and stability claims Ordering Information Cat # Description 544 Level 1 ..................................................................................... 6 x 3 mL 545 Level 2 ..................................................................................... 6 x 3 mL 546 Level 3 ..................................................................................... 6 x 3 mL 545X Trilevel MiniPak (1 of each level) .................................................................. 3 x 3 mL EQAS Independent QC Unity An independent, external assessment of laboratory Ongoing, proactive, unbiased daily QC that QC Data Management tools that help you create performance in comparison to your peers. helps identify errors as they occur or begin to trend. a strategy to reduce risk and streamline QC workflow. QCNet.com/eqas QCNet.com/independentqc QCNet.com/datamanagement Bio-Rad For further information, please contact the Bio-Rad office nearest you Laboratories, Inc. or visit our website at www.bio-rad.com/qualitycontrol Clinical Website www.bio-rad.com/diagnostics Australia -

Understanding Asthma
Understanding Asthma The Mount Sinai − National Jewish Health Respiratory Institute was formed by the nation’s leading respiratory hospital National Jewish Health, based in Denver, and top ranked academic medical center the Icahn School of Medicine at Mount Sinai in New York City. Combining the strengths of both organizations into an integrated Respiratory Institute brings together leading expertise in diagnosing and treating all forms of respiratory illness and lung disease, including asthma, chronic obstructive pulmonary disease (COPD), interstitial lung disease (ILD) and bronchiectasis. The Respiratory Institute is based in New York City on the campus of Mount Sinai. njhealth.org Understanding Asthma An educational health series from National Jewish Health IN THIS ISSUE What Is Asthma? 2 How Does Asthma Develop? 4 How Is Asthma Diagnosed? 5 What Are the Goals of Treatment? 7 How Is Asthma Managed? 7 What Things Make Asthma Worse and How Can You Control Them? 8 Nocturnal Asthma 18 Occupational Asthma 19 Medication Therapy 20 Monitoring Your Asthma 29 Using an Action Plan 33 Living with Asthma 34 Note: This information is provided to you as an educational service of National Jewish Health. It is not meant as a substitute for your own doctor. © Copyright 1998, revised 2014, 2018 National Jewish Health What Is Asthma? This booklet, prepared by National Jewish Health in Denver, is intended to provide information to people with asthma. Asthma is a chronic respiratory disease — sometimes worrisome and inconvenient — but a manageable condition. With proper understanding, good medical care and monitoring, you can keep asthma well controlled. That’s our treatment goal at National Jewish Health: to teach patients and families how to manage asthma, so that they can lead full and productive lives. -

Presentation on Key Ideas of Elementary Mathematics
Key Ideas of Elementary Mathematics Sybilla Beckmann Department of Mathematics University of Georgia Lesson Study Conference, May 2007 Sybilla Beckmann (University of Georgia) Key Ideas of Elementary Mathematics 1/52 US curricula are unfocused A Splintered Vision, 1997 report based on the TIMSS curriculum analysis. US state math curriculum documents: “The planned coverage included so many topics that we cannot find a single, or even a few, major topics at any grade that are the focus of these curricular intentions. These official documents, individually or as a composite, are unfocused. They express policies, goals, and intended content coverage in mathematics and the sciences with little emphasis on particular, strategic topics.” Sybilla Beckmann (University of Georgia) Key Ideas of Elementary Mathematics 2/52 US instruction is unfocused From A Splintered Vision: “US eighth grade mathematics and science teachers typically teach far more topic areas than their counterparts in Germany and Japan.” “The five surveyed topic areas covered most extensively by US eighth grade mathematics teachers accounted for less than half of their year’s instructional periods. In contrast, the five most extensively covered Japanese eighth grade topic areas accounted for almost 75 percent of their year’s instructional periods.” Sybilla Beckmann (University of Georgia) Key Ideas of Elementary Mathematics 3/52 Breaking the “mile-wide-inch-deep” habit Every mathematical skill and concept has some useful application has some connection to other concepts and skills So what mathematics should we focus on? Sybilla Beckmann (University of Georgia) Key Ideas of Elementary Mathematics 4/52 What focus? Statistics and probability are increasingly important in science and in the modern workplace. -

Cellulosic Ethanol Production Via Aqueous Ammonia Soaking Pretreatment and Simultaneous Saccharification and Fermentation Asli Isci Iowa State University
Iowa State University Capstones, Theses and Retrospective Theses and Dissertations Dissertations 2008 Cellulosic ethanol production via aqueous ammonia soaking pretreatment and simultaneous saccharification and fermentation Asli Isci Iowa State University Follow this and additional works at: https://lib.dr.iastate.edu/rtd Part of the Agriculture Commons, and the Bioresource and Agricultural Engineering Commons Recommended Citation Isci, Asli, "Cellulosic ethanol production via aqueous ammonia soaking pretreatment and simultaneous saccharification and fermentation" (2008). Retrospective Theses and Dissertations. 15695. https://lib.dr.iastate.edu/rtd/15695 This Dissertation is brought to you for free and open access by the Iowa State University Capstones, Theses and Dissertations at Iowa State University Digital Repository. It has been accepted for inclusion in Retrospective Theses and Dissertations by an authorized administrator of Iowa State University Digital Repository. For more information, please contact [email protected]. Cellulosic ethanol production via aqueous ammonia soaking pretreatment and simultaneous saccharification and fermentation by Asli Isci A dissertation submitted to the graduate faculty in partial fulfillment of the requirements for the degree of DOCTOR OF PHILOSOPHY Co-majors: Agricultural and Biosystems Engineering; Biorenewable Resources and Technology Program of Study Committee: Robert P. Anex, Major Professor D. Raj Raman Anthony L. Pometto III. Kenneth J. Moore Robert C. Brown Iowa State University Ames, Iowa -
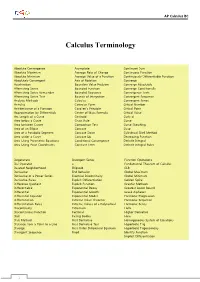
Calculus Terminology
AP Calculus BC Calculus Terminology Absolute Convergence Asymptote Continued Sum Absolute Maximum Average Rate of Change Continuous Function Absolute Minimum Average Value of a Function Continuously Differentiable Function Absolutely Convergent Axis of Rotation Converge Acceleration Boundary Value Problem Converge Absolutely Alternating Series Bounded Function Converge Conditionally Alternating Series Remainder Bounded Sequence Convergence Tests Alternating Series Test Bounds of Integration Convergent Sequence Analytic Methods Calculus Convergent Series Annulus Cartesian Form Critical Number Antiderivative of a Function Cavalieri’s Principle Critical Point Approximation by Differentials Center of Mass Formula Critical Value Arc Length of a Curve Centroid Curly d Area below a Curve Chain Rule Curve Area between Curves Comparison Test Curve Sketching Area of an Ellipse Concave Cusp Area of a Parabolic Segment Concave Down Cylindrical Shell Method Area under a Curve Concave Up Decreasing Function Area Using Parametric Equations Conditional Convergence Definite Integral Area Using Polar Coordinates Constant Term Definite Integral Rules Degenerate Divergent Series Function Operations Del Operator e Fundamental Theorem of Calculus Deleted Neighborhood Ellipsoid GLB Derivative End Behavior Global Maximum Derivative of a Power Series Essential Discontinuity Global Minimum Derivative Rules Explicit Differentiation Golden Spiral Difference Quotient Explicit Function Graphic Methods Differentiable Exponential Decay Greatest Lower Bound Differential -

Dehydrogenation of Ethanol to Acetaldehyde Over Different Metals Supported on Carbon Catalysts
catalysts Article Dehydrogenation of Ethanol to Acetaldehyde over Different Metals Supported on Carbon Catalysts Jeerati Ob-eye , Piyasan Praserthdam and Bunjerd Jongsomjit * Center of Excellence on Catalysis and Catalytic Reaction Engineering, Department of Chemical Engineering, Faculty of Engineering, Chulalongkorn University, Bangkok 10330, Thailand; [email protected] (J.O.-e.); [email protected] (P.P.) * Correspondence: [email protected]; Tel.: +66-2-218-6874 Received: 29 November 2018; Accepted: 27 December 2018; Published: 9 January 2019 Abstract: Recently, the interest in ethanol production from renewable natural sources in Thailand has been receiving much attention as an alternative form of energy. The low-cost accessibility of ethanol has been seen as an interesting topic, leading to the extensive study of the formation of distinct chemicals, such as ethylene, diethyl ether, acetaldehyde, and ethyl acetate, starting from ethanol as a raw material. In this paper, ethanol dehydrogenation to acetaldehyde in a one-step reaction was investigated by using commercial activated carbon with four different metal-doped catalysts. The reaction was conducted in a packed-bed micro-tubular reactor under a temperature range of 250–400 ◦C. The best results were found by using the copper doped on an activated carbon catalyst. Under this specified condition, ethanol conversion of 65.3% with acetaldehyde selectivity of 96.3% at 350 ◦C was achieved. This was probably due to the optimal acidity of copper doped on the activated carbon catalyst, as proven by the temperature-programmed desorption of ammonia (NH3-TPD). In addition, the other three catalyst samples (activated carbon, ceria, and cobalt doped on activated carbon) also favored high selectivity to acetaldehyde (>90%). -

Finding Hazards
FINDING HAZARDS OSHA 11 Finding Hazards 1 Osha 11 Finding Hazards 2 FINDING HAZARDS Learning Objectives By the end of this lesson, students will be able to: • Define the term “job hazard” • Identify a variety of health and safety hazards found at typical worksites where young people are employed. • Locate various types of hazards in an actual workplace. Time Needed: 45 Minutes Materials Needed • Flipchart Paper • Markers (5 colors per student group) • PowerPoint Slides: #1: Job Hazards #2: Sample Hazard Map #3: Finding Hazards: Key Points • Appendix A handouts (Optional) Preparing To Teach This Lesson Before you present this lesson: 1. Obtain a flipchart and markers or use a chalkboard and chalk. 2. Locate slides #1-3 on your CD and review them. If necessary, copy onto transparencies. 3. For the Hazard Mapping activity, you will need flipchart paper and a set of five colored markers (black, red, green, blue, orange) for each small group. Detailed Instructor’s Notes A. Introduction: What is a job hazard? (15 minutes) 1. Remind the class that a job hazard is anything at work that can hurt you, either physically or mentally. Explain that some job hazards are very obvious, but others are not. In order to be better prepared to be safe on the job, it is necessary to be able to identify different types of hazards. Tell the class that hazards can be divided into four categories. Write the categories across the top of a piece of flipchart paper and show PowerPoint Slide #1, Job Hazards. • Safety hazards can cause immediate accidents and injuries.